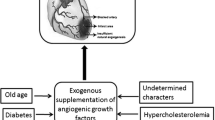
Abstract
Therapeutic angiogenesis is based on the premise that the development of new blood vessels can be augmented by exogenous administration of the appropriate growth factors. Over the last years, successful preclinical studies and promising results of early clinical trials have created great excitement about the potential of therapeutic angiogenesis for patients with advanced ischemic heart disease. The authors provide an overview of the biology of angiogenesis, the basic characteristics of angiogenic factors, and the different routes of their delivery. They discuss experimental studies in animal models of myocardial ischemia and outline available clinical studies on therapeutic angiogenesis for myocardial ischemia. Related safety issues are also addressed followed by a critical perspective about the future of proangiogenic therapies for ischemic cardiovascular disorders. Despite the established proof of concept and reasonable safety, however, results of the latest trials on therapeutic angiogenesis for myocardial ischemia have provided inconsistent results and the definite means of inducing clinically useful therapeutic angiogenesis remain elusive. More studies are required to gain further insights into the biology of angiogenesis and address pharmacological limitations of current approaches of angiogenic therapy. The authors hope and envisage that in the not-too-distant future, these investigative efforts will lead to important new strategies for treatment of myocardial ischemic syndromes. Means of non-invasive individualized pharmacological therapeutic neovascularization may be the next major advance in the treatment of ischaemic heart disease.





Similar content being viewed by others
Abbreviations
- EPC:
-
Endothelial precursor cell
- VEGF:
-
Vascular endothelial growth factor
- FGF:
-
Fibroblast growth factor
- VEGFR:
-
Vascular endothelial growth factor receptor
- NP/NRP:
-
Neuropilin
- FGFR:
-
Fibroblast growth factor receptor
- HLGAG:
-
Heparin-like glycosaminoglycans
- RTK:
-
Receptor tyrosine kinase
- MCP-1:
-
Monocyte chemotactic protein-1
- GM-CSF:
-
Granulocyte-macrophage colony-stimulating factor
- HGF:
-
Hepatocyte growth factor
- PDGF:
-
Platelet derived growth factor
- TGF-a/TGF-b:
-
Transforming growth factor-a/-b
- TNF-a:
-
Tumour necrosis factor-a
- BDNF:
-
Brain-derived neurotrophic factor
- CAD:
-
Coronary artery disease
- EMI:
-
Experimental myocardial iscehima
- MRI:
-
Magnetic resonance imaging
- G-CSF:
-
Granulocyte colony stimulating factor
- GM-CSF:
-
Granulocyte macrophage colony stimulating factor
- CABG:
-
Coronary artery bypass graft
- SPECT:
-
Single photon emission computed tomography
- LV:
-
Left ventricular
- LVEF:
-
Left ventricular ejection fraction
- CPC:
-
Cardiac progenitor cell
- BM:
-
Bone marrow
- BMC:
-
Bone marrow cell
- MSC:
-
Mesenchymal stem cell
- BM-MNC:
-
Bone marrow mononuclear cell
- AMI:
-
Acute myocardial infarction
- TMR:
-
Transmyocardial laser revascularization
- PMR:
-
Percutaneous myocardial revascularization
References
Murray CJ, Lopez AD (1997) Mortality by cause for eight regions of the world: global burden of disease study. Lancet 349(9061):1269–1276
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285(21):1182–1186
Gimbrone MA Jr, Cotran RS, Folkman J (1973) Endothelial regeneration: studies with human endothelial cells in culture. Ser Haematol 6(4):453–455
Shing Y, Folkman J, Sullivan R, Butterfield C, Murray J, Klagsbrun M (1984) Heparin affinity: purification of a tumor-derived capillary endothelial cell growth factor. Science 223(4642):1296–1299
