Summary.
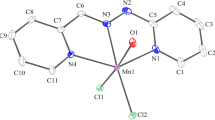
Group 2B transition metal complexes of bis[N-(2,6-diisopropylphenyl)imino]acenaphthene (o,o′-iPr2C6H3-BIAN), namely, [Hg(o,o′-iPr2C6H3-BIAN)Cl2] (1), [Zn(o,o′-iPr2C6H3-BIAN)2](ClO4)2 (2), and [Cd(o,o′-iPr2C6H3-BIAN)2](ClO4)2 (3) have been synthesized and characterized. In complexes 2 and 3, IR, NMR, and conductivity measurements confirm the coordination of two (o,o′-iPr2C6H3-BIAN) ligands to the metal center with two discrete perchlorate anions. X-Ray crystal structure of 1 indicates a distorted tetrahedral geometry with two nitrogen atoms from (o,o′-iPr2C6H3-BIAN) ligand and two chloride atoms coordinating to the Hg(II) center.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Ayaan, U. Synthesis and Structural Studies of Group 2B Transition Metal Complexes with the Bulky Nitrogen Ligand Bis[N-(2,6-diisopropylphenyl) imino]acenaphthene. Monatshefte für Chemie 135, 919–925 (2004). https://doi.org/10.1007/s00706-004-0181-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-004-0181-y