Abstract
Background
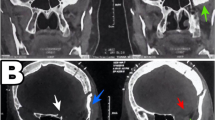
Surgical management of giant medial sphenoid meningiomas (≥5 cm in maximum dimension) is extremely challenging due to their intimate relationship with vital neural structures like the optic nerve, cranial nerves of the cavernous sinus and the cavernous internal carotid artery. Their surgical management is presented incorporating a radiological scoring system that predicts the grade of tumour excision.
Materials and methods
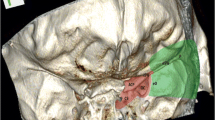
20 patients of giant medial sphenoidal wing meningioma (maximum tumour dimension range: 5.2 to 9.5 cm; mean maximum dimension = 6.12 ± 1.06 cm) with mainly visual and extraocular movement deficits, and raised intracranial pressure, underwent surgery. A preoperative radiological scoring system (range 1–12) was proposed considering tumour volume (using Kawamoto’s method); extension into the surrounding surgical corridors; extent of cavernous sinus invasion (based on the tumour relationship to the cavernous internal carotid artery); associated hyperostosis and/or >50% calcification; and, associated brain oedema. Both the conventional frontotemporal craniotomy (n = 13) and its extension to orbitozygomatic osteotomy (n = 7) were utilized. The cavernous sinus was explored in 4 patients and the hyperostotic sphenoid ridge drilled in five patients.
Findings
Total excision was achieved in nine patients; small tumour remnants within the cavernous sinus, interpeduncular fossa or suprasellar cistern were left in eight patients; and less than 10% of tumour was left in three patients. A patient with a completely calcified meningioma died due to myocardial infarction. When the preoperative radiological score was ≥7, there was considerable difficulty in achieving total tumour excision. A mean follow of 17.58 ± 15.05 months revealed improvement in visual acuity/field defects in three, stabilisation in 11, and deterioration of ipsilateral visual acuity in five patients. Symptoms of raised pressure, cognitive dysfunction, aphasia and proptosis showed improvement.
Conclusion
A relatively conservative approach to these extensive lesions resulted in good outcome in a majority of our patients. Both the standard as well as skull base approaches may be utilized for successful removal of giant medial sphenoidal wing meningiomas. A preoperative radiological score of ≥7 predicts a greater degree of difficulty in achieving complete surgical extirpation.




Similar content being viewed by others
References
Abdel Aziz KM, Froliech SC, Cohen PL, Sanam A, Keller IT, Van Loveran HR (2002) The one piece orbitozygomatic approach: the MaCarty burr hole and the inferior orbital fissure as keys to technique and application. Acta Neurochir (Wien) 144:15–42
Al-Mefty O (1990) Clinoidal meningiomas. J Neurosurg 73:840–849
Al-Mefty O, Anand VK (1990) Zygomatic approach to skull-base lesions. J Neurosurg 73:668–673
Al-Mefty O, Ayoubi S, Smith RR (1991) Direct surgery of the cavernous sinus: patient selection. Acta Neurochir Suppl (Wien) 53:117–121
Al-Mefty O, Smith RR (1988) Surgery of tumours invading the cavernous sinus. Surg Neurol 30:370–381
Bonnal J, Thibaut A, Brotchi J, Born J (1980) Invading meningiomas of the sphenoid ridge. J Neurosurg 53:587–599
Cook A (1971) Total removal of large global meningiomas of the medial aspect of the sphenoid ridge: technical note. J Neurosurg 34:107–113
Cushing H, Eisenhardt L (1938) Meningiomas: their classification, regional behavior, life history, and surgical end results. Charles C Thomas, Springfield, pp 311–319
Cusimano M, Sekhar LN, Sen CN, Pomonis S, Wright DC, Biglan A, Jennetta P (1995) The results of surgery for benign tumours of the cavernous sinus. Neurosurgery 37:1–10
Day JD (2000) Skull base surgical techniques for large sphenocavernous meningiomas: technical note. Neurosurgery 46:754–760
