Abstract
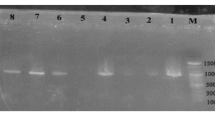
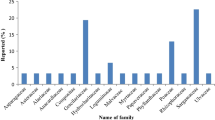
White spot syndrome virus (WSSV), the most contagious pathogen of cultured shrimp, causes mass mortality, leading to huge economic loss to the shrimp industry. The lack of effective therapeutic or prophylactic measures has aggravated the situation, necessitating the development of antiviral agents. With this objective, the antiviral activity in the aqueous extract of a mangrove plant Ceriops tagal in Penaeus monodon was evaluated. The Ceriops tagal aqueous extract (CTAE) was non-toxic to shrimps at 50 mg/ml when injected intramuscularly at a dosage of 10 μL/animal (0.5 mg/animal) and showed a protective effect against WSSV at 30 mg/ml when mixed with WSSV suspension at a 1:1 ratio. When the extract was administered along with the diet and the animals were challenged orally, there was a dose-dependent increase in survival, culminating in 100 % survival at a concentration of 500 mg/kg body weight/day. Neither hypertrophied nuclei nor the viral envelope protein VP28 could be demonstrated in surviving shrimps using histology and indirect immunofluorescence histochemistry (IIFH), respectively. To elucidate the mode of action, the temporal expression of WSSV genes and shrimp immune genes, including antimicrobial peptides, was attempted. None of the viral genes were found to be expressed in shrimps that were fed with the extract and challenged or in those that were administered CTAE-exposed WSSV. The overall results suggest that the aqueous extract from C. tagal can protect P. monodon from white spot syndrome virus infection.







Similar content being viewed by others
References
Achuthankutty CT, Desai UM (2004) Treatment of white spot syndrome virus (WSSV) in penaeid shrimp aquaculture using plant extract. In: Proceedings of MBR 2004 national seminar on New Frontiers in marine bioscience research, India, pp 63–67
Anil TM, Shankar KM, Mohan CV (2002) Monoclonal antibodies developed for sensitive detection and comparison of white spot syndrome virus isolates in India. Dis Aquat Org 51:67–75
Balasubramanian G, Sarathi M, Kumar S, Hameed A (2007) Screening the antiviral activity of Indian medicinal plants against white spot syndrome virus in shrimp. Aquaculture 263:15–19
Balasubramanian G, Sarathi M, Venkatesan C, Thomas J, Hameed ASS (2008) Oral administration of antiviral plant extract of Cynodon dactylon on a large scale production against white spot syndrome virus (WSSV) in Penaeus monodon. Aquaculture 279:2–5
Chang C, Su M, Chen H, Liao I (2003) Dietary β-1, 3-glucan effectively improves immunity and survival of Penaeus monodon challenged with white spot syndrome virus. Fish Shellfish Immunol 15:297–310
Chotigeat W, Tongsupa S, Supamataya K, Phongdara A (2004) Effect of fucoidan on disease resistance of black tiger shrimp. Aquaculture 233:23–30
Citarasu T, Sivaram V, Immanuel G, Rout N, Murugan V (2006) Influence of selected Indian immunostimulant herbs against white spot syndrome virus (WSSV) infection in black tiger shrimp, Penaeus monodon with reference to haematological, biochemical and immunological changes. Fish Shellfish Immunol 21:372–384
Duke J, Wain K (1981) Medicinal plants of the world. Computer index with more than 85,000 entries. 3 vols Medicinal plants of the world. - cited in Duke J. A (1983): Handbook of Energy Crops. Unpublished. [http://www.hort.purdue.edu/newcrop/Indices/index_ab.html]
Escobedo-Bonilla CM, Alday-Sanz V, Wille M, Sorgeloos P, Pensaert MB, Nauwynck HJ (2008) A review on the morphology, molecular characterization, morphogenesis and pathogenesis of white spot syndrome virus. J Fish Dis 31:1–18
Friesen P (1997) Regulation of Baculovirus early gene expression. In: Miller LK (ed) The Baculoviruses. Plenum Press, New York, pp 141–170
Ghosh A, Misra S, Dutta A, Choudhury A (1985) Pentacyclic triterpenoids and sterols from seven species of Mangrove. Phytochem 24:1725–1727
Gomez VP, GonzaŁ lez AL, Co¤rdova AC, Meyer ML, Coronado JAF, Ruiz AŁP (2009) Probiotic microorganisms and antiviral plants reduce mortality and prevalence of WSSV in shrimp (Litopenaeus vannamei) cultured under laboratory conditions. Aquac Res 40:1481–1489
Ha YM, Soo JG, Thi-Hoai N, Chae HR, Ki HK, Yoon-Kwon N, Sung-Koo K (2008) Vaccination of shrimp (Penaeus chinensis) against white spot syndromevirus (WSSV). J Microbiol Biotechnol 18:964–967
Jiravanichpaisal P, Puanglarp N, Petkon S, Donnuea S, Söderhäll I, Söderhäll K (2007) Expression of immune-related genes in larval stages of the giant tiger shrimp, Penaeus monodon. Fish Shellfish Immunol 23:815–824
