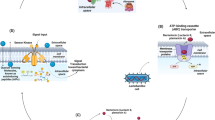
Abstract
Naturally occurring polyamines are ubiquitously distributed and play important roles in cell development, amino acid and protein synthesis, oxidative DNA damage, proliferation, and cellular differentiation. Macrophages are essential in the innate immune response, and contribute to tissue remodeling. Naïve macrophages have two major potential fates: polarization to (1) the classical pro-inflammatory M1 defense response to bacterial pathogens and tumor cells, and (2) the alternatively activated M2 response, induced in the presence of parasites and wounding, and also implicated in the development of tumor-associated macrophages. ODC, the rate-limiting enzyme in polyamine synthesis, leads to an increase in putrescine levels, which impairs M1 gene transcription. Additionally, spermidine and spermine can regulate translation of pro-inflammatory mediators in activated macrophages. In this review, we focus on polyamines in macrophage activation patterns in the context of gastrointestinal inflammation and carcinogenesis. We seek to clarify mechanisms of innate immune regulation by polyamine metabolism and potential novel therapeutic targets.


Similar content being viewed by others
References
Alexander ET, Minton A, Peters MC, Phanstiel O 4th, Gilmour SK (2017) A novel polyamine blockade therapy activates an anti-tumor immune response. Oncotarget 8:84140–84152. https://doi.org/10.18632/oncotarget.20493
Anderson CF, Mosser DM (2002) A novel phenotype for an activated macrophage: the type 2 activated macrophage. J Leukoc Biol 72:101–106. https://doi.org/10.1189/jlb.72.1.101
Asim M, Chaturvedi R, Hoge S, Lewis ND, Singh K, Barry DP, Algood HS, de Sablet T, Gobert AP, Wilson KT (2010) Helicobacter pylori induces ERK-dependent formation of a phospho-c-Fos c-Jun activator protein-1 complex that causes apoptosis in macrophages. J Biol Chem 285:20343–20357. https://doi.org/10.1074/jbc.M110.116988
Bassiri H, Benavides A, Haber M, Gilmour SK, Norris MD, Hogarty MD (2015) Translational development of difluoromethylornithine (DFMO) for the treatment of neuroblastoma. Transl Pediatr 4:226–238. https://doi.org/10.3978/j.issn.2224-4336.2015.04.06
Baydoun AR, Morgan DM (1998) Inhibition of ornithine decarboxylase potentiates nitric oxide production in LPS-activated J774 cells. Br J Pharmacol 125:1511–1516. https://doi.org/10.1038/sj.bjp.0702231
Benoit M, Desnues B, Mege J-L (2008) Macrophage polarization in bacterial infections. J Immunol 181:3733–3739. https://doi.org/10.4049/jimmunol.181.6.3733
Brooks WH (2013) Increased polyamines alter chromatin and stabilize autoantigens in autoimmune diseases. Front Immunol 4:91. https://doi.org/10.3389/fimmu.2013.00091
Bussière FI, Chaturvedi R, Cheng Y, Gobert AP, Asim M, Blumberg DR, Xu H, Kim PY, Hacker A, Casero RA, Wilson KT (2005) Spermine causes loss of innate immune response to Helicobacter pylori by inhibition of inducible nitric-oxide synthase translation. J Biol Chem 280:2409–2412. https://doi.org/10.1074/jbc.C400498200
Chaturvedi R, Cheng Y, Asim M, Bussière FI, Xu H, Gobert AP, Hacker A, Casero RA, Wilson KT (2004) Induction of polyamine oxidase 1 by Helicobacter pylori causes macrophage apoptosis by hydrogen peroxide release and mitochondrial membrane depolarization. J Biol Chem 279:40161–40173. https://doi.org/10.1074/jbc.M401370200
