Abstract
Based on the intrinsic electrochemical features of brucine integrated with carbon nanotubes (brucine/SWNTs), dimeric quinoid brucine was electrochemically generated by electroactivation of a brucine/SWNTs-modified GC electrode and used as a novel electrocatalyst for efficient electro-oxidation of hydroxylamine (HA). The electrocatalytic activity was investigated with cyclic voltammetry in the range pH 2.0 to pH 11.0, and the best electrocatalytic performance of the electrocatalyst was obtained at pH 10.0. By taking advantage of the electrocatalytic activity of the dimeric quinoid brucine toward HA, we have developed an electrochemical sensor for HA measurements based on a brucine/SWNTS-modified GC electrode using amperometry with the applied potential of + 0.1 V (vs. Ag/AgCl). Under the optimized conditions, the current response toward HA concentration shows a linear relationship in the dynamic ranges of 0.1–10 μM and 10–1000 μM with a detection limit of 0.021 μM based on the 3σ criterion. The sensor was used to assay HA in pharmaceuticals including hydroxyurea tablets and pralidoxime iodide injections with satisfactory results. The spike-and-recovery for samples of tap water (n = 9) and lake water (n = 9) was within 97.17–100.16%.

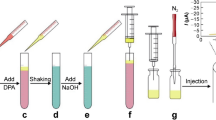
Schematic illustration of electrochemical sensing of hydroxylamine (HA) enabled by integrating brucine with single-walled carbon nanotube (brucine/SWNTs) based on electro-activation of brucine/SWNTs-modified GC electrode







Similar content being viewed by others
References
Gross P (1985) Biologic activity of hydroxylamine: a review. Crit Rev Toxicol 14:87–99
Liu GF, Li XC, Han BJ, Chen LW, Zhu LN, Campos LC (2017) Efficient degradation of sulfamethoxazole by the Fe(II)/HSO5− process enhanced by hydroxylamine: efficiency and mechanism. J Hazard Mater 322:461–446
Coumes CCD, Chopin-Dumas J, Devisme F (2001) Kinetics of the reaction of methyl iodide with hydroxylamine in an aqueous solution within the framework of nuclear spent fuel reprocessing. Ind Eng Chem Res 40:3721–3731
Park H, Kim DH (2018) Dissociative adsorption of a multifunctional compound on a semiconductor surface: a theoretical study of the adsorption of hydroxylamine on Ge (100). Phys Chem Chem Phys 20:15335–15343
Caranto JD, Lancaster KM (2018) Nitric oxide is an obligate bacterial nitrification intermediate produced by hydroxylamine oxidoreductase. Proc Natl Acad Sci U S A 115:EB325–EB325
Kobayashi S, Hira D, Yoshida K, Toyofuku M, Shida Y, Ogasawara W, Yamaguchi T, Araki N, Oshiki M (2018) Nitric oxide production from nitrite reduction and hydroxylamine oxidation by copper-containing dissimilatory nitrite reductase (NirK) from the aerobic ammonia-oxidizing archaeon, nitrososphaera viennensis. Microbes Environ 33:428–434
Hirasawa M, Tripathy JN, Sommer F, Somasundaram R, Chung JS, Nestander M, Kruthiventi M, Zabet-Moghaddam M, Johnson MK, Merchant SS, Allen JP, Knaff DB (2010) Enzymatic properties of the ferredoxin-dependent nitrite reductase from Chlamydomonas reinhardtii. Evidence for hydroxylamine as a late intermediate in ammonia production. Photosynth Res 103:67–77
Taylor AT, Crist SB, Jones OW (1970) Alteration of native DNA transcription by the mutagen hydroxylamine. Cancer Res 30:3095–3099
Troll W, Belman S, Levine E (1963) The effect of metabolites of 2-naphthylamine and the mutagen hydroxylamine on the thermal stability of DNA and polyribonucleotides. Cancer Res 23:841–847
Afkhami A, Madrakian T, Maleki A (2005) Spectrophotometric determination of hydroxylamine and nitrite in mixture in water and biological samples after micelle-mediated extraction. Anal Biochem 347:162–164
Song M, Wu S, Lu PB, Qiao YN, Hang TJ (2016) A selective and sensitive pre-column derivatization HPLC method for the trace analysis of genotoxic impurity hydroxylamine in active pharmaceutical ingredients. Anal Methods 8:8352–8361
Li J, Peng ZQ, Wang EK (2018) Tackling grand challenges of the 21st century with electroanalytical chemistry. J Am Chem Soc 140:10629–10638
Minteer SD (2018) Advances in electroanalytical chemistry. J Am Chem Soc 140:2701–2703
Rezaei B, Ensafi AA, Jamshidi-mofrad E (2015) A sensitive electrochemical sensor for hydroxylamine determination: using multiwall carbon nanotube paste electrode (MWCNTPE) and promazine hydrochloride as homogenous mediator. Sensor Actuat B-Chem 211:138–145
Ardakani MM, Karimi M, Mirdehghan S, Zare M, Mazidi R (2008) Electrocatalytic determination of hydroxylamine with alizarin red S as a homogenous mediator on the glassy carbon electrode. Sensor Actuat B-Chem 132:52–59
Li J, Lin XQ (2007) Electrocatalytic oxidation of hydrazine and hydroxylamine at gold nanoparticle-polypyrrole nanowire modified glassy carbon electrode. Sensor Actuat B-Chem 126:527–535
