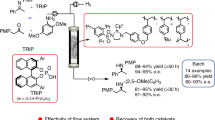
Abstract
The asymmetric hydrogenation of benchmark substrates dimethyl itaconate and (Z)-α-acetamidocinnamic acid methyl ester with chiral pentane-2,4-diyl-based phosphine–phosphite Rh complexes immobilized on the support with heteropolyacid (phosphotungstic acid) as anchoring agent has been studied. The complexes have been supported on commercially available Al2O3 by the Augustine method. The novel heterogeneous catalysts were applied in a high-throughput flow reactor. The effect of the pressure, temperature, substrate concentration, and flow rate was thoroughly screened to optimize reaction conditions. The immobilized catalysts proved to be remarkably stable and could be used 6 h in the microfluidic-based reactor without a significant loss of activity and selectivity. Furthermore, under optimized conditions, the hydrogenation product could be obtained with high activity (TOF >2000 h−1) and enantioselectivity (up to 99% ee). As the first precedent, the potential of supported Rh(P-OP)-complexes under flow conditions has been presented.
Graphical abstract

Similar content being viewed by others
References
Ding K, Uozumi Y (2008) Handbook of asymmetric heterogeneous catalysis. Wiley-VCH, Weinheim
De Vos DE, Vankelecom IFJ, Jacobs PA (2000) Chiral catalyst immobilization and recycling. Wiley-VCH, Weinheim
Zhao D, Ding K (2013) ACS Catal 3:928
Song CE, Lee SG (2002) Chem Rev 102:3495
Zsigmond Á, Balatoni I, Bogar K, Notheisz F, Joó F (2004) J Catal 227:428
McMorn P, Hutchings GJ (2004) Chem Soc Rev 33:108
König M, Linhardt A, Brüggemann O, Teasdale I (2016) Monatsh Chem 147:1575
Augustine R, Tanielyan S, Anderson S, Yang H (1999) Chem Commun:1257
Stephenson P, Licence P, Ross SK, Poliakoff M (2004) Green Chem 6:521
Stephenson P, Kondor B, Licence P, Scovell K, Ross SK, Poliakoff M (2006) Adv Synth Catal 348:1605
Burk MJ (1991) J Am Chem Soc 113:8518
Al Herz MA, Tsoligkas AN, Simmons MJH, Wood J (2011) Appl Catal A 396:148
Duque R, Pogorzelec PJ, Cole-Hamilton DJ (2013) Angew Chem Int Ed 52:1
Sakai N, Mano S, Nozaki K, Takaya H (1993) J Am Chem Soc 115:7033
Nozaki K, Takaya K, Hiyama T (1997) Top Catal 4:175
Falk A, Gödertz A-L, Schmalz HG (2013) Angew Chem Int Ed 52:1576
Falk A, Cavalieri A, Nichol GS, Vogt D, Schmalz H-G (2015) Adv Synth Catal 357:3317
Arribas I, Rubio M, Kleman P, Pizzano A (2013) J Org Chem 78:3997
Dindaroğlu M, Akyol S, Şimşir H, Neudörfl J-M, Burke A, Schmalz H-G (2013) Tetrahedron Asymmetry 24:657
Gavrilov KN, Zheglov SV, Shiryaev AA, Potapova OV, Gavrilov VK, Volov AN, Zamilatskov IA (2013) Russ Chem Bull 62:1097
Vaquero M, Vargas S, Suárez A, García-Garrido SE, Álvarez E, Mancera M, Pizzano A (2012) Organometallics 31:3551
Fernández-Pérez H, Benet-Buchholz J, Vidal-Ferran A (2013) Org Lett 15:3634
Núñez-Rico J-L, Vidal-Ferran A (2013) Org Lett 15:2066
Lao JR, Benet-Buchholz J, Vidal-Ferran A (2014) Organometallics 33:2960
Núñez-Rico JL, Etayo P, Fernández-Pérez H, Vidal-Ferran A (2012) Adv Synth Catal 354:3025
Farkas G, Balogh S, Madarász J, Szöllősy Á, Darvas F, Ürge L, Gouygou M, Bakos J (2012) Dalton Trans 41:9493
Farkas G, Császár Z, Balogh S, Szöllősy Á, Gouygou M, Bakos J (2013) Catal Commun 36:94
Fernández-Pérez H, Etayo P, Panossian P, Vidal-Ferran A (2011) Chem Rev 111:2119
Pizzano A (2016) Chem Rec 16:2599
Heutz FJL, Kamer PCJ (2016) Dalton Trans 45:2116
Kleman P, Barbaro P, Pizzano A (2015) Green Chem 17:3826
Farkas G, Balogh S, Szöllősy Á, Ürge L, Darvas F, Bakos J (2011) Tetrahedron Asymmetry 22:2104
Madarász J, Farkas G, Balogh S, Szöllősy Á, Kovács J, Darvas F, Ürge L, Bakos J (2011) J Flow Chem 2:62
Brunauer S, Emmett PH, Teller E (1938) J Am Chem Soc 60:309
Barrett P, Joyner LG, Halenda PP (1951) J Am Chem Soc 73:373
Acknowledgements
We thank ThalesNano Nanotechnology Inc. for the H-Cube™ microreactor and Mr. Béla Édes for skilful assistance in analytical measurements and synthetic experiments. The research was supported by the ‘National Excellence Program’ in the framework of the project TÁMOP-4.2.4.A/2-11/1-2012-0001, by the National Research, Development and Innovation Office (NKFIH, K 115539) and by the project GINOP-2.3.2-15-2016-00053.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Madarász, J., Nánási, B., Kovács, J. et al. Immobilized phosphine–phosphite rhodium complexes: highly active and enantioselective catalysts for asymmetric hydrogenation under continuous flow conditions. Monatsh Chem 149, 19–25 (2018). https://doi.org/10.1007/s00706-017-2048-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-017-2048-z