Chaturvedi R, Asim M, Lewis ND, Algood HMS, Cover TL, Kim PY, Wilson KT (2007) l-arginine availability regulates inducible nitric oxide synthase-dependent host defense against Helicobacter pylori. Infect Immun 75:4305–4315. https://doi.org/10.1128/IAI.00578-07
Chaturvedi R, Asim M, Hoge S, Lewis ND, Singh K, Barry DP, de Sablet T, Piazuelo MB, Sarvaria AR, Cheng Y (2010) Polyamines impair immunity to Helicobacter pylori by inhibiting l-arginine uptake required for nitric oxide production. Gastroenterology 139:1686–1698. https://doi.org/10.1053/j.gastro.2010.06.060
Chaturvedi R, Asim M, Romero-Gallo J, Barry DP, Hoge S, de Sablet T, Delgado AG, Wroblewski LE, Piazuelo MB, Yan F (2011) Spermine oxidase mediates the gastric cancer risk associated with Helicobacter pylori CagA. Gastroenterology 141:1696–1708. https://doi.org/10.1053/j.gastro.2011.07.045
Chaturvedi R, de Sablet T, Peek RM, Wilson KT (2012) Spermine oxidase, a polyamine catabolic enzyme that links Helicobacter pylori CagA and gastric cancer risk. Gut Microbes 3:48–56. https://doi.org/10.4161/gmic.19345
Chaturvedi R, Asim M, Barry DP, Frye JW, Casero RA Jr, Wilson KT (2014a) Spermine oxidase is a regulator of macrophage host response to Helicobacter pylori: enhancement of antimicrobial nitric oxide generation by depletion of spermine. Amino Acids 46:531–542. https://doi.org/10.1007/s00726-013-1531-z
Chaturvedi R, Asim M, Piazuelo MB, Yan F, Barry DP, Sierra JC, Delgado AG, Hill S, Casero RA Jr, Bravo LE, Dominguez RL, Correa P, Polk DB, Washington MK, Rose KL, Schey KL, Morgan DR, Peek RM Jr, Wilson KT (2014b) Activation of EGFR and ERBB2 by Helicobacter pylori results in survival of gastric epithelial cells with DNA damage. Gastroenterology 146:1739–51.e14. https://doi.org/10.1053/j.gastro.2014.02.005
Chaturvedi R, de Sablet T, Asim M, Piazuelo MB, Barry DP, Verriere TG, Sierra JC, Hardbower DM, Delgado AG, Schneider BG (2015) Increased Helicobacter pylori-associated gastric cancer risk in the Andean region of Colombia is mediated by spermine oxidase. Oncogene 34:3429–3440. https://doi.org/10.1038/onc.2014.273
Coburn LA, Singh K, Asim M, Barry DP, Allaman MM, Al-Greene NT, Hardbower DM, Polosukhina D, Williams CS, Delgado AG (2018) Loss of solute carrier family 7 member 2 exacerbates inflammation-associated colon tumorigenesis. Oncogene 38:1067–1079. https://doi.org/10.1038/s41388-018-0492-9
Duleu S, Vincendeau P, Courtois P, Semballa S, Lagroye I, Daulouède S, Boucher J-L, Wilson KT, Veyret B, Gobert AP (2004) Mouse strain susceptibility to trypanosome infection: an arginase-dependent effect. J Immunol 172:6298–6303. https://doi.org/10.4049/jimmunol.172.10.6298
Eaden JA, Abrams KR, Mayberry JF (2001) The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut 48:526–535. https://doi.org/10.1136/gut.48.4.526
Ekbom A, Helmick C, Zack M, Adami H-O (1990) Ulcerative colitis and colorectal cancer. N Engl J Med 323:1228–1233. https://doi.org/10.1056/NEJM199011013231802
El Kasmi KC, Qualls JE, Pesce JT, Smith AM, Thompson RW, Henao-Tamayo M, Basaraba RJ, König T, Schleicher U, Koo M-S (2008) Toll-like receptor–induced arginase 1 in macrophages thwarts effective immunity against intracellular pathogens. Nat Immunol 9:1399–1406. https://doi.org/10.1038/ni.1671
