Abstract
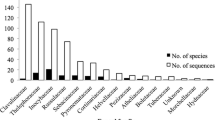
To examine the geographic patterns in Alnus-associated ectomycorrhizal (ECM) fungal assemblages and determine how they may relate to host plant biogeography, we studied ECM assemblages associated with two Alnus species (Alnus acuminata and Alnus jorullensis) in montane Mexico and compared them with Alnus-associated ECM assemblages located elsewhere in the Americas. ECM root samples were collected from four sites in Mexico (two per host species), identified with ITS and LSU rRNA gene sequences, and assessed using both taxon- (richness, diversity, evenness indices) and sequence divergence-based (UniFrac clustering and significance) analyses. Only 23 ECM taxa were encountered. Clavulina, an ECM lineage never before reported with Alnus, contained the dominant taxon overall. ECM assemblage structure varied between hosts, but UniFrac significance tests indicated that both associated with similar ECM lineage diversity. There was a strikingly high sequence similarity among a diverse array of the ECM taxa in Mexico and those in Alnus forests in Argentina, the United States, and Europe. The Mexican and United States assemblages had greater overlap than those present in Argentina, supporting the host–ECM fungi co-migration hypothesis from a common north temperate origin. Our results indicate that Alnus-associated ECM assemblages have clear patterns in richness and composition across a wide range of geographic locations. Additional data from boreal western North America as well as the eastern United States and Canada will be particularly informative in further understanding the co-biogeographic patterns of Alnus and ECM fungi in the Americas.



Similar content being viewed by others
References
Amend A, Seifert K, Samson R, Bruns TD (2010) Indoor fungal composition is geographically patterned and more diverse in temperate zones than in the tropics. Proc Natl Acad Sci USA 107:13478–13753
Beccera A, Zak M, Horton T, Micolini J (2005) Ectomycorrhizal and arbuscular mycorrhizal colonization of Alnus acuminata from Caliegua National Park (Argentina). Mycorrhiza 15:525–531
Benson D, Dawson J (2007) Recent advances in the biogeography and genecology of symbiotic Frankia and its host plants. Phys Plant 130:318–330
Bonito G, Gryganski A, Trappe JM, Vilgalys R (2010) A global meta-analysis of Tuber ITS rDNA sequences: species diversity, host associations and long-distance dispersal. Mol Ecol 19:4994–5008
Bormann B, Cromack K, Russell W (1994) Influences of red alder on soils and long-term ecosystem productivity. In: Hibbs D, DeBell D, Tarrant R (eds) The biology and management of red alder. Oregon State University Press, Corvallis, pp 47–56
Bruns T, Kennedy P (2009) Individuals, populations, communities and function: the growing field of ectomycorrhizal ecology. New Phytol 182:12–14
Chen Z, Li J (2004) Phylogenetics and biogeography of Alnus (Betulaceae) inferred from sequences of nuclear ribosomal DNA ITS region. Int J Plant Sci 165:325–335
Colwell R (2005) EstimateS: statistical estimation of species richness and shared species from samples. Version 7.5. User's guide and application. http://purl.oclc.org/estimates
Crane P (1989) Early fossil history and evolution of the Betulaceae. In: Crane P, Blackmore S (eds) Evolution, systematics, and fossil history of the Hamamelidae, vol 2, “Higher” Hamamelidae. Clarendon, Oxford, pp 87–116
den Bakker H, Zuccarello G, Kuyper T, Noordeloos M (2004) Evolution and host specificity in the ectomycorrhizal genus Leccinum. New Phytol 163:201–215
Dickie IA, Moyersoen B (2008) Towards a global view of ectomycorrhizal ecology. New Phytol 180:263–265
Edgar R (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Furlow J (1979) The systematics of the American species of Alnus (Betulaceae). Rhodora 81(1–121):151–248
Gardes M, Bruns T (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rust. Mol Ecol 2:113–118
Geml J, Tulloss R, Laursen GA, Sazanova NA, Taylor DL (2008) Evidence of strong inter- and intracontinental phylogenetic structure in Amanita muscaria, a wind-dispersed ectomycorrhizal basidiomycete. Mol Phylogenet Evol 48:694–701
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Halling RE (2001) Ectomycorrhizae: co-evolution, significance, and biogeography. Ann Mo Bot Gard 88:5–13
