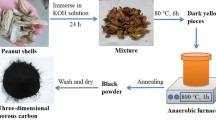
Abstract
Porous carbons prepared by carbonization of solid leather wastes were employed for supercapacitor applications using KOH electrolyte. Advanced instrumentation techniques were used to determine the physicochemical properties of the porous carbon samples. Three-electrode configuration was used to investigate the electrochemical properties of porous carbon electrodes through cyclic voltammetry studies, galvanostatic charge–discharge tests, electrochemical impedance spectroscopy (EIS) techniques in 1 M KOH electrolyte. Porous carbons exhibit high specific surface areas in a range of 613–716 m2 g−1, as well as superior energy storage capacity as electrodes for supercapacitors. Porous carbon electrodes activated at 900 °C exhibited specific capacitance up to 1800 F/g in alkali medium. All the solid leather waste carbons established stable cycle performance over 5000 cycles. These desirable capacitive performances enable solid leather waste-derived carbons as a source of new materials for low-cost energy storage devices as well as supercapacitors. This paper put forwards a new concept of ‘waste to value-added products’ that can be a helping hand for leather industries and its solid waste management disposal problems. The highlights of this research work suggest that leather industries could implement and make it feasible for commercialization.
Graphical Abstract








Similar content being viewed by others
Abbreviations
- AgCl:
-
Silver chloride
- BET:
-
Brunauer–Emmett–Teller
- BJH:
-
Barrett–Joyner– Halenda
- C6:
-
Porous carbon activated at 600 °C
- C7:
-
Porous carbon activated at 700 °C
- C8:
-
Porous carbon activated at 800 °C
- C9:
-
Porous carbon activated at 900 °C
- CP:
-
Precarbonized carbon prepared from solid leather waste
- Cu:
-
Copper
- CV:
-
Cyclic voltammetry
- EIS:
-
Electrochemical impedance spectroscopy
- GCD:
-
Galvanostatic charge–discharge
- H2SO4 :
-
Sulfuric acid
- H3PO4 :
-
Phosphoric acid
- HCl:
-
Hydrochloric acid
- HK:
-
Horvath–Kawazoe
- HR-TEM:
-
High-resolution transmission electron microscopy
- K :
-
Potassium
- K2SO4 :
-
Potassium sulfate
- KOH:
-
Potassium hydroxide
- OH:
-
Hydroxide
- pH:
-
Power of hydrogen
- PPI:
-
Pores per inch
- SAED:
-
Selected area electron diffraction pattern
- SEM:
-
Scanning electron microscopy
- XRD:
-
X-ray diffraction
- Ag:
-
Silver
- C:
-
Capacitance in Farad per grams
- Cr:
-
Chromium
- d :
-
Spacing of lattice planes
- ESR:
-
Equivalent series resistance
- f :
-
Frequency
- Hz:
-
Hertz
- I :
-
Current in amperes
- Kα :
-
K-alpha radiation
- m :
-
Mass in grams
- °C:
-
Temperature in degree celsius
- R :
-
Resistance in ohms
- t :
-
Time in seconds
- v :
-
Scan rate in volt per seconds
- V :
-
Voltage in volts
- V − V 0 :
-
Potential window
- W :
-
Warburg impendence in ohms
- Z :
-
Impedance
- ∆:
-
Difference operator
- θ :
-
Angle in degrees
- λ :
-
Wavelength
- Ώ:
-
Ohms unit of resistance
- −:
-
Negative ions
- +:
-
Positive ions
- °:
-
Degree
- ct:
-
Charge transfer resistance
- im:
-
Imaginary part of impedance
- k :
-
Knee frequency
- re :
-
Real part of impedance
- s :
-
Solution resistance
- sp:
-
Specific capacitance
References
Andreas HA, Lussier K, Oickle AM (2009) Effect of Fe-contamination on rate of self-discharge in carbon-based aqueous electrochemical capacitors. J Power Sources 187:275–283. doi:10.1016/j.jpowsour.2008.10.096
Cassano A, Drioli E, Molinari R, Bertolutti C (1997) Quality improvement of recycled chromium in the tanning operation by membrane processes. Desalination 108:193–203. doi:10.1016/S0011-9164(97)00027-1
Fic K, Lota G, Meller M, Frackowiak E (2012) Novel insight into neutral medium as electrolyte for high-voltage supercapacitors. Energy Environ Sci 5:5842–5850. doi:10.1039/C1EE02262H
Frackowiak E, Beguin F (2001) Carbon materials for the electrochemical storage of energy in capacitors carbon. Carbon 39:937–950. doi:10.1016/S0008-6223(00)00183-4
Hayashi J, Kazehaya A, Muroyama K, Watkinson AP (2000) Carbon materials for the electrochemical storage of energy in capacitors. Carbon 38:1873–1878. doi:10.1016/S0008-6223(00)00027-0
Jurcakova DH, Puziy AM, Poddubnaya OI, Garcia FS, Tascon JMD, Lu GQ (2009) Highly stable performance of supercapacitors from phosphorus-enriched carbons. J Am Chem Soc 131:5026–5027. doi:10.1021/ja809265m
Jurewicz K, Delpeux S, Bretagne V, Beguin F, Frackowiak F (2001) Supercapacitors from nanotubes/polypyrrole composites. Chem Phys Lett 347:36–40. doi:10.1016/S00092614(01)01037-5
Kantarli IC, Yanik J (2010) Activated carbon from leather shaving wastes and its application in removal of toxic materials. J Hazard Mater 179:348–356. doi:10.1016/j.jhazmat.2010.03.012