Fleming BD, Mosser DM (2011) Regulatory macrophages: setting the threshold for therapy. Eur J Immunol 41:2498–2502. https://doi.org/10.1002/eji.201141717
Gabrilovich DI, Nagaraj S (2009) Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 9:162–174. https://doi.org/10.1038/nri2506
Galli SJ, Borregaard N, Wynn TA (2011) Phenotypic and functional plasticity of cells of innate immunity: macrophages, mast cells and neutrophils. Nat Immunol 12:1035–1044. https://doi.org/10.1038/ni.2109
Georgopoulos K (2002) Haematopoietic cell-fate decisions, chromatin regulation and Ikaros. Nat Rev Immunol 2:162–174. https://doi.org/10.1038/nri747
Gobert AP, Daulouede S, Lepoivre M, Boucher JL, Bouteille B, Buguet A, Cespuglio R, Veyret B, Vincendeau P (2000) l-Arginine availability modulates local nitric oxide production and parasite killing in experimental trypanosomiasis. Infect Immun 68:4653–4657. https://doi.org/10.1128/IAI.68.8.4653-4657.2000
Gobert AP, Cheng Y, Wang J-Y, Boucher J-L, Iyer RK, Cederbaum SD, Casero RA, Newton JC, Wilson KT (2002) Helicobacter pylori induces macrophage apoptosis by activation of arginase II. J Immunol 168:4692–4700. https://doi.org/10.4049/jimmunol.168.9.4692
Gobert AP, Cheng Y, Akhtar M, Mersey BD, Blumberg DR, Cross RK, Chaturvedi R, Drachenberg CB, Boucher J-L, Hacker A, Casero RA, Wilson KT (2004) Protective role of arginase in a mouse model of colitis. J Immunol 173:2109–2117. https://doi.org/10.4049/jimmunol.173.3.2109
Hardbower DM, Asim M, Murray-Stewart T, Casero RA Jr, Verriere T, Lewis ND, Chaturvedi R, Piazuelo MB, Wilson KT (2016) Arginase 2 deletion leads to enhanced M1 macrophage activation and upregulated polyamine metabolism in response to Helicobacter pylori infection. Amino Acids 48:2375–2388. https://doi.org/10.1007/s00726-016-2231-2
Hardbower DM, Asim M, Luis PB, Singh K, Barry DP, Yang C, Steeves MA, Cleveland JL, Schneider C, Piazuelo MB, Gobert AP, Wilson KT (2017a) Ornithine decarboxylase regulates M1 macrophage activation and mucosal inflammation via histone modifications. Proc Natl Acad Sci USA 114:E751–E760. https://doi.org/10.1073/pnas.1614958114
Hardbower DM, Coburn LA, Asim M, Singh K, Sierra JC, Barry DP, Gobert AP, Piazuelo MB, Washington MK, Wilson KT (2017b) EGFR-mediated macrophage activation promotes colitis-associated tumorigenesis. Oncogene 36:3807–3819. https://doi.org/10.1038/onc.2017.23
Hasko G, Kuhel DG, Marton A, Nemeth ZH, Deitch EA, Szabó C (2000) Spermine differentially regulates the production of interleukin-12 p40 and interleukin-10 and suppresses the release of the T helper 1 cytokine interferon-gamma. Shock 14:144–149
Hayashi S, Murakami Y, Matsufuji S (1996) Ornithine decarboxylase antizyme: a novel type of regulatory protein. Trends Biochem Sci 21:27–30. https://doi.org/10.1016/S0968-0004(06)80024-1
Hobbs CA, Gilmour SK (2000) High levels of intracellular polyamines promote histone acetyltransferase activity resulting in chromatin hyperacetylation. J Cell Biochem 77:345–360. https://doi.org/10.1002/(SICI)1097-4644(20000601)77:3%3c345:AID-JCB1%3e3.0.CO;2-P
Huang J, DeGraves FJ, Lenz SD, Gao D, Feng P, Li D, Schlapp T, Kaltenboeck B (2002) The quantity of nitric oxide released by macrophages regulates Chlamydia-induced disease. Proc Natl Acad Sci 99:3914–3919. https://doi.org/10.1073/pnas.062578399