Halling RE (1989) A synopsis of Colombian boletes. Mycotaxon 34:93–113
Halling RE (1997) Boletaceae (Agaricales): latitudinal biodiversity and biological interactions in Costa Rica and Colombia. Rev Biol Trop 44(suppl 4):111–114
Hamady M, Lozupone C, Knight R (2010) Fast UniFrac: facilitating high-throughput phylogenetic analyses of microbial communities including analysis of pyrosequencing and PhyloChip data. ISME J 2010:17–27
Horton TR, Bruns TD (2001) The molecular revolution in ectomycorrhizal ecology: peeking into the black-box. Mol Ecol 10(8):1855–1871
Ishida T, Nara K, Hogetsu T (2007) Host effects on ectomycorrhizal fungal communities: insight from eight host species in mixed conifer–broadleaf forests. New Phytol 174:430–440
Kennedy P, Hill L (2010) A molecular and phylogenetic analysis of the structure and specificity of Alnus rubra ectomycorrhizal assemblages. Fungal Ecol 3:195–204
Kennedy PG, Izzo AD, Bruns TD (2003) There is high potential for the formation of common mycorrhizal networks between understorey and canopy trees in a mixed evergreen forest. J Ecol 91:1071–1080
Krebs C (1999) Ecological methodology. Benjamin/Cummings, Menlo Park
Lilleskov E, Hobbie E, Fahey T (2002) Ectomycorrhizal fungal taxa differing in response to nitrogen deposition also differ in pure culture organic nitrogen use and natural abundance of nitrogen isotopes. New Phytol 154:219–231
Lozupone C, Knight R (2008) Species divergence and the measurement of microbial diversity. FEMS Microbiol Rev 32:557–578
Matheny PB, Aime MC, Bougher NL, Buyck B, Desjardin DE, Horak E, Kropp BR, Lodge DJ, Soytong K, Trappe JM, Hibbett DS (2009) Out of the Palaeotropics? Historical biogeography and diversification of the cosmopolitan ectomycorrhizal mushroom family Inocybaceae. J Biogeogr 36:577–592
Molina R (1981) Ectomycorrhizal specificity in the genus Alnus. Can J Bot 59:325–334
Morris MH, Perez-Perez MA, Smith M, Bledsoe C (2009) Influence of host species on ectomycorrhizal communities associated with two co-occurring oaks (Quercus spp.) in a tropical cloud forest. FEMS Microbiol Ecol 69:274–287
Morris MH, Smith M, Rizzo D, Rejmanek M, Bledsoe C (2008) Contrasting ectomycorrhizal fungal communities on the roots of co-occurring oaks (Quercus spp.) in a California woodland. New Phytol 178:167–176
Moser M, Horak E (1975) Cortinarius Fries und. nahe verwande Gatttungen in Sudamerika. Beih Nova Hedwig 52:1–628
Moser M (2001) Some species of Cortinarius associated with Alnus. J des J E C 3:47–101
Murat C, Diez C, Luis P, Delaruelle C, Dupre C, Chevalier G, Bonfante P, Martin F (2004) Polymorphism at the ribosomal DNA ITS and its relations to postglacial re-colonization routes of the Perigord truffle Tuber melanosporum. New Phytol 164:401–411
Navarro E, Bousquet J, Moiroud A, Munive A, Piou D, Normand P (2003) Molecular phylogeny of Alnus (Betulaceae) inferred from nuclear ribosomal DNA ITS sequences. Plant Soil 254:207–217
Peay K, Kennedy P, Bruns T (2008) Fungal community ecology: a hybrid beast with a molecular master. Bioscience 58:799–810
Pickles BJ, Genney DR, Potts JM, Lennon J, Anderson I, Alexander I (2010) Spatial and temporal ecology of Scots pine ectomycorrhizas. New Phytol 186:755–768
Porter TM, Skillman JE, Moncalvo J-M (2008) Fruiting body and soil rDNA sampling detects complementary assemblage of Agaricomycotina (Basidiomycota, Fungi) in a hemlock-dominated forest plot in southern Ontario. Mol Ecol 17:3037–3050
Pritsch K, Beccera A, Polme S, Tedersoo L (2010) Description and identification of Alnus acuminata ectomycorrhizae from Argentinean alder stands. Mycologia 102:1263–1273
Pritsch K, Boyle H, Munch J, Buscot F (1997) Characterization and identification of black alder ectomycorrhizas by PCR/RFLP analyses of the rDNA internal transcribed spacer (ITS). New Phytol 137:357–369
Selosse M-A, Le Tacon F (1998) The land flora: a phototroph–fungus relationship? Trends Ecol Evol 13:15–20
Smith M, Douhan G, Kremier A, Rizzo D (2009) Are true multihost fungi the exception or the rule? Dominant ectomycorrhizal fungi on Pinus sabinia differ from those on co-occurring Quercus species. New Phytol 182:295–299
Smith S, Read D (2008) Mycorrhizal symbiosis. Academic, San Diego
