Abstract
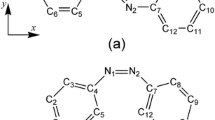
A theoretical analysis of the linear and nonlinear optical properties of six push–pull π-conjugated molecules with stilbene, azobenzene and benzilideneaniline as a backbone is presented. The photophysical properties of the investigated systems were determined by using response functions combined with density functional theory (DFT). Several different exchange-correlation potentials were applied in order to determine parameters describing the one- and two-photon spectra of the studied molecules. In particular, the recently proposed Coulomb-attenuated model (CAM-B3LYP) was used to describe charge-transfer (CT) excited states. In order to compare theoretical predictions with available experimental data, calculations with inclusion of solvent effects were performed. The BLYP and the CAM-B3LYP functionals were found to yield values of two-photon absorption (TPA) probabilities closer to experimental values than the B3LYP functional or the HF wavefunction. Moreover, molecular static hyperpolarisabilities were determined using both DFT and second-order Møller-Plesset perturbation (MP2) theory. Likewise, the CAM-B3LYP functional was found to outperform other applied exchange-correlation potentials in determining first hyperpolarisability (β). Moreover, it was confirmed on a purely theoretical basis that the presence of a –C=C– bridge between the phenyl rings leads to a much larger nonlinear optical response in comparison with a –N=N– bridge.

Similar content being viewed by others
References
Skotheim TJ (1986) Handbook of conducting polymers. Dekker, New York
Chemla DS, Zyss J (1987) Nonlinear optical properties of organic molecules and crystals. Academic, New York
Prasad PN, Williams DJ (1991) Introduction to nonlinear optical effects in molecules and polymers. Wiley, New York
Nalwa HS, Siezo M (1994) Nonlinear optics of organic molecules and polymers. CRC, Boco Raton
Zyss J (1994) Molecular nonlinear optics: materials, physics and devices. Academic, New York
Dalton LR (2001) Nonlinear optical polymeric materials: from chromophore design to commercial applications. Advances in polymer science, vol 158. Springer, Heidelberg
Dalton LR (2001) The role of nonlinear optical devices in the optical communications age. Kluwer, Dordrecht
Marde SRR, Perry JW (1994) Science 263:1706–1707
Denning RG (1995) J Mater Chem 5:365–378
Goonesekera A, Ducharme S (1999) J Appl Phys 85:6506
Stahelin M, Burland DM, Rice JE (1992) Chem Phys Lett 191:245–250
De Boni L, Piovesan E, Misoguti L, Zilio SC, Mendonca CR (2007) J Phys Chem A 111:6222–6224
Oliveira SL, Correa DS, Misoguti L, Constantino CJL, Aroca RF, Zilio SC, Mendonca CR (2005) Adv Mater 17:1890–1893
De Boni L, Misoguti L, Zilio SC, Mendonca CR (2005) Chem Phys Chem 6:1121–1125
De Boni L, Constantino CJL, Misoguti L, Aroca RF, Zilio SC, Mendonca CR (2003) Chem Phys Lett 371:744–749
van Walree CA, Franssen O, Marsman AW, Flipse MC, Jenneskens LW (1997) J Chem Soc Perkin Trans 2:799–807
van Walree CA, Marsman AW, Flipse MC, Jenneskens LW, Smeets WJJ, Spek AL (1997) J Chem Soc Perkin Trans 2:809–819
Baev A, Prasad PN, Samoc M (2005) J Chem Phys 122:224309
Chandra Jha P, Anusooya Pati Y, Ramasesha S (2005) Mol Phys 14:1859–1873
Day PN, Nguyen KA, Pachter R (2006) J Chem Phys 125:094103
Suponitsky KY, Tafur S, Masunov AE (2008) J Chem Phys 129:044109
Champagne B (1996) Chem Phys Lett 261:57–65
Jacquemin D, André J, Perpéte B (2004) J Chem Phys 121:4389–4396