Huang Y, Marton LJ, Woster PM, Casero RA (2009) Polyamine analogues targeting epigenetic gene regulation. Essays Biochem 46:95–110. https://doi.org/10.1042/bse0460007
Iniesta V, Gómez-Nieto LC, Corraliza I (2001) The inhibition of arginase by Nω-hydroxy-l-arginine controls the growth of Leishmania inside macrophages. J Exp Med 193:777–784. https://doi.org/10.1084/jem.193.6.777
Jain V (2018) Role of polyamines in asthma pathophysiology. Med Sci (Basel, Switzerland) 6(1):4. https://doi.org/10.3390/medsci6010004
Jiang F, Gao Y, Dong C, Xiong S (2018) ODC1 inhibits the inflammatory response and ROS-induced apoptosis in macrophages. Biochem Biophys Res Commun 504:734–741. https://doi.org/10.1016/J.BBRC.2018.09.023
Kakuda DK, Sweet MJ, Mac Leod CL, Hume DA, Markovich D (1999) CAT2-mediated l-arginine transport and nitric oxide production in activated macrophages. Biochem J 340(2):549–553. https://doi.org/10.1042/bj3400549
Kaparakis M, Walduck AK, Price JD, Pedersen JS, van Rooijen N, Pearse MJ, Wijburg OLC, Strugnell RA (2008) Macrophages are mediators of gastritis in acute Helicobacter pylori infection in C57BL/6 mice. Infect Immun 76:2235–2239. https://doi.org/10.1128/IAI.01481-07
Lass A, Zimmermann R, Haemmerle G, Riederer M, Schoiswohl G, Schweiger M, Kienesberger P, Strauss JG, Gorkiewicz G, Zechner R (2006) Adipose triglyceride lipase-mediated lipolysis of cellular fat stores is activated by CGI-58 and defective in Chanarin–Dorfman Syndrome. Cell Metab 3:309–319. https://doi.org/10.1016/j.cmet.2006.03.005
Lech M, Anders H-J (2013) Macrophages and fibrosis: how resident and infiltrating mononuclear phagocytes orchestrate all phases of tissue injury and repair. Biochim Biophys Acta Mol Basis Dis 1832:989–997. https://doi.org/10.1016/j.bbadis.2012.12.001
Lewis ND, Asim M, Barry DP, Singh K, de Sablet T, Boucher J-L, Gobert AP, Chaturvedi R, Wilson KT (2010) Arginase II restricts host defense to Helicobacter pylori by attenuating inducible nitric oxide synthase translation in macrophages. J Immunol 184(5):2572–2582. https://doi.org/10.4049/jimmunol.0902436
Lewis ND, Asim M, Barry DP, de Sablet T, Singh K, Piazuelo MB, Gobert AP, Chaturvedi R, Wilson KT (2011) Immune evasion by Helicobacter pylori is mediated by induction of macrophage arginase II. J Immunol 186:3632–3641. https://doi.org/10.4049/jimmunol.1003431
Liao C-P, Lasbury ME, Wang S-H, Zhang C, Durant PJ, Murakami Y, Matsufuji S, Lee C-H (2009) Pneumocystis mediates overexpression of antizyme inhibitor resulting in increased polyamine levels and apoptosis in alveolar macrophages. J Biol Chem 284:8174–8184. https://doi.org/10.1074/jbc.M805787200
Lichtnekert J, Kawakami T, Parks WC, Duffield JS (2013) Changes in macrophage phenotype as the immune response evolves. Curr Opin Pharmacol 13:555–564. https://doi.org/10.1016/j.coph.2013.05.013
Linsalata M, Orlando A, Russo F (2014) Pharmacological and dietary agents for colorectal cancer chemoprevention: effects on polyamine metabolism. Int J Oncol 45:1802–1812. https://doi.org/10.3892/ijo.2014.2597
Mandal A, Das S, Kumar A, Roy S, Verma S, Ghosh AK, Singh R, Abhishek K, Saini S, Sardar AH, Purkait B, Kumar A, Mandal C, Das P (2017) L-arginine uptake by cationic amino acid transporter promotes intra-macrophage survival of Leishmania donovani by enhancing arginase-mediated polyamine synthesis. Front Immunol 8:839. https://doi.org/10.3389/fimmu.2017.00839