Taberlet P, Gielly L, Pautou G, Bouvet J (1991) Universal primers for amplification of three non-coding regions of chloroplast DNA. Plant Mol Biol 17:1105–1109
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Tedersoo L, May TW, Smith M (2010) Ectomycorrhizal lifestyle in fungi: global diversity, distribution, and evolution of phylogenetic lineages. Mycorrhiza 20:217–263
Tedersoo L, Suvi T, Jairus T, Ostonen I, Polme S (2009) Revisiting ectomycorrhizal fungi of the genus Alnus: differential host specificity, diversity and determinants of the fungal community. New Phytol 182:727–735
Toljander J, Eberhardt U, Toljander Y, Paul L, Taylor A (2006) Species composition of an ectomycorrhizal fungal community along a local nutrient gradient in a boreal forest. New Phytol 170:873–884
Van der Heijden EW, Kuyper TW (2003) Ecological strategies of ectomycorrhizal of Salix repens: root manipulation versus root replacement. Oikos 103:668–680
Van der Heijden M, Sanders I (2002) Mycorrhizal Ecology. Springer, Berlin
Wu Q-X, Mueller GM, Lutzoni FM, Huang Y-Q, Guo S-Y (2000) Phylogenetic and biogeographic relationships of eastern Asian and eastern north American disjunct Suillus species (fungi) as inferred from nuclear ribosomal RNA ITS sequences. Mol Phylogenet Evol 17:37–47
Acknowledgements
The authors thank A. Kong for assistance in identifying the location of Malinche site 2, G. Williams-Linera and C. Alvarez for assistance in locating Naolinco site 1, L. Mora for assistance with soil analyses, and P-A. Moreau and P. B. Matheny for access to data presented in Fig. 3 as well as discussions about Alnus-associated taxa in the genus Inocybe. A. Amend, G. Bonilla, R. Molina, M. Palomino, K. Peay, F. Reverchon, E. Vellinga, M. Weber, and two reviewers provided many constructive comments on previous versions of this manuscript. Funding was provided by the Lewis & Clark College Student Academic Affairs Board (L.M. Higgins), a Lewis & Clark College Rogers Summer Research fellowship (L.M. Higgins and P.G. Kennedy), UNAM-PAPIIT IN218210 (R. Garibay-Orijel), a CIES Fulbright-Garcia Robles award (P.G. Kennedy), and National Science Foundation grant DEB-1020735 (P.G. Kennedy).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
S1
Location of the four field sites in Mexico. Map images modified from MapQuest and Google Earth. (PDF 4.00 mb)
S2
Results of UNITE and NCBI database queries for the ectomycorrhizal fungal (EMF) taxa sampled on Alnus acuminata and A. jorullensis. (PDF 613 kb)
S3
Results of UNITE and NCBI database queries for the EMF taxa sampled on P. montezumae. (PDF 610 kb)
S4
A weighted UniFrac Jackknife cluster analysis of the EMF assemblages present at the four study sites. Node values represent percent of Jackknife grouping based on 1,000 replications. UniFrac tests of significance revealed no significant differences in lineage evolution among sites despite the strong clustering of assemblages by host species. The scale bar indicates distance among clusters in UniFrac units (a distance of 0 means that the two sites have identical EMF lineages and a distance of 1 means that the two sites contain mutually exclusive EMF lineages). Very similar results were obtained for an unweighted UniFrac analysis of the same data. (PDF 123 kb)
S5
Pair-wise comparisons between various EMF taxa in the genera Tomentella, Cortinarius, Lactarius, and Inocybe. Comparisons were conducted on using the alignment function in NCBI BLAST(http://www.ncbi.nlm.nih.gov/blast/Blast.cgi?PAGE=Nucleotides& (PDF 401 kb) PROGRAM=blastn&BLAST_PROGRAMS=megaBlast&PAGE_TYPE=BlastSearch& HOW_DEFAULTS=on&BLAST_SPEC=blast2seq &QUERY=&SUBJECTS=). Taxa are designated here by their GenBank, UNITE, or collection number and correspond with those presented in Fig. 3. Values represent percent of similarity with the number of matching bases out of the total in parentheses. (PDF 1.22 mb)
Rights and permissions
About this article
Cite this article
Kennedy, P.G., Garibay-Orijel, R., Higgins, L.M. et al. Ectomycorrhizal fungi in Mexican Alnus forests support the host co-migration hypothesis and continental-scale patterns in phylogeography. Mycorrhiza 21, 559–568 (2011). https://doi.org/10.1007/s00572-011-0366-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-011-0366-2