Day JD, Ginnotta SL, Fukushima T (1994) Extradural temporopolar approach to lesions of the upper basilar artery and infrachiasmatic regions. J Neurosurg 81:230–235
De Jesus O, Sekhar LN, Parikh HK, Wright DC, Wagner DP (1996) Long-term follow-up of patients with meningiomas involving the cavernous sinus: recurrence, progression, and quality of life. Neurosurgery 39:915–920
DeMonte F, Smith H, Al-Mefty O (1994) Outcome of aggressive removal of cavernous sinus meningiomas. J Neurosurg 81:245–251
Dolenc VV (1979) Microsurgical removal of large sphenoidal bone meningiomas. Acta Neurochir (Wien) 28(Suppl):391–396
Dolenc VV (1983) Direct microsurgical repair of intracavernous vascular lesions. J Neurosurg 58:824–831
Dolenc VV (1989) Anatomy of the cavernous sinus. In: Dolenc VV (ed) Anatomy and surgery of the cavernous sinus. Springer, Berlin Heidelberg New York, pp 3–7
Dolenc VV, Kregar T, Ferluga M, Fettich M, Morina A (1987) Treatment of tumours invading the cavernous sinus. In: Dolenc VV (ed) The cavernous sinus: a multidisciplinary approach to tumorous and vascular lesions. Springer, Berlin Heidelberg New York, pp 377–391
Dufour H, Murracciole X, Metellus P, Regis J, Chinot O, Grisoli F (2001) Long-term tumour control and functional outcome in patients with cavernous sinus meningiomas treated by radiotherapy with or without previous surgery: is there an alternative to aggressive tumour removal? Neurosurgery 48:285–294
Duma CM, Lunsford LD, Kondziolka D, Harsh GR IV, Flickinger JC (1993) Stereotactic radiosurgery of cavernous sinus meningiomas as an addition or alternative to microsurgery. Neurosurgery 32:699–705
El-Kalliny M, van Loveren HR, Keller JT, Tew JM Jr (1992) Tumours of the lateral wall of the cavernous sinus. J Neurosurg 77:508–514
Hakuba A, Tanaka K, Suzuki T, Nishimura S (1989) A combined orbitozygomatic infratemporal epidural and subdural approach for lesions involving the entire cavernous sinus. J Neurosurg 71:699–704
Hirsch WL, Sekhar LN, Lanzino G, Pomonis S, Sen CN (1993) Meningiomas involving the cavernous sinus: value of imaging for predicting surgical complications. AJR Am J Roentgenol 160:1083–1088
Inoue T, Rhoton AL Jr, Theele D, Barry ME (1990) Surgical approaches to the cavernous sinus: a microsurgical study. Neurosurgery 26:903–932
Kawamoto Y, Uozumi T, Kiya K, Fujioka Y, Kurisu K, Kawamoto K, Sugiyama K (1995) Clinicopathologic growth factors of acoustic neuromas. Surg Neurol 43:546–52
Kawase T, Toya S, Shiobara R, Kimura C, Nakajima H (1987) Skull base approaches for the meningiomas invading the cavernous sinus. In: Dolenc VV (ed) The cavernous sinus: multidisciplinary approach to vascular and tumorous lesions. Springer-Verlag, Vienna, pp 346–354
Kleinpeter G, Bock F (1990) Invasion of the cavernous sinus by medial sphenoid meningioma: “Radical” surgery and recurrence. Acta Neurochir (Wien) 103:87–91
Klink D, Sampath P, Miller NR, Brem H, Long DM (2000) Long-term visual outcome after nonradical microsurgery in patients with parasellar and cavernous sinus meningiomas. Neurosurgery 47:24–31
Knosp E, Perneczky A, Koos WT, Fries G, Matula C (1996) Meningiomas of the space of the cavernous sinus. Neurosurgery 38:434–442
Kotapka MJ, Kalia KK, Martinez AJ, Sekhar LN (1994) Infiltration of the carotid artery by cavernous sinus meningiomas. J Neurosurg 81:252–255
Krisht A, Barnett DW, Barrow DL, Bonner G (1994) The blood supply of the intracavernous cranial nerves: an anatomic study. Neurosurgery 34:275–279
Lanzino G, Hirsch WL, Pomonis S, Sen CN, Sekhar LN (1992) Cavernous sinus tumours: neuroradiologic and neurosurgical considerations on 150 operated cases. J Neurosurg Sci 36:183–196
Larsen JJ, van Loveren HR, Balko MG, Tew JM Jr (1995) Evidence of meningioma infiltration into cranial nerves: clinical implications for cavernous sinus meningiomas. J Neurosurg 83:596–599
