Abstract
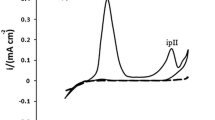
The inhibiting behavior of bis-(2-benzothiazolyl)-disulfide on mild steel corrosion was evaluated in 1 M HCl solution. Weight loss, potentiodynamic polarization, and electrochemical impedance spectroscopy (EIS) techniques were used in this study. In EIS technique, by performing Kramers–Kronig transformations, the experimental results validated, before fitting any theoretical model on them. The obtained results showed that the inhibition efficiency (%IE) increases by increasing the concentration of bis-(2-benzothiazolyl)-disulfide up to 2.02 × 10−4 M (%IE ∼ 62–68). Between the results obtained from various used techniques, a good agreement was found. Polarization curves indicate that the inhibition of the inhibitor is a mixed anodic–cathodic nature and Langmuir isotherm is found as an accurate isotherm describing the adsorption behavior. It also found that its inhibition mechanism of bis-(2-benzothiazolyl)-disulfide is at the interval of physical adsorption and chemical binding. The inhibition efficiency of the inhibitor decreased by rising of temperature in the range of 25–55 °C and these results, verified adsorption behavior of the inhibitor.






Similar content being viewed by others
References
Ashassi-Sorkhabi H, Eshaghi M (2009) J Solid State Electrochem 13:1297
Abdeli M, Parvini-Ahmadi N, Khosroshahi RA (2009) J Solid State Electrochem 14:1317
Abdeli M, Parvini-Ahmadi N, Khosroshahi RA (2009) Corrosion inhibition of mild steel in hydrochloric acid by 6Aminohexanoic acid. Mater Corros 61:147–151
Abdel Rehim SS, Hazzazi OA, Amin MA, Khaled FK (2008) Corros Sci 50:2258
Machnikova E, Whitmire KH, Hackerman N (2008) Electrochim Acta 53:6024
Xianghong L, Shuduan D, Hui F, Guannan M (2009) Corros Sci 51:620
Ashassi-Sorkhabi H, Eshaghi M (2009) Mater Chem Phys 114:267
Ashassi-Sorkhabi H, Seifzadeh D, Hosseini MG (2008) Corros Sci 50:3363
Ashassi-Sorkhabi H, Seifzadeh D (2008) J Appl Electrochem 38:1545
Ashassi-Sorkhabi H, Masoumi B, Ejbari P, Asghari E (2009) Corrosion inhibition of mild steel in acidic media by Basic yellow 13 dye. J Appl Electrochem 39:1497–1501
Hamdy HH (2007) Electrochim Acta 53:1722
Bentiss F, Traisnel M, Gengembre L, Lagrenée M (1999) Appl Surf Sci 152:237
Popova A, Sokolova E, Raicheva S, Christov M (2003) Corros Sci 45:33
Solmaz R, Kardas G, Yazıcı B, Erbil M (2008) Colloids Surf A 312:7
El Azhar M, Mernari B, Traisnel M, Bentiss F, Lagrenée M (2001) Corros Sci 43:2229
Ramesh S, Rajeswari S (2004) Electrochim Acta 49:811
Elkadi L, Mernari B, Traisnel M, Bentiss F, Lagrenée M (2000) Corros Sci 42:703
Popova A, Christov M, Zwetanova A (2007) Corros Sci 49:2131
Weihua L, Qiao H, Changling P, Baorong H (2007) Electrochim Acta 52:6386
Qing Q, Shuan J, Wei B, Lei L (2007) Electrochim Acta 52:6811
Zhang J, Monteiro PJM (2001) Cem Concr Res 31:603
Bruzzoni P, Carranza RM, Collet Lacoste JR, Crespo EA (2002) Electrochim Acta 48:341
Priyantha N, Jayaweera P, Macdonald DD, Sun A (2004) J Electroanal Chem 572:409
Hassanzadeh A (2007) Corros Sci 49:1895
Avci G (2008) Colloids Surf A 317:730
Behpour M, Ghoreishi SM, Soltani N, Salavati-Niasari M, Hamadanian M, Gandomi A (2008) Corros Sci 50:2172
Libin TL, Xueming L, Lin L, Qing Q, Guannan M, Guangheng L (2005) Mater Chem Phys 94:353
Rammelt U, Koehler S, Reinhard G (2008) Corros Sci 50:1659
Acknowledgment
The authors thank the Material Faculty of Sahand University of Technology for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdeli, M., Parvini Ahmadi, N. & Azari Khosroshahi, R. Influence of bis-(2-benzothiazolyl)-disulfide on corrosion inhibition of mild steel in hydrochloric acid media. J Solid State Electrochem 15, 1867–1873 (2011). https://doi.org/10.1007/s10008-010-1162-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-010-1162-1