Abstract
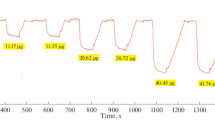
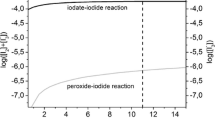
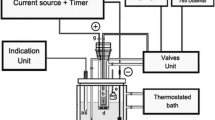
This paper describes a method of determining the purity of potassium iodate by constant-current coulometry. The determination can be divided into two steps. First, a Na2S2O3 solution is prepared and its reductive value is determined. Second, the purity value of an oxidimetric quantity of potassium iodate is determined. This paper discusses the conditions of the reaction process and evaluates type A and B standard uncertainty of this method. The expanded relative uncertainty of this method is 0.02% (k=2).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 15 June 2000 Accepted: 27 December 2001
Rights and permissions
About this article
Cite this article
Ma, L. Determination of the purity of potassium iodate by constant-current coulometry. Accred Qual Assur 7, 163–167 (2002). https://doi.org/10.1007/s00769-002-0441-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00769-002-0441-7