Martinez FO, Gordon S (2014) The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep 6:13. https://doi.org/10.12703/P6-13
Miao H, Ou J, Peng Y, Zhang X, Chen Y, Hao L, Xie G, Wang Z, Pang X, Ruan Z, Li J, Yu L, Xue B, Shi H, Shi C, Liang H (2016) Macrophage ABHD5 promotes colorectal cancer growth by suppressing spermidine production by SRM. Nat Commun 7:11716. https://doi.org/10.1038/ncomms11716
Mosser DM (2003) The many faces of macrophage activation. J Leukoc Biol 73:209–212. https://doi.org/10.1189/jlb.0602325
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8:958–969. https://doi.org/10.1038/nri2448
Murray PJ, Wynn TA (2011) Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol 11:723–737. https://doi.org/10.1038/nri3073
Pasini A, Caldarera CM, Giordano E (2014) Chromatin remodeling by polyamines and polyamine analogs. Amino Acids 46:595–603. https://doi.org/10.1007/s00726-013-1550-9
Peek RM Jr, Fiske C, Wilson KT (2010) Role of innate immunity in Helicobacter pylori-induced gastric malignancy. Physiol Rev 90:831–858. https://doi.org/10.1152/physrev.00039.2009
Pegg AE (2006) Regulation of ornithine decarboxylase. J Biol Chem 281:14529–14532. https://doi.org/10.1074/jbc.R500031200
Pegg AE (2009) Mammalian polyamine metabolism and function. IUBMB Life 61:880–894. https://doi.org/10.1002/iub.230
Pegg AE, McCann PP (1982) Polyamine metabolism and function. Am J Physiol Physiol 243:C212–C221. https://doi.org/10.1152/ajpcell.1982.243.5.C212
Saulnier Sholler GL, Gerner EW, Bergendahl G, MacArthur RB, VanderWerff A, Ashikaga T, Bond JP, Ferguson W, Roberts W, Wada RK, Eslin D, Kraveka JM, Kaplan J, Mitchell D, Parikh NS, Neville K, Sender L, Higgins T, Kawakita M et al (2015) A Phase I trial of DFMO targeting polyamine addiction in patients with relapsed/refractory neuroblastoma. PLoS ONE 10:e0127246–e0127246. https://doi.org/10.1371/journal.pone.0127246
Shlyueva D, Stampfel G, Stark A (2014) Transcriptional enhancers: from properties to genome-wide predictions. Nat Rev Genet 15:272–286. https://doi.org/10.1038/nrg3682
Singh K, Coburn LA, Asim M, Barry DP, Allaman MM, Shi C, Washington MK, Luis PB, Schneider C, Delgado AG (2018) Ornithine decarboxylase in macrophages exacerbates colitis and promotes colitis-associated colon carcinogenesis by impairing M1 immune responses. Cancer Res 78:4303–4315. https://doi.org/10.1158/0008-5472.CAN-18-0116
Strauss-Ayali D, Conrad SM, Mosser DM (2007) Monocyte subpopulations and their differentiation patterns during infection. J Leukoc Biol 82:244–252. https://doi.org/10.1189/jlb.0307191
Szabó C, Southan GJ, Wood E, Thiemermann C, Vane JR (1994) Inhibition by spermine of the induction of nitric oxide synthase in J774.2 macrophages: requirement of a serum factor. Br J Pharmacol 112:355–356. https://doi.org/10.1111/j.1476-5381.1994.tb13078.x
Van den Bossche J, Bogaert P, van Hengel J, Guérin CJ, Berx G, Movahedi K, Van den Bergh R, Pereira-Fernandes A, Geuns JMC, Pircher H, Dorny P, Grooten J, De Baetselier P, Van Ginderachter JA (2009) Alternatively activated macrophages engage in homotypic and heterotypic interactions through IL-4 and polyamine-induced E-cadherin/catenin complexes. Blood 114:4664–4674. https://doi.org/10.1182/blood-2009-05-221598