Antonov L, Kamada K, Ohta K, Kamounah FS (2003) Phys Chem Chem Phys 5:1193–1197
Ohta K, Antonov L, Yamada S, Kamada K (2007) J Chem Phys 127:084504–084515
Oudar JL, Chemla DS (1977) J Chem Phys 66:2664–2668
Kawata S, Kawata Y (2000) Chem Rev 100:1777–1788
Delaire JA, Nakatani K (2000) Chem Rev 100:1817–1846
Adamo C, Scuseria GE, Barone V (1999) J Chem Phys 111:2889–2899
Jamorski-Jödicke C, Lüthi HP (2002) J Chem Phys 117:4146–4156
Cavillot V, Champagne B (2002) Chem Phys Lett 354:449–457
Becke AD (1993) J Chem Phys 98:5648–5652
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865–3868
Yanai T, Tew DP, Handy NC (2004) Chem Phys Lett 393:51–57
Tawada Y, Tsuneda T, Yanagisawa S, Yanai T, Hirao K (2004) J Chem Phys 120:8425–8433
Peach MJG, Benfield P, Helgaker T, Tozer DJ (2008) J Chem Phys 128:044118
Yanai T, Harrison RJ, Handy NC (2005) Mol Phys 103:413–424
Peach MJG, Cohen AJ, Tozer DJ (2006) Phys Chem Chem Phys 8:4543–4549
Iikura H, Tsuneda T, Yanai T, Hirao K (2001) J Chem Phys 115:3540–3544
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, revision C.02. Gaussian, Wallingford CT
DALTON (2005) A molecular electronic structure program, Release 2.0 see http://www.kjemi.uio.no/software/dalton/dalton.html
Schmidt MW, Baldridge KK, Boatz JA, Elbert St, Gordon MS, Jensen JH, Koseki S, Matsunaga N, Nguyen KA, Su S, Windus TL, Dupuis M, Montgomery JA Jr (1993) The general atomic and molecular electronic structure system. J Comput Chem 14:1347–1363
Hättig C, Hald K (2002) Phys Chem Chem Phys 4:2111–2118
Hättig C, Köhn A (2002) J Chem Phys 117:6939–6951
Fliegl H, Köhn A, Hättig C, Ahlrichs R (2003) J Am Chem Soc 125:9821–9827
Zaleśny R, Bartkowiak W, Styrcz S, Leszczynski J (2002) J Phys Chem A 106:4032–4037
Krawczyk P, Kaczmarek A, Zaleśny R, Matczyszyn K, Bartkowiak W, Ziółkowski M, Cysewski P (2009) J Mol Model 15:581–590
Zaleśny R, Matczyszyn K, Kaczmarek A, Bartkowiak W, Cysewski P (2007) J Mol Model 13:785–791
Chen PC, Chiech YC (2003) Theochem 624:191–200
Rau H (1973) Angew Chem Int Ed Engl 12:224–235
Jacquemin D, Bouhy M, Perpéte EA (2006) J Chem Phys 124:204321
Jacquemin D, Preat J, Wahtelet V, Fontaine M, Perpéte EA (2005) J Am Chem Soc 128:2072–2083
Peach MJG, Helgaker T, Sałek P, Keal TW, Lutnaes OB, Tozer DJ, Handy NC (2006) Phys Chem Chem Phys 8:558–562
Jacquemin D, Perpéte EA, Scalmani G, Frisch MJ, Kobayashi R, Adamo C (2008) J Chem Phys 126:144105
Jacquemin, Perpéte EA, Scuseria GE, Ciofini I, Adamo C (2008) Chem Phys Lett 465:226–229
Liptay W (1974) In: Lim EC (ed) Excited states, vol 1. Academic, New York, p 129
Ch JP, Wang Y, Ågren H (2008) Chem Phys Chem 9:111–116
Day PN, Nguyen KA, Pachter R (2005) J Phys Chem B 109:1803–1814
Zaleśny R, Wójcik G, Mossakowska I, Bartkowiak W, Avramopoulos A, Papadopoulos MG (2009) Theochem 907:46–50
Medved M, Noga J, Jacquemin D, Assfeld X, Perpéte EA (2007) Theochem 821:160–165
Acknowledgements
Computational grants from the Poznan Supercomputing and Networking Center (PCSS) and ACK CYFRONET AGH are acknowledged. The author thanks Dr. Żaneta Czyżnikowska for computing excited state dipole moments using the CC2 approach.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krawczyk, P. DFT study of linear and nonlinear optical properties of donor-acceptor substituted stilbenes, azobenzenes and benzilideneanilines. J Mol Model 16, 659–668 (2010). https://doi.org/10.1007/s00894-009-0623-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-009-0623-x