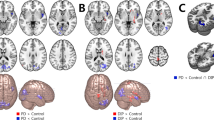
Abstract
Current quantification methods of 123I-FP-CIT SPECT rely on anatomical parcellation of the striatum. We propose here to implement a new method based on MRI segmentation and functional atlas of the basal ganglia (MR-ATLAS) that could provide a reliable quantification within the sensorimotor, associative, and limbic territories of the striatum. Patients with Parkinson’s disease (PD), idiopathic rapid eye movement sleep behavioral disorder (iRBD), and healthy controls underwent 123I-FP-CIT SPECT, MRI, motor, and cognitive assessments. SPECT data were corrected for partial volume effects and registered to a functional atlas of the striatum to allow quantification in every functional region of the striatum (nucleus accumbens, limbic, associative, and sensorimotor parts of the striatum). The MR-ATLAS quantification method is proved to be reliable in every territory of the striatum. In addition, good correlations were found between cognitive dysexecutive tests and the binding within the functional (limbic) territories of the striatum using the MR-ATLAS method, slightly better than correlations found using the anatomical quantification method. This new MR-ATLAS method provides a robust and useful tool for studying the dopaminergic system in PD, particularly with respect to cognitive functions. It may also be relevant to further unravel the relationship between dopaminergic denervation and cognitive or behavioral symptoms.






Similar content being viewed by others
Availability of data and materials
Not applicable.
Code availability
On demand to the corresponding author: gathers publicly available codes (see Methods for details).
References
Ashburner J, Friston KJ (2011) Diffeomorphic registration using geodesic shooting and Gauss-Newton optimisation. Neuroimage 55:954–967. https://doi.org/10.1016/j.neuroimage.2010.12.049
Beckmann M, Johansen-Berg H, Rushworth MFSS (2009) Connectivity-based parcellation of human cingulate cortex and its relation to functional specialization. J Neurosci 29:1175–1190. https://doi.org/10.1523/JNEUROSCI.3328-08.2009
Brogley JE (2019) DatQuant: the future of diagnosing Parkinson disease. J Nucl Med Technol 47:21–26. https://doi.org/10.2967/jnmt.118.222349
Chang L-T (1978) A method for attenuation correction in radionuclide computed tomography. IEEE Trans Nucl Sci 25:638–643. https://doi.org/10.1109/TNS.1978.4329385
Chen B, Wang S, Sun W et al (2017) Functional and structural changes in gray matter of Parkinson’s disease patients with mild cognitive impairment. Eur J Radiol 93:16–23. https://doi.org/10.1016/j.ejrad.2017.05.018
Choi EY, Ding SL, Haber SN (2017) Combinatorial inputs to the ventral striatum from the temporal cortex, frontal cortex, and amygdala: implications for segmenting the striatum. eNeuro. https://doi.org/10.1523/ENEURO.0392-17.2017
Donoso M, Collins AGE, Koechlin E (2014) Foundations of human reasoning in the prefrontal cortex. Science 344:1481–1486. https://doi.org/10.1126/science.1252254
Dubois B, Slachevsky A, Litvan I, Pillon B (2000) The FAB: a frontal assessment battery at bedside. Neurology 55:1621–1626. https://doi.org/10.1212/WNL.57.3.565
Firbank MJ, Yarnall AJ, Lawson RA et al (2017) Cerebral glucose metabolism and cognition in newly diagnosed Parkinson’s disease: ICICLE-PD study. J Neurol Neurosurg Psychiatry 88:310–316. https://doi.org/10.1136/jnnp-2016-313918
Gerlach M, Gsell W, Kornhuber J et al (1996) A post mortem study on neurochemical markers of dopaminergic, GABA-ergic and glutamatergic neurons in basal ganglia-thalamocortical circuits in Parkinson syndrome. Brain Res 741:142–152
Goetz CG, Tilley BC, Shaftman SR et al (2008) Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 23:2129–2170. https://doi.org/10.1002/mds.22340
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184. https://doi.org/10.1136/jnnp.55.3.181
Iranzo A, Santamaría J, Valldeoriola F et al (2017) Dopamine transporter imaging deficit predicts early transition to synucleinopathy in idiopathic rapid eye movement sleep behavior disorder. Ann Neurol 82:419–428. https://doi.org/10.1002/ana.25026
Koch W, Radau PE, Hamann C, Tatsch K (2005) Clinical testing of an optimized software solution for an automated, observer-independent evaluation of dopamine transporter SPECT studies. J Nucl Med 46:1109–1118
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15:155–163. https://doi.org/10.1016/j.jcm.2016.02.012
Kopp B, Rösser N, Tabeling S et al (2013) Performance on the Frontal Assessment Battery is sensitive to frontal lobe damage in stroke patients. BMC Neurol 13:179. https://doi.org/10.1186/1471-2377-13-179
Løvstad M, Funderud I, Meling T et al (2012) Anterior cingulate cortex and cognitive control: neuropsychological and electrophysiological findings in two patients with lesions to dorsomedial prefrontal cortex. Brain Cogn 80:237–249. https://doi.org/10.1016/j.bandc.2012.07.008
