Abstract
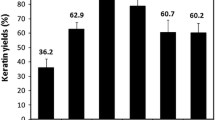
Feathers biomass from poultry industry is considered as an important waste product, which creates serious environmental problems. In this study, keratin was extracted from waste chicken feathers using sodium sulfide as a reducing agent under optimized conditions. The extracted keratin particles were used to develop a bioploymeric film by adding microcrystalline cellulose as nano-additive agent. The calculated yield of 80.2% was obtained for keratin from feathers dry weight 25 g (w/w). The extracted keratin was characterized using Fourier transform infrared spectroscopy, scanning electron microscopy (SEM), thermogravimetric analysis, differential scanning calorimetry, wide-angle X-ray diffraction. The physiochemical characteristics of the feathers were compared with the keratin powder. The regenerated keratin particles preserved their chemical composition, thermal strength and stability after chemical extraction. The extracted keratin particles showed 10–20-µm spongy porous microparticles in SEM analysis. The keratin powder was used to synthesize a bioplastic film using glycerol (3.5%) and microcrystalline cellulose (0.2%) in NaOH for 48 h at 60 °C. The calculated thickness of bioplastic film was 1.12 × 10−4 mm with tensile strength of 3.62 ± 0.6 MPa. The Young’s modulus and break elongation for synthesized bioplastic film were 1.52 ± 0.34 MPa and 15.8 ± 2.2%, respectively. The feather and keratin showed maximum similarity index of 64.74% (l-alanyl, l-alanyl, l-alanine, p-nitroanilide) and 64.32% with d-pantethine, respectively, using OMNIC Specta software. Overall, the study presented a highly efficient method to convert the waste feather biomass into a bioplastic film which can be used in biopolymer, biomedical and pharmaceutical industries.








Similar content being viewed by others
References
Sharma S et al (2016) Extraction and characterization of keratin from chicken feather waste biomass: a study
Aluigi A, Zoccola M, Vineis C, Tonin C, Ferrero F, Canetti M (2007) Study on the structure and properties of wool keratin regenerated from formic acid. Int J Biol Macromol 41:266–273
Aluigi A, Vineis C, Varesano A, Mazzuchetti G, Ferrero F, Tonin C (2008) Structure and properties of keratin/PEO blend nanofibres. Eur Polym J 44:2465–2475
Arai KM, Takahashi R, Yokote Y, Akahane K (1983) Amino-acid sequence of feather keratin from fowl. Eur J Biochem 132:501–507
Barone JR (2009) Lignocellulosic fiber-reinforced keratin polymer composites. J Polym Environ 17:143–151
Barone JR, Schmidt WF (2005) Polyethylene reinforced with keratin fibers obtained from chicken feathers. Compos Sci Technol 65:173–181
Bertsch A, Coello N (2005) A biotechnological process for treatment and recycling poultry feathers as a feed ingredient. Biores Technol 96:1703–1708
Brandelli A, Sala L, Kalil SJ (2015) Microbial enzymes for bioconversion of poultry waste into added-value products. Food Res Int 73:3–12
Cao J (2000) Is the α–β transition of keratin a transition of α-helices to β-pleated sheets? Part I. In situ XRD studies. J Mol Struct 553:101–107
Cavello I, Cavalitto S, Hours R (2012) Biodegradation of a keratin waste and the concomitant production of detergent stable serine proteases from Paecilomyces lilacinus. Appl Biochem Biotechnol 167:945–958
Dahl KH, McKinley-McKee JS (1981) The reactivity of affinity labels: a kinetic study of the reaction of alkyl halides with thiolate anions—a model reaction for protein alkylation. Bioorg Chem 10:329–341