Maciag T, Mehlman T, Friesel R, Schreiber AB (1984) Heparin binds endothelial cell growth factor, the principal endothelial cell mitogen in bovine brain. Science 225(4665):932–935
Bohlen P, Esch F, Baird A, Gospodarowicz D (1985) Acidic fibroblast growth factor (FGF) from bovine brain: amino-terminal sequence and comparison with basic FGF. EMBO J 4(8):1951–1956
Fett JW, Strydom DJ, Lobb RR et al (1985) Isolation and characterization of angiogenin, an angiogenic protein from human carcinoma cells. Biochemistry 24(20):5480–5486
Sasayama S, Fujita M (1992) Recent insights into coronary collateral circulation. Circulation 85(3):1197–1204
Charney R, Cohen M (1993) The role of the coronary collateral circulation in limiting myocardial ischemia and infarct size. Am Heart J 126(4):937–945
Schaper W, Ito WD (1996) Molecular mechanisms of coronary collateral vessel growth. Circ Res 79(5):911–919
Isner JM, Pieczek A, Schainfeld R et al (1996) Clinical evidence of angiogenesis after arterial gene transfer of phVEGF165 in patient with ischaemic limb. Lancet 348(9024):370–374
Baumgartner I, Pieczek A, Manor O et al (1998) Constitutive expression of phVEGF165 after intramuscular gene transfer promotes collateral vessel development in patients with critical limb ischemia. Circulation 97(12):1114–1123
Mukherjee D, Bhatt DL, Roe MT, Patel V, Ellis SG (1999) Direct myocardial revascularization and angiogenesis–how many patients might be eligible? Am J Cardiol 84(5):598–600, A598
Tabibiazar R, Rockson SG (2001) Angiogenesis and the ischaemic heart. Eur Heart J 22(11):903–918
Risau W, Flamme I (1995) Vasculogenesis. Annu Rev Cell Dev Biol 11:73–91
Risau W, Sariola H, Zerwes HG et al (1988) Vasculogenesis and angiogenesis in embryonic-stem-cell-derived embryoid bodies. Development 102(3):471–478
Luttun A, Carmeliet P (2003) De novo vasculogenesis in the heart. Cardiovasc Res 58(2):378–389
Risau W (1995) Differentiation of endothelium. FASEB J 9(10):926–933
Weiss MJ, Orkin SH (1996) In vitro differentiation of murine embryonic stem cells. New approaches to old problems. J Clin Invest 97(3):591–595
Asahara T, Murohara T, Sullivan A et al (1997) Isolation of putative progenitor endothelial cells for angiogenesis. Science 275(5302):964–967
Asahara T, Masuda H, Takahashi T et al (1999) Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res 85(3):221–228
Takahashi T, Kalka C, Masuda H et al (1999) Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat Med 5(4):434–438
Kalka C, Masuda H, Takahashi T et al (2000) Transplantation of ex vivo expanded endothelial progenitor cells for therapeutic neovascularization. Proc Natl Acad Sci USA 97(7):3422–3427
Risau W (1997) Mechanisms of angiogenesis. Nature 386(6626):671–674
Levenberg S (2005) Engineering blood vessels from stem cells: recent advances and applications. Curr Opin Biotechnol 16(5):516–523
Forsythe JA, Jiang BH, Iyer NV et al (1996) Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol 16(9):4604–4613
Levy AP, Levy NS, Wegner S, Goldberg MA (1995) Transcriptional regulation of the rat vascular endothelial growth factor gene by hypoxia. J Biol Chem 270(22):13333–13340
Hicklin DJ, Ellis LM (2005) Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 23(5):1011–1027
Bergers G, Benjamin LE (2003) Tumorigenesis and the angiogenic switch. Nat Rev Cancer 3(6):401–410
Fang J, Yan L, Shing Y, Moses MA (2001) HIF-1alpha-mediated up-regulation of vascular endothelial growth factor, independent of basic fibroblast growth factor, is important in the switch to the angiogenic phenotype during early tumorigenesis. Cancer Res 61(15):5731–5735