Luijten M, Machielsen MWJ, Veltman DJ et al (2014) Systematic review of ERP and fMRI studies investigating inhibitory control and error processing in people with substance dependence and behavioural addictions. J Psychiatry Neurosci 39:149–169
Marquand AF, Haak KV, Beckmann CF (2017) Functional corticostriatal connection topographies predict goal-directed behaviour in humans. Nat Hum Behav 1:1–9. https://doi.org/10.1038/s41562-017-0146
Martinez D, Slifstein M, Broft A et al (2003) Imaging human mesolimbic dopamine transmission with positron emission tomography. Part II: amphetamine-induced dopamine release in the functional subdivisions of the striatum. J Cereb Blood Flow Metab 23:285–300. https://doi.org/10.1097/01.WCB.0000048520.34839.1A
Mawlawi O, Martinez D, Slifstein M et al (2001) Imaging human mesolimbic dopamine transmission with positron emission tomography: I. Accuracy and precision of D2 receptor parameter measurements in ventral striatum. J Cereb Blood Flow Metab 21:1034–1057. https://doi.org/10.1097/00004647-200109000-00002
Menon V, Adleman NE, White CD et al (2001) Error-related brain activation during a Go/NoGo response inhibition task. Hum Brain Mapp 12:131–143. https://doi.org/10.1002/1097-0193(200103)12:3%3c131::AID-HBM1010%3e3.0.CO;2-C
Morris LS, Kundu P, Dowell N et al (2016) Fronto-striatal organization: defining functional and microstructural substrates of behavioural flexibility. Cortex 74:118–133. https://doi.org/10.1016/j.cortex.2015.11.004
Nandhagopal R, Kuramoto L, Schulzer M et al (2009) Longitudinal progression of sporadic Parkinson’s disease: a multi-tracer positron emission tomography study. Brain 132:2970–2979. https://doi.org/10.1093/brain/awp209
Nasreddine Z, Phillips N, Bédirian V et al (2005) The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x
Nieuwenhuis S, Yeung N, Van Den Wildenberg W, Ridderinkhof KR (2003) Electrophysiological correlates of anterior cingulate function in a go/no-go task: effects of response conflict and trial type frequency. Cogn Affect Behav Neurosci 3:17–26. https://doi.org/10.3758/CABN.3.1.17
Nobili F, Naseri M, De Carli F et al (2013) Automatic semi-quantification of [123 I]FP-CIT SPECT scans in healthy volunteers using BasGan version 2: results from the ENC-DAT database. Eur J Nucl Med Mol Imaging 40:565–573. https://doi.org/10.1007/s00259-012-2304-8
O’Brien JT, Colloby S, Fenwick J, Williams ED (2004) Dopamine transporter loss visualized with FP-CIT. Arch Neurol 61:919–925
Palomero-Gallagher N, Hoffstaedter F, Mohlberg H et al (2019) Human pregenual anterior cingulate cortex: structural, functional, and connectional heterogeneity. Cereb Cortex 29:2552–2574. https://doi.org/10.1093/cercor/bhy124
Patenaude B, Smith SM, Kennedy DN, Jenkinson M (2011) A Bayesian model of shape and appearance for subcortical brain segmentation. Neuroimage 56:907–922. https://doi.org/10.1016/j.neuroimage.2011.02.046
Postuma RB, Iranzo A, Hu M et al (2019) Risk and predictors of dementia and parkinsonism in idiopathic REM sleep behaviour disorder: a multicentre study. Brain 142:744–759. https://doi.org/10.1093/brain/awz030
Rousset OG, Deep P, Kuwabara H et al (2000) Effect of partial volume correction on estimates of the influx and cerebral metabolism of 6-[18F]fluoro-L-dopa studied with pet in normal control and Parkinson’s disease subjects. Synapse 37:81–89. https://doi.org/10.1002/1098-2396(200008)37:2%3c81::aid-syn1%3e3.0.co;2-%23
Shenhav A, Botvinick MM, Cohen JD (2013) The expected value of control: an integrative theory of anterior cingulate cortex function. Neuron 79:217–240
Smith CT, Crawford JL, Dang LC et al (2019) Partial-volume correction increases estimated dopamine D2-like receptor binding potential and reduces adult age differences. J Cereb Blood Flow Metab 39:822–833. https://doi.org/10.1177/0271678X17737693
Thomas BA, Erlandsson K, Modat M et al (2011) The importance of appropriate partial volume correction for PET quantification in Alzheimer’s disease. Eur J Nucl Med Mol Imaging 38:1104–1119. https://doi.org/10.1007/s00259-011-1745-9
Torta DM, Cauda F (2011) Different functions in the cingulate cortex, a meta-analytic connectivity modeling study. Neuroimage 56:2157–2172. https://doi.org/10.1016/j.neuroimage.2011.03.066
Tossici-Bolt L, Hoffmann SMA, Kemp PM et al (2006) Quantification of [123I]FP-CIT SPECT brain images: an accurate technique for measurement of the specific binding ratio. Eur J Nucl Med Mol Imaging 33:1491–1499. https://doi.org/10.1007/s00259-006-0155-x
Tzourio-Mazoyer N, Landeau B, Papathanassiou D et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15:273–289