Edwards H, Hunt D, Sibley M (1998) FT-Raman spectroscopic study of keratotic materials: horn, hoof and tortoiseshell. Spectrochim Acta Part A Mol Biomol Spectrosc 54:745–757
Endo R, Kamei K, Iida I, Kawahara Y (2008) Dimensional stability of waterlogged wood treated with hydrolyzed feather keratin. J Archaeol Sci 35:1240–1246
Eslahi N, Dadashian F, Nejad NH (2013) An investigation on keratin extraction from wool and feather waste by enzymatic hydrolysis. Prep Biochem Biotechnol 43:624–648
Fang Y, Catron B, Zhang Y, Zhao L, Caruso J, Hu Q (2010) Distribution and in vitro availability of selenium in selenium-containing storage protein from selenium-enriched rice utilizing optimized extraction. J Agric Food Chem 58:9731–9738
Feughelman M, Lyman D, Willis B (2002) The parallel helices of the intermediate filaments of α-keratin. Int J Biol Macromol 30:95–96
Fu K, Griebenow K, Hsieh L, Klibanov AM, Langera R (1999) FTIR characterization of the secondary structure of proteins encapsulated within PLGA microspheres. J Controll Release 58:357–366
Garrido T, Leceta I, de la Caba K, Guerrero P (2018) Chicken feathers as a natural source of sulphur to develop sustainable protein films with enhanced properties. Int J Biol Macromol 106:523–531
Guo Y, Tang H, Li G, Xie D (2014) Effects of cow dung biochar amendment on adsorption and leaching of nutrient from an acid yellow soil irrigated with biogas slurry. Water Air Soil Pollut 225:1820
Ha S-W, Tonelli AE, Hudson SM (2005) Structural studies of Bombyx mori silk fibroin during regeneration from solutions and wet fiber spinning. Biomacromolecules 6:1722–1731
Happey F, Wormell R (1949) 53—Regenerated keratin fibres from wool. J Text Inst Trans 40:T855–T869
He M, Zhang B, Dou Y, Yin G, Cui Y (2017) Blend modification of feather keratin‐based films using sodium alginate. J Appl Polym Sci 134:44680
Idris A, Vijayaraghavan R, Rana UA, Fredericks D, Patti A, MacFarlane D (2013) Dissolution of feather keratin in ionic liquids. Green Chem 15:525–534
Idris A, Vijayaraghavan R, Patti A, MacFarlane D (2014) Distillable protic ionic liquids for keratin dissolution and recovery. ACS Sustain Chem Eng 2:1888–1894
Ji Y, Chen J, Lv J, Li Z, Xing L, Ding S (2014) Extraction of keratin with ionic liquids from poultry feather. Sep Purif Technol 132:577–583
Jones CB, Mecham D (1943) The dispersion of keratins. I. Studies on the dispersion and degradation of certain keratins by sodium sulfide. Arch Biochem 2:209
Kamarudin NB, Sharma S, Gupta A, Kee CG, Chik SMSBT, Gupta R (2017) Statistical investigation of extraction parameters of keratin from chicken feather using design-expert. 3 Biotech 7:127
Khosa MA, Ullah A (2014) In-situ modification, regeneration, and application of keratin biopolymer for arsenic removal. J Hazard Mater 278:360–371
Lasekan A, Bakar FA, Hashim D (2013) Potential of chicken by-products as sources of useful biological resources. Waste Manag 33:552–565
Lee H et al (2015) Human hair keratin-based biofilm for potent application to periodontal tissue regeneration. Macromol Res 23:300–308
Ma B, Qiao X, Hou X, Yang Y (2016) Pure keratin membrane and fibers from chicken feather. Int J Biol Macromol 89:614–621
Ma B, Chen W, Qiao X, Pan G, Jakpa W, Hou X, Yang Y (2017) Tunable wettability and tensile strength of chitosan membranes using keratin microparticles as reinforcement. J Appl Polym Sci 134:44667
Martelli SM, Moore G, Paes SS, Gandolfo C, Laurindo JB (2006a) Influence of plasticizers on the water sorption isotherms and water vapor permeability of chicken feather keratin films. LWT Food Sci Technol 39:292–301