Lee JH, Jeun SS, Evans J, Kosmorsky G (2001) Surgical management of clinoidal meningiomas. Neurosurgery 48:1012–1021
Maroon J, Kennerdell HS, Vidovich DV, Abla A, Sternau L (1994) Recurrent spheno-orbital meningioma. J Neurosurg 80:202–208
Mathiesen T, Lindquist C, Kihlstorm L, Karlsson B (1996) Recurrence of cranial base meningiomas. Neurosurgery 39:2–7
McDermott MW, Durity FA, Rootman J, Woodhurst WB (1990) Combined frontotemporal-orbitozygomatic approach for tumours of the sphenoid wing and orbit. Neurosurgery 26(1):107–116
Miralbell R, Lingwood RM, De La Monte S, Mirinmanoff RO (1992) The role of radiotherapy in the subtotally resected meningiomas. J Neurooncol 13:157–164
Nakamura M, Roser F, Jakobs C, Vorkapic P, Samii M (2006) Medial sphenoid wing meningiomas: clinical outcome and recurrence rate. Neurosurgery 58:626–629
Ojemann R (1992) Medial sphenoid wing (clinoidal) meningiomas. Clin Neurosurgery 40:321–383
O’Sullivan MG, van Loveren HR, Tew JM Jr (1997) The surgical resectibility of meningiomas of the cavernous sinus. Neurosurgery 40:238–244
Perneczky A, Knosp E, Matula C (1988) Cavernous sinus surgery: approach through the lateral wall. Acta Neurochir (Wien) 92:76–82
Pieper DR, Al-Mefty O, Hanada Y, Buechner D (1999) Hyperostosis associated with meningioma of the cranial base: secondary changes or tumour invasion. Neurosurgery 44:742–747
Risi P, Uske A, de Tribolet N (1994) Meningiomas involving the anterior clinoid process. British J Neurosurg 8:295–305
Roser F, Nakamura M, Jacobs C, Vorkapic P, Samii M (2005) Sphenoid wing meningiomas with osseous involvement. Surg Neurol 64:37–43
Seifert V, Dietz H (1992) Combined orbito-frontal, sub- and infratemporal fossa approach to skull base neoplasms: surgical technique and clinical application. Acta Neurochir (Wien) 114:139–144
Sekhar LN, Moller AR (1986) Operative management of tumours involving the cavernous sinus. J Neurosurg 64:879–889
Sen C, Hague K (1997) Meningiomas involving the cavernous sinus: histological factors affecting the degree of resection. J Neurosurg 87:535–543
Shaffrey ME, Dolenc VV, Lanzino GL, Wolcott WP, Shaffrey CI (1999) Invasion of the internal carotid artery by cavernous sinus meningiomas. Surg Neurol 52:167–71
Tomasello F, de Divitiis O, Angileri F, Salpietro M, d’Avella D (2003) Large sphenocavernous meningiomas: is there still a role for the intradural approach via the pterional-transsylvian route? Acta Neurochir (Wein) 145:273–282
Umansky F, Valarezo A, Elidan J (1994) The superior wall of the cavernous sinus A microanatomical study. J Neurosurg 81:914–920
Van Loveren HR, Keller JT, El-Kalliny M, Scodary D, Tew JM Jr (1991) The Dolenc technique for cavernous sinus exploration (cadaveric prosection). J Neurosurg 74:837–844
Yasargil MG (1996) Microneurosurgery of CNS tumours. In: Yasargil MG (ed) Microneurosurgery, vol. IVB. George Thieme Verlag, New York, p 136
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
This article describes the neurosurgical practice concerning the management of MMC in Nigeria. Although the authors describe a selected population of MMC patients, several aspects draw attention. First the number of patients with MMC is considerable compared to European countries. The timing of treatment is relatively late (after 7 days), and 5 patients died before treatment. Four patients died of ventriculitis and one patient died of a symptomatic Chiari. Only 18 out of 36 patients required treatment of hydrocephalus of whom 13 patients could be treated with third ventriculostomy. This is a very high success rate of primary endoscopic treatment of hydrocephalus in this patient group while apparently a large number of patients do not develop hydrocephalus at all. The follow-up of these patients has not been mentioned by the authors which may be due to the local circumstances.