Premalatha S, Chandrasekaran M, Bapu G (2017) Preparation of cobalt-RuO2 nanocomposite modified electrode for highly sensitive and selective determination of hydroxylamine. Sensor Actuat B-Chem 252:375–384
Benvidi A, Jahanbani S, Akbari A, Zare HR (2015) Simultaneous determination of hydrazine and hydroxylamine on a magnetic bar carbon paste electrode modified with reduced graphene oxide/Fe3O4 nanoparticles and a heterogeneous mediator. J Electroanal Chem 758:68–77
Yang YJ, Yao C, Li WK (2017) Immobilization of phosphotungstic acid on multiwalled carbon nanotubes with cetyltrimethyl ammonium bromide as the molecular linker for enhanced oxidation of hydroxylamine. J Electroanal Chem 799:386–392
Tajik S, Mahmoudi-Moghaddam H, Beitollahi H (2019) Screen-printed electrode modified with La3+-doped Co3O4 nanocubes for electrochemical determination of hydroxylamine. J Electrochem Soc 166:B402–B406
Hemmati MH, Ekrami-Kakhki MS (2018) Electrocatalytic oxidation of hydroxylamine on graphite screen printed electrode modified with iron oxide nanoparticles. Anal Bioanal Electrochem 10:439–449
Zhang CH, Wang GF, Liu M, Feng YH, Zhang ZD, Fang B (2010) A hydroxylamine electrochemical sensor based on electrodeposition of porous ZnO nanofilms onto carbon nanotubes films modified electrode. Electrochim Acta 55:2835–2840
Rostami S, Azizi SN, Ghasemi S (2017) Simultaneous electrochemical determination of hydrazine and hydroxylamine by CuO doped in ZSM-5 nanoparticles as a new amperometric sensor. New J Chem 41:13712–13723
Zhao X, Bai J, Bo XJ, Guo LP (2019) A novel electrochemical sensor based on 2D CuTCPP nanosheets and platelet ordered mesoporous carbon composites for hydroxylamine and chlorogenic acid. Anal Chim Acta 1075:71–80
Nyasulu FW (1990) Brucine. 1. Electrochemistry of brucine. Electroanalysis 2:327–331
Savalia R, Chatterjee S (2017) Sensitive detection of brucine an anti-metastatic drug for hepatocellular carcinoma at carbon nanotubes-nafion composite based biosensor. Biosens Bioelectron 98:371–377
Zhao LJ, Zhao FQ, Zeng BZ (2014) Preparation of surface-imprinted polymer grafted with water-compatible external layer via RAFT precipitation polymerization for highly selective and sensitive electrochemical determination of brucine. Biosens Bioelectron 60:71–76
Wendlandt AE, Stahl SS (2015) Quinone-catalyzed selective oxidation of organic molecules. Angew Chem Int Edit 54:14638–14658
Regino MCS, Brajter-Toth A (1999) Real time characterization of catalysis by on-line electrochemistry mass spectrometry. Investigation of quinone electrocatalysis of amine oxidation. Electroanalysis 11:374–379
Costentin C, Robert M, Saveant JM (2010) Update 1 of: electrochemical approach to the mechanistic study of proton-coupled electron transfer. Chem Rev 110:PR1–PR40
Wang SF, Xie F, Hu RF (2007) Electrochemical study of brucine on an electrode modified with magnetic carbon-coated nickel nanoparticles. Anal Bioanal Chem 387:933–939
Masini J, Aragon S, Nyasulu F (1997) Electrochemistry of brucine .2. The brucine based determination of nitrate. Anal Chem 69:1077–1081
Zlotos DP, Buller S, Holzgrabe U, Mohr K (2003) Bisquaternary dimers of strychnine and brucine. A new class of potent enhancers of antagonist binding to muscarinic M2 receptors. Biorg Med Chem 11:2627–2634
Zhang LL, Liu YC, Wang YM, Xu M, Hu XY (2018) UV-Vis spectroscopy combined with chemometric study on the interactions of three dietary flavonoids with copper ions. Food Chem 263:208–215
Xu LY, Tian HW, Yao HP, Shi TS (2018) New kinetic and mechanistic findings in the oxidation of hydroxylamine by cerium(IV) in perchloric acid media. Int J Chem Kinet 50:856–862
Makarycheva-Mikhailova AV, Stanbury DM, McKee ML (2007) Kinetic isotope effect and extended pH-dependent studies of the oxidation of hydroxylamine by hexachloroiridate(IV): ET and PCET. J Phys Chem B 111:6942–6948
Funding
This work was financially supported by the National Natural Science Foundation of China (Grant No. 21505002), 2017 Anhui Province Foundation for the Returned Scholars (No. 19), Excellent Young Talents Fund Program of Higher Education Institutions of Anhui Province (Grant No. gxyq2018026), and Provincial Natural Science Foundation of Anhui (Grant No.1708085QH188).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no competing interest.
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 502 kb)
Rights and permissions
About this article
Cite this article
Xi, W., Zhai, J., Zhang, Y. et al. Integrating brucine with carbon nanotubes toward electrochemical sensing of hydroxylamine. Microchim Acta 187, 343 (2020). https://doi.org/10.1007/s00604-020-04315-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-04315-6