Martelli SM, Moore GRP, Laurindo JB (2006b) Mechanical properties, water vapor permeability and water affinity of feather keratin films plasticized with sorbitol. J Polym Environ 14:215–222
Martinez-Hernandez AL, Velasco-Santos C, De Icaza M, Castano VM (2005) Microstructural characterisation of keratin fibres from chicken feathers. Int J Environ Pollut 23:162–178
Menefee E, Yee G (1965) Thermally-induced structural changes in wool. Text Res J 35:801–812
Mohanty AK, Misra M, Drzal LT (2005) Natural fibers, biopolymers, and biocomposites. CRC Press, Boca Raton
Mokrejs P, Svoboda P, Hrncirik J, Janacova D, Vasek V (2010) Processing poultry feathers into keratin hydrolysate through alkaline-enzymatic hydrolysis. Waste Manag Res 29(3):260–267
Moore GRP, Martelli SM, Gandolfo C, do Amaral Sobral PJ, Laurindo JB (2006) Influence of the glycerol concentration on some physical properties of feather keratin films. Food Hydrocoll 20:975–982
Moritz J, Latshaw J (2001) Indicators of nutritional value of hydrolyzed feather meal. Poult Sci 80:79–86
Nishikawa N, Tanizawa Y, Tanaka S, Horiguchi Y, Asakura T (1998) Structural change of keratin protein in human hair by permanent waving treatment. Polymer 39:3835–3840
Onifade A, Al-Sane N, Al-Musallam A, Al-Zarban S (1998) A review: potentials for biotechnological applications of keratin-degrading microorganisms and their enzymes for nutritional improvement of feathers and other keratins as livestock feed resources. Biores Technol 66:1–11
Pavia DL, Lampman GM, Kriz GS, Vyvyan JA (2008) Introduction to spectroscopy. Cengage Learning, Boston
Pillai C (2010) Challenges for natural monomers and polymers: novel design strategies and engineering to develop advanced polymers. Des Monomers Polym 13:87–121
Poole AJ, Church JS (2015) The effects of physical and chemical treatments on Na2S produced feather keratin films. Int J Biol Macromol 73:99–108
Poole AJ, Church JS, Huson MG (2008) Environmentally sustainable fibers from regenerated protein. Biomacromolecules 10:1–8
Poole AJ, Lyons RE, Church JS (2011) Dissolving feather keratin using sodium sulfide for bio-polymer applications. J Polym Environ 19:995–1004
Popescu C, Augustin P (1999) Effect of chlorination treatment on the thermogravimetric behaviour of wool fibres. J Therm Anal Calorim 57:509–515
Rad ZP, Tavanai H, Moradi A (2012) Production of feather keratin nanopowder through electrospraying. J Aerosol Sci 51:49–56
Ramakrishnan N, Sharma S, Gupta A, Alashwal BY (2018) Keratin based bioplastic film from chicken feathers and its characterization. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2018.01.037
Rao DR, Gupta V (1992) Crystallite orientation in wool fibers. J Appl Polym Sci 46:1109–1112
Reddy N, Yang Y (2007) Structure and properties of chicken feather barbs as natural protein fibers. J Polym Environ 15:81–87
Schrooyen PM, Dijkstra PJ, Oberthür RC, Bantjes A, Feijen J (2000) Partially carboxymethylated feather keratins. 1. Properties in aqueous systems. J Agric Food Chem 48:4326–4334
Schrooyen PM, Dijkstra PJ, Oberthür RC, Bantjes A, Feijen J (2001a) Partially carboxymethylated feather keratins. 2. Thermal and mechanical properties of films. J Agric Food Chem 49:221–230
Schrooyen PM, Dijkstra PJ, Oberthür RC, Bantjes A, Feijen J (2001b) Stabilization of solutions of feather keratins by sodium dodecyl sulfate. J Colloid Interface Sci 240:30–39
Senoz E, Wool RP (2010) Microporous carbon–nitrogen fibers from keratin fibers by pyrolysis. J Appl Polym Sci 118:1752–1765