The characteristic features of this Nigerian series of patients with MMC treated after one week differs from most series in the literature.Selection bias may be the most probable explanation, but the authors contribute valuable data about their Nigerian practice.
E Hoving
University of Groningen Hospital
Behari et al. reported their experience in the management of giant (max diameter > 5 cm) medial sphenoid wing meningiomas. The series includes 20 patients treated from January 2003 till January 2007. A gross total excision (grade II in the modified De Monte grading scale) was obtained in 9 patients and a subtotal excision in 11 (Grade IVa in 8 and IVb in 3). The outcome was good in 13 patients, fair in 6. One patient died for a massive myocardial infarction three days after reoperation for an extradural hematoma evacuation. The authors propose a preoperative neuroradiologic scoring system to predict extent of resection.
In my experience medial sphenoid wing meningiomas are an heterogeneous group of tumors. Clinoidal and sphenocavernous both are comprised among medial sphenoid wings, but they have different origin, different clinical and neuroradiological features and need different surgical strategy.
Sphenocavernous meningiomas tend to spread lateral-inferiorly into the middle cranial fossa so that ICA and optic and oculomotor nerves are generally involved later. This difference may explain the huge dimensions they may attain before becoming symptomatic. Moreover, they usually spare the paraclinoid segment of the ICA and proximal control of this vessel can be safely achieved through a pure intradural strategy. These lesions are a formidable challenge for the neurosurgeons and their optimal treatment strategy is nowadays controversial. Some authors advocate a so called conservative approach with resection of the intracranial portion of the tumor without attempting to remove the intracavernous tumor; others suggest a more aggressive approach with extensive bone work, extradural dissection for proximal neurovascular control and resection of the tumor included the intracavernous portion. In my experience a pure intradural strategy aimed at the resection of the intracranial portion gives the patient the best chance of a good neurological outcome, with long-term tumor control provided that the remnant, if present, can be followed up and treated with radiosurgery in case of tumor progression.
The authors stated that their preoperative scoring system is able to predict resectability of these lesions. The score is composed by 5 subheadings: tumor volume, Hyperostosis/calcification, edema, cavernous sinus extension, and extension of the meningioma to extra regions (Sellar/parasellar, Superior orbital fissure-orbit, prepontine-Interpeduncolar cistern-posterior fossa). I believe that this score may be useful although it is not balanced because of the different weight of each subheading. Presence of a hyperostosis or a calcified mass > 50% of the whole tumor volume is per se always, in their series, cause of subtotal resection, while peritumoral edema is not correlated to the degree of resection. Moreover even tumor volume has, in the authors series, and also in my experience, a minor weight in the extent of surgical excision.
However their attempt of identifying factors able to predict extent of resection deserves great consideration by the neurosurgical community. Unfortunately, it is impossible at present to achieve preoperative information on consistency and firm adherence of the tumor to major arteries and perforators. These are important variables limiting often radicality in tumor removal. Although, the authors clearly stated the limitation of such a scoring system, they are to be acknowledged for their experience.
F. Tomasello
Messina, Italy
F.F. Angileri
Messina, Italy
Rights and permissions
About this article
Cite this article
Behari, S., Giri, P.J., Shukla, D. et al. Surgical strategies for giant medial sphenoid wing meningiomas: a new scoring system for predicting extent of resection. Acta Neurochir (Wien) 150, 865–877 (2008). https://doi.org/10.1007/s00701-008-0006-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-008-0006-6