Lightner D (1996) A handbook of pathology and diagnostic procedures for diseases of penaeid shrimp. World Aquaculture Society, Baton Rouge, p 304
Lightner D (2005) Biosecurity in shrimp farming: pathogen exclusion through use of SPF stock and routine surveillance. J World Aquac Soc 36:229–247
Lin P, Fu Q (1995) Environmental ecology and economic utilization of mangroves in China. Higher Education Press, Beijing, pp 1–95
Lin Y, Vaseeharan B, Chen J (2008) Molecular cloning and phylogenetic analysis on alpha2-macroglobulin (alpha2-M) of white shrimp Litopenaeus vannamei. Dev Comp Immunol 32:317–329
Liu H, Soderhall K, Jiravanichpaisal P (2009) Antiviral immunity in crustaceans. Fish Shellfish Immunol 27:79–88
Liu WJ, Chang YS, Wang CH, Kou G, Lo CF (2005) Microarray and RT-PCR screening for white spot syndrome virus immediate-early genes in cycloheximide-treated shrimp. Virol 334:327–341
Lo C, Leu J, Ho C, Chen C, Peng S, Chen Y, Chou C, Yeh P, Huang C, Chou H, Wang C, Kou G (1996) Detection of baculovirus associated with white spot syndrome (WSBV) in penaeid shrimps using polymerase chain reaction. Dis Aquat Org 25:133–141
Manjusha M, Varghese R, Philip R, Mohandas A, Bright Singh IS (2009) Pathological changes in Fenneropenaeus indicus experimentally infected with white spot virus and virus morphogenesis. J Invertebr Pathol 103:225–232
Mayo M (2002) A summary of taxonomic changes recently approved by ICTV. Arch Virol 147:1655–1656
Namikoshi A, Wu JL, Yamashita T, Nishizawa T, Nishioka T, Arimoto M, Muroga K (2004) Vaccination trials with Penaeus japonicus to induce resistance to white spot syndrome virus. Aquaculture 229:25–35
Naskar K, Mandal R (1999) Ecology and bio diversity of Indian mangroves. Daya Books 2:397–400
Rahman MM, Escobedo-Bonilla CM, Wille M, Alday Sanz V, Audoorn L, Neyts J, Pensaert MB, Sorgeloos P, Nauwynck HJ (2006) Clinical effect of cidofovir and a diet supplemented with Spirulina platensis in white spot syndrome virus (WSSV) infected specific pathogen-free Litopenaeus vannamei juveniles. Aquaculture 255:600–605
Rastogi RP, Mehrotra BN (1991) Compendium of Indian medicinal plants. Central drug research institute and publications and information directorate, New Delhi, p 515
Roch P, Yang Y, Toubiana M, Aumelas A (2008) NMR structure of musselmytilin, and antiviral-antibacterial activities of derived synthetic peptides. Dev Comp Immunol 32:227–238
Singh ISB, Manjusha M, Pai SS, Philip R (2005) Fenneropenaeus indicus is protected from white spot disease by oral administration of inactivated white spot syndrome virus. Dis Aquat Org 66:265–270
Sudheer NS, Philip Rosamma, Bright Singh IS (2011) In vivo screening of mangrove plants for anti WSSV activity in Penaeus monodon, and evaluation of Ceriops tagal as a potential source of antiviral molecules. Aquaculture 311:36–41
Supamattaya K, Kiriratnikom S, Boonyaratpalin M, Borowitzka L (2005) Effect of a Dunaliella extract on growth performance, health condition, immune response and disease resistance in black tiger shrimp (Penaeus monodon). Aquaculture 248:207–216
Takahasi Y, Uchara K, Watanabe R, Okumura T, Yamashita T, Omura H, Yomo T, Kawano T, Kanemitsu A et al (1998) Efficacy of oral administration of fucoidan, a sulphated polysaccharide in controlling white spot syndrome in kuruma shrimp in Japan. In: Flegel TW (ed) Advances in shrimp biotechnology. National center for genetic engineering and biotechnology, Bangkok, pp 171–173
Tan Y, Shi Z (2008) Proteomic analyses of the shrimp white spot syndrome virus. Virol Sin 23:157–166
Wu JY, Xiao Q, Xu J, Li MY, Pan JY, Yang M (2008) Natural products from true mangrove flora: source, chemistry and bioactivities. Nat Prod Rep 25:955–981
Zhang J, Li F, Wang Z, Xiang J (2007) Cloning and recombinant expression of a crustin-like gene from Chinese shrimp, Fenneropenaeus chinensis. J Biotechnol 127:605–614
Acknowledgments
This work was carried out with financial assistance from the Department of Biotechnology under the projects BT/PR3509/AAQ/03/173/2000 and BT/PR7419/AAQ/03/274/2006. We acknowledge the Department of Science and Technology, Lakshadweep, for the partial supply of mangrove plants, and Mr. Krishna Iyer, Scientist, CIFT, Cochin (Rtd) for statistical analysis. We thankfully acknowledge Dr. A. Mohandas, Emeritus Professor, NCAAH, for editing the manuscript. The first author thanks DBT for a fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sudheer, N.S., Philip, R. & Bright Singh, I.S. Anti–white spot syndrome virus activity of Ceriops tagal aqueous extract in giant tiger shrimp Penaeus monodon . Arch Virol 157, 1665–1675 (2012). https://doi.org/10.1007/s00705-012-1346-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-012-1346-3