Jain RK (2005) Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science 307(5706):58–62
Ferrara N (2002) Role of vascular endothelial growth factor in physiologic and pathologic angiogenesis: therapeutic implications. Semin Oncol 29(6 Suppl 16):10–14
Connolly DT, Heuvelman DM, Nelson R et al (1989) Tumor vascular permeability factor stimulates endothelial cell growth and angiogenesis. J Clin Invest 84(5):1470–1478
Unemori EN, Ferrara N, Bauer EA, Amento EP (1992) Vascular endothelial growth factor induces interstitial collagenase expression in human endothelial cells. J Cell Physiol 153(3):557–562
Zachary I, Morgan RD (2010) Therapeutic angiogenesis for cardiovascular disease: biological context, challenges, prospects. Heart 97(3):181–189
Stimpfl M, Tong D, Fasching B et al (2002) Vascular endothelial growth factor splice variants and their prognostic value in breast and ovarian cancer. Clin Cancer Res 8(7):2253–2259
Carmeliet P, Collen D (1997) Molecular analysis of blood vessel formation and disease. Am J Physiol 273(5 Pt 2):H2091–H2104
Shibuya M, Ito N, Claesson-Welsh L (1999) Structure and function of vascular endothelial growth factor receptor-1 and -2. Curr Top Microbiol Immunol 237:59–83
Shalaby F, Rossant J, Yamaguchi TP et al (1995) Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 376(6535):62–66
Kawasaki T, Kitsukawa T, Bekku Y et al (1999) A requirement for neuropilin-1 in embryonic vessel formation. Development 126(21):4895–4902
Baka S, Clamp AR, Jayson GC (2006) A review of the latest clinical compounds to inhibit VEGF in pathological angiogenesis. Expert Opin Ther Targets 10(6):867–876
Guo D, Jia Q, Song HY, Warren RS, Donner DB (1995) Vascular endothelial cell growth factor promotes tyrosine phosphorylation of mediators of signal transduction that contain SH2 domains. Association with endothelial cell proliferation. J Biol Chem 270(12):6729–6733
Costa C, Soares R, Reis-Filho JS, Leitao D, Amendoeira I, Schmitt FC (2002) Cyclo-oxygenase 2 expression is associated with angiogenesis and lymph node metastasis in human breast cancer. J Clin Pathol 55(6):429–434
Waltenberger J, Mayr U, Pentz S, Hombach V (1996) Functional upregulation of the vascular endothelial growth factor receptor KDR by hypoxia. Circulation 94(7):1647–1654
Li J, Brown LF, Hibberd MG, Grossman JD, Morgan JP, Simons M (1996) VEGF, flk-1, and flt-1 expression in a rat myocardial infarction model of angiogenesis. Am J Physiol 270(5 Pt 2):H1803–H1811
Kalka C, Masuda H, Takahashi T et al (2000) Vascular endothelial growth factor(165) gene transfer augments circulating endothelial progenitor cells in human subjects. Circ Res 86(12):1198–1202
Asahara T, Takahashi T, Masuda H et al (1999) VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO J 18(14):3964–3972
Venkataraman G, Raman R, Sasisekharan V, Sasisekharan R (1999) Molecular characteristics of fibroblast growth factor-fibroblast growth factor receptor-heparin-like glycosaminoglycan complex. Proc Natl Acad Sci USA 96(7):3658–3663
Carmeliet P (2000) Mechanisms of angiogenesis and arteriogenesis. Nat Med 6(4):389–395
Itoh N, Ornitz DM (2011) Fibroblast growth factors: from molecular evolution to roles in development, metabolism and disease. J Biochem 149(2):121–130
Ornitz DM, Itoh N (2001) Fibroblast growth factors. Genome Biol 2(3):3005 (reviews)
Lee PL, Johnson DE, Cousens LS, Fried VA, Williams LT (1989) Purification and complementary DNA cloning of a receptor for basic fibroblast growth factor. Science 245(4913):57–60
Mignatti P, Morimoto