Sharma S, Gupta A (2016) Sustainable management of keratin waste biomass: applications and future perspectives. Braz Arch Biol Technol 59:e16150684
Sharma S, Gupta A, Chik S, Kee CG, Mistry BM, Kim DH, Sharma G (2017a) Characterization of keratin microparticles from feather biomass with potent antioxidant and anticancer activities. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2017.06.015
Sharma S, Gupta A, Chik SMSBT, Kee CYG, Poddar PK (2017b) Dissolution and characterization of biofunctional keratin particles extracted from chicken feathers. In: IOP conference series: materials science and engineering, vol 1. IOP Publishing, p 012013
Tanabe T, Okitsu N, Tachibana A, Yamauchi K (2002) Preparation and characterization of keratin–chitosan composite film. Biomaterials 23:817–825
Tesfaye T, Sithole B, Ramjugernath D, Chunilall V (2017) Valorisation of chicken feathers: characterisation of physical properties and morphological structure. J Clean Prod 149:349–365
Tiwary E, Gupta R (2010) Medium optimization for a novel 58 kDa dimeric keratinase from Bacillus licheniformis ER-15: biochemical characterization and application in feather degradation and dehairing of hides. Biores Technol 101:6103–6110
Touaibia D, Benayada B (2005) Removal of mercury (II) from aqueous solution by adsorption on keratin powder prepared from Algerian sheep hooves. Desalination 186:75–80
Ullah A, Vasanthan T, Bressler D, Elias AL, Wu J (2011) Bioplastics from feather quill. Biomacromolecules 12:3826–3832
Vasconcelos A, Freddi G, Cavaco-Paulo A (2008) Biodegradable materials based on silk fibroin and keratin. Biomacromolecules 9:1299–1305
Wang Y-X, Cao X-J (2012) Extracting keratin from chicken feathers by using a hydrophobic ionic liquid. Process Biochem 47:896–899
Wojciechowska E, Włochowicz A, Wesełucha-Birczyńska A (1999) Application of Fourier-transform infrared and Raman spectroscopy to study degradation of the wool fiber keratin. J Mol Struct 511:307–318
Xu W, Ke G, Wu J, Wang X (2006) Modification of wool fiber using steam explosion. Eur Polym J 42:2168–2173
Yamauchi K, Yamauchi A, Kusunoki T, Kohda A, Konishi Y (1996) Preparation of stable aqueous solution of keratins, and physiochemical and biodegradational properties of films. J Biomed Mater Res 31:439–444
Yin X-C, Li F-Y, He Y-F, Wang Y, Wang R-M (2013) Study on effective extraction of chicken feather keratins and their films for controlling drug release. Biomater Sci 1:528–536
Zhang J et al (2013) Isolation and characterization of biofunctional keratin particles extracted from wool wastes. Powder Technol 246:356–362
Zhang Y, Zhao W, Yang R (2015) Steam flash explosion assisted dissolution of keratin from feathers. ACS Sustain Chem Eng 3:2036–2042
Zoccola M, Aluigi A, Tonin C (2009) Characterisation of keratin biomass from butchery and wool industry wastes. J Mol Struct 938:35–40
Acknowledgment
Authors are thankful to University Malaysia Pahang (UMP) for proving facilities and financial support; author [SS] is thankful to UMP for providing Doctoral Scholarship Scheme (DSS) scholarship as financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest among all authors.
Rights and permissions
About this article
Cite this article
Sharma, S., Gupta, A., Kumar, A. et al. An efficient conversion of waste feather keratin into ecofriendly bioplastic film. Clean Techn Environ Policy 20, 2157–2167 (2018). https://doi.org/10.1007/s10098-018-1498-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-018-1498-2