Kong J, Yue Q, Huang L, Gao Y, Sun Y, Gao B, Li Q, Wang Y (2013) Preparation, characterization and evaluation of adsorptive properties of leather waste based activated carbon via physical and chemical activation. Chem Eng J 221:62–71. doi:10.1016/j.cej.2013.02.021
Kotz R, Carlen M (2000) Principles and applications of electrochemical capacitors. Electrochim Acta 45:2483–2498. doi:10.1016/S0013-4686(00)00354-6
Niketha K, Kennedy LJ, Vijaya JJ (2016a) Preparation and characterization of hierarchical porous carbons derived from solid leather waste for supercapacitor applications. J Hazard Mater 318:173–185. doi:10.1016/j.jhazmat.2016.06.037
Niketha K, Kennedy LJ, Udaya A, Vijaya JJ (2016b) Electrical conductivity studies of nanoporous carbon derived from leather waste: effect of pressure, temperature and porosity. J Nanosci Nanotechnol 16:8829–8838. doi:10.1166/jnn.2016.11652
Nithya VD, Kalaiselvan R, Kalpana D, Vasylechko L (2013) Synthesis of Bi2WO6 nanoparticles and its electrochemical properties in different electrolytes for pseudocapacitor electrodes. Electrochim Acta 109:720–731. doi:10.1016/j.electacta.2013.07.138
Oickle AM, Andreas HA (2011) Examination of water electrolysis and oxygen reduction as self-discharge mechanisms for carbon-based, aqueous electrolyte electrochemical capacitors. J Phys Chem C 115:4283–4288. doi:10.1021/jp1067439
Perret P, Khani Z, Brousse T, Belanger D, Guay D (2011) Carbon/PbO2 asymmetric electrochemical capacitor based on methane sulfonic acid electrolyte. Electrochim Acta 56:8122–8128. doi:10.1016/j.electacta.2011.05.125
Petruzelli D, Passino R, Tiravanti G (1995) Ion exchange process for chromium removal and recovery from tannery wastes. Ind Eng Chem 34:2612–2617. doi:10.1021/ie00047a009
Sreeram KJ, Saravanabhavan S, Rao JR, Nair BU (2004) Use of chromium-collagen wastes for the removal of tannins from wastewaters. Ind Eng Chem Res 43:5310–5317. doi:10.1021/ie034273p
Sugimoto W, Shibutani T, Murakami Y, Takasu Y (2002) Charge storage capabilities of rutile-type RuO2VO2 solid solution for electrochemical supercapacitors. Electrochem Solid-State Lett 5:170–172. doi:10.1149/1.1483155
Tang Y, Chen T, Yu S, Qiao Y, Mu S, Zhang S, Zhao Y, Hou L, Huang W, Gao F (2015) A highly electronic conductive cobalt nickel sulphide dendrite/quasi-spherical nanocomposite for a supercapacitor electrode with ultrahigh areal specific capacitance. J Power Sources 295:314–322. doi:10.1016/j.jpowsour.2015.07.035
Tsay KC, Zhang L, Zhang J (2012) Effects of electrode layer composition/thickness and electrolyte concentration on both specific capacitance and energy density of supercapacitor. Electrochim Acta 60:428–436. doi:10.1016/j.electacta.2011.11.087
Wang C, Zhou Y, Sun L, Wan P, Zhang X, Qiu J (2013a) Sustainable synthesis of phosphorus- and nitrogen-co-doped porous carbons with tunable surface properties for supercapacitors. J Power Sources 239:81–88. doi:10.1016/j.jpowsour.2013.03.126
Wang XJ, Yan ZZ, Pang H, Wang WQ, Li GC, Ma YH, Zhang H, Li XX, Chen J (2013b) NH4CoPO4·H2O micro flowers and porous Co2P2O micro flowers: effective electrochemical supercapacitor behavior in different alkaline electrolytes. Int J Electrochem Sci 8: 3768–3785. http://www.electrochemsci.org/papers/vol8/80303768.pdf
Wang RY, Li Q, Cheng LL, Li HL, Wang BY, Zhao SX, Guo PZ (2014) Electrochemical properties of manganese ferrite-based supercapacitors in aqueous electrolyte: the effect of ionic radius. Colloids Surf A 457:94–99. doi:10.1016/j.colsurfa.2014.05.059
Wenzel BM, Zimmer TH, Fernandez CS, Marcilio NR, Godinho M (2012) Aluminothermic reduction of Cr2O3 contained in the ash of thermally treated leather waste. Braz J Chem Eng 30:141–154. doi:10.1590/S0104-66322013000100016
Yılmaz O, Kantarli IC, Yuksel M, Saglam M, Yanik J (2007) Conversion of leather wastes to useful products. Resour Conserv Recycl 49:436–448. doi:10.1016/j.resconrec.2006.05.006
Zhong C, Deng Y, Hu W, Qiao J, Zhang L, Zhang J (2015) A review of electrolyte materials and compositions for electrochemical supercapacitors. Chem Soc Rev 44(21):7484–7539. doi:10.1039/c5cs00303b
Acknowledgments
The authors express thanks to authorities and staff of Sri Chamundi Leathers, Kolappakkam, Chennai, for their valuable help. The first author authentically thanks the VIT University Chennai management for granting the Teaching Research Associateship for carrying out the research work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Konikkara, N., Punithavelan, N., Kennedy, L.J. et al. A new approach to solid waste management: fabrication of supercapacitor electrodes from solid leather wastes using aqueous KOH electrolyte. Clean Techn Environ Policy 19, 1087–1098 (2017). https://doi.org/10.1007/s10098-016-1301-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-016-1301-1