Van den Bossche J, Lamers WH, Koehler ES, Geuns JMC, Alhonen L, Uimari A, Pirnes-Karhu S, Van Overmeire E, Morias Y, Brys L, Vereecke L, De Baetselier P, Van Ginderachter JA (2012) Pivotal advance: arginase-1-independent polyamine production stimulates the expression of IL-4-induced alternatively activated macrophage markers while inhibiting LPS-induced expression of inflammatory genes. J Leukoc Biol 91:685–699. https://doi.org/10.1189/jlb.0911453
Wilson KT, Crabtree JE (2007) Immunology of Helicobacter pylori: insights into the failure of the immune response and perspectives on vaccine studies. Gastroenterology 133:288–308. https://doi.org/10.1053/j.gastro.2007.05.008
Wroblewski LE, Peek RM Jr, Wilson KT (2010) Helicobacter pylori and gastric cancer: factors that modulate disease risk. Clin Microbiol Rev 23:713–739. https://doi.org/10.1128/CMR.00011-10
Wynn TA, Chawla A, Pollard JW (2013) Macrophage biology in development, homeostasis and disease. Nature 496:445–455. https://doi.org/10.1038/nature12034
Yang L, Zhang Y (2017) Tumor-associated macrophages: from basic research to clinical application. J Hematol Oncol 10:58. https://doi.org/10.1186/s13045-017-0430-2
Yeramian A, Martin L, Serrat N, Arpa L, Soler C, Bertran J, McLeod C, Palacín M, Modolell M, Lloberas J (2006) Arginine transport via cationic amino acid transporter 2 plays a critical regulatory role in classical or alternative activation of macrophages. J Immunol 176:5918–5924. https://doi.org/10.4049/jimmunol.176.10.5918
Zhang M, Caragine T, Wang H, Cohen PS, Botchkina G, Soda K, Bianchi M, Ulrich P, Cerami A, Sherry B, Tracey KJ (1997) Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: a counterregulatory mechanism that restrains the immune response. J Exp Med 185:1759–1768. https://doi.org/10.1084/jem.185.10.1759
Zhou S, Gu J, Liu R, Wei S, Wang Q, Shen H, Dai Y, Zhou H, Zhang F, Lu L (2018) Spermine alleviates acute liver injury by inhibiting liver-resident macrophage pro-inflammatory response through ATG5-dependent autophagy. Front Immunol 9:948. https://doi.org/10.3389/fimmu.2018.00948
Acknowledgements
We thank the members of the Wilson Lab for their work related to the original research summarized in this article.
Funding
This work was funded by NIH Grants R01AT004821, R01CA190612, P01CA116087, and P01CA028842 (K.T.W.), Merit Review Grant I01BX001453 from the United States Department of Veterans Affairs Biomedical Laboratory R&D (BLRD) Service (K.T.W.), the Vanderbilt Digestive Disease Research Center, supported by NIH Grant P30DK058404, the Vanderbilt Ingram Cancer Center, supported by NIH Grant P30CA068485, Department of Defense Grant W81XWH-18-1-0301 (K.T.W.), the Thomas F. Frist Sr. Endowment (K.T.W.), and the Vanderbilt Center for Mucosal Inflammation and Cancer (K.T.W.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no conflict of interest exists.
Human rights and participants
All studies from the Wilson Lab that are referenced here were conducted under proper ethical standards for either animal or human research.
Additional information
Handling Editor: E. Agostinelli.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Latour, Y.L., Gobert, A.P. & Wilson, K.T. The role of polyamines in the regulation of macrophage polarization and function. Amino Acids 52, 151–160 (2020). https://doi.org/10.1007/s00726-019-02719-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-019-02719-0