Yang J, Huang SC, Mega M et al (1996) Investigation of partial volume correction methods for brain FDG PET studies. IEEE Trans Nucl Sci 43:3322–3327. https://doi.org/10.1109/23.552745
Yelnik J, Bardinet E, Dormont D et al (2007) A three-dimensional, histological and deformable atlas of the human basal ganglia. I. Atlas construction based on immunohistochemical and MRI data. Neuroimage 34:618–638. https://doi.org/10.1016/j.neuroimage.2006.09.026
Acknowledgements
The authors thank the Clinical Investigations Centre (CIC) for its involvement in the study organization and management, Prof. Richard Levy and Dr. Virginie Czernecki for their helpful discussion around this manuscript, and Dr. Renaud La Joie for his help with the Figures.
ICEBERG study group
Nominated consortia representative: Prof. Marie Vidailhet, MD—Département de Neurologie, Groupe Hospitalier Pitié-Salpêtrière, 47-83 boulevard de l’Hôpital, 75651 PARIS Cedex 13, France, marie.vidailhet@aphp.fr. Steering committee: Marie Vidailhet, MD [(principal investigator of ICEBERG), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Jean-Christophe Corvol, MD, PhD [(scientific lead), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Isabelle Arnulf, MD, PhD (member of the steering committee), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Sleep Disorders Unit, Paris, France), Stéphane Lehéricy, MD, PhD (member of the steering committee), (Institut du Cerveau et de la Moelle Epinière—ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neuroradiology, Paris, France); Clinical data: Marie Vidailhet, MD [(coordination), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Graziella Mangone, MD, PhD [(co-coordination), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Jean-Christophe Corvol, MD, PhD (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), Isabelle Arnulf, MD, PhD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Sleep Disorders Unit, Paris, France), Jonas Ihle, MD (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), Caroline Weill, MD (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), David Grabli, MD, PhD (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), Florence Cormier-Dequaire, MD, PhD (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), Louise Laure Mariani, MD, PhD (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), Bertrand Degos, MD, PhD (Service de Neurologie, Hôpital Avicenne, Hôpitaux Universitaires de Paris—Seine Saint Denis, AP-HP, Bobigny, France; Center for Interdisciplinary Research in Biology, Collège de France, INSERM U1050, CNRS UMR7241, Labex Memolife, Paris Sciences et Lettres, Paris, France); Neuropsychological data: Richard Levy, MD, PhD [(coordination), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Fanny Pineau, MSc [(neuropsychologist), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Julie Socha, MSc [(neuropsychologist), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Eve Benchetrit, MSc [(neuropsychologist), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Virginie Czernecki, PhD [(neuropsychologist), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)]; Eye movement: Sophie Rivaud-Pechoux, PhD (coordination), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France); Elodie Hainque, MD, PhD (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France); Sleep assessment: Isabelle Arnulf, MD, PhD (coordination), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Sleep Disorders Unit, Paris, France), Smaranda Leu Semenescu, MD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Sleep Disorders Unit, Paris, France), Pauline Dodet, MD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Sleep Disorders Unit, Paris, France); Genetic data: Jean-Christophe Corvol, MD, PhD [(coordination), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Graziella Mangone, MD, PhD [(co-coordination), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Samir Bekadar, MSc [(biostatistician), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Alexis Brice, MD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Genetics, Paris, France), Suzanne Lesage, PhD (genetic analyses) (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France); Metabolomics: Fanny Mochel, MD, PhD (coordination), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Genetics, Paris, France), Farid Ichou, PhD (ICAN, Pitié-Salpêtrière Hospital, Paris), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Genetics, Paris, France), Vincent Perlbarg, PhD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), Benoit Colsch, PhD (CEA-INRA UMR 0496, DRF/Institut Joliot/SPI, Université Paris-Saclay, MetaboHUB, France), Arthur Tenenhaus, PhD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; L2S Laboratory at CentraleSupélec, France); Brain MRI data: Stéphane Lehericy, MD, PhD (coordination), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neuroradiology, Paris, France), Rahul Gaurav, MS, [(data analysis), (Institut du Cerveau et de la Moelle Epinièrem ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Nadya Pyatigorskaya, MD, PhD (data analysis), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neuroradiology, Paris, France), Lydia Yahia-Cherif, PhD [(Biostatistics), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Romain Valabregue, PhD (data analysis) (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France), Cécile Galléa, PhD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France); Datscan imaging data: Marie-Odile Habert, MD (coordination), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Nuclear Medicine, Paris, France; Sorbonne Université, CNRS, INSERM, Laboratoire d’Imagerie Biomédicale, LIB, Paris, France); Voice recording: Dijana Petrovska, PhD [(Telecom Sud Paris, Evry, coordination), (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; TELECOM SudParis, CNRS SAMOVAR UMR 5157, Département Electronique et Physique, MINES TELECOM, Evry, France)], Laetitia Jeancolas, PhD (Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France; TELECOM SudParis, CNRS SAMOVAR UMR 5157, Département Electronique et Physique, MINES TELECOM, Evry, France); Study management: Vanessa Brochard [(coordination), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Alizé Chalançon [(clinical research assistant), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)], Carole Dongmo-Kenfack [(clinical research assistant), (AP-HP Sorbonne Université, Hôpital Pitié-Salpêtrière, Department of Neurology, Paris, France; Institut du Cerveau et de la Moelle Epinière, ICM, Sorbonne Université, INSERM U1127, CNRS 7225, Paris, France)]; study sponsoring: INSERM, Paris.
Funding
IHU-A_ICM ANR‐11‐INBS‐0006, Fondation d'Entreprise EDF, Mr A. Mallart, Energiepole (Belgique), Mr J-J. Legrand and Société Française de Médecine Esthétique (France).
Author information
Authors and Affiliations
Consortia
Contributions
NV conceived the presented methodological pipeline, contributed to data collection, performed analyses, and wrote the manuscript. GB contributed to data collection and to the writing of the manuscript. M-OH, AK, JCC, GM, SL, MV, and DG contributed to the design of the study, data collection, and writing of manuscript. JA, OJ, RV, and SFV contributed to data analysis.
Corresponding author
Ethics declarations
Conflict of interest
Independent of this work, NV received research support from Fondation Bettencourt-Schueller, Fondation Servier and Fondation pour la Recherche sur l’Alzheimer; travel grant from the Movement Disorders Society, Merz-Pharma and GE Healthcare SAS; and is an unpaid sub-investigator in NCT04241068 (aducanumab, Biogen), NCT04437511 (donanemab, Eli-Lilly), NCT04592341 (gantenerumab, Roche), NCT03887455 (lecanemab, Eisai), NCT03352557 (gosuranemab, Biogen), NCT03828747 and NCT03289143 (semorinemab, Roche), NCT04619420 (JNJ-63733657, Janssen—Johnson & Johnson), NCT04374136 (AL001, Alector), NCT04592874 (AL002, Alector). M-OH receives Consultant Fees from the Blue Earth Company. JCC has served in advisory boards for Air Liquide, Biogen, Denali, Ever Pharma, Idorsia, Prevail Therapeutic, Theranexus, UCB; and received grants from Ipsen, the Michael J Fox Foundation, and Sanofi. SL received grants from ‘Investissements d’avenir’ [grant number ANR-10-IAIHU-06 and ANR-11-INBS-0006], Fondation Bettencourt Schuller (Ultrabrain project) and Biogen Inc. DG received grants “Contrat Interface” from INSERM, AP-HP (DRC-PHRC) and France Parkinson, served on scientific advisory boards for AbbVie and Zambon; received research funding from Air Liquide; received speech honorarium from UCB, Teva, Medtronic, Abbvie and Merz; received travel funding from Teva, Novartis, Abbvie and Merz. GB, AK, JA, OJ, RV, SFV, GM, and MV declare no conflict of interest related to this work.
Ethics approval and consent to participate
The study was approved by the local ethic committee Comité de Protection des Personnes (CPP) “Ile-de-France VI” (IRB: 2014-A00725-42/48-14). Informed consent was obtained from all individual participants included in the study.
Consent for publication
All the authors have read and agreed with current content of the submitted manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Members of the “ICEBERG Study Group” are listed in acknowledgement section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Villain, N., Béra, G., Habert, MO. et al. Dopamine denervation in the functional territories of the striatum: a new MR and atlas-based 123I-FP-CIT SPECT quantification method. J Neural Transm 128, 1841–1852 (2021). https://doi.org/10.1007/s00702-021-02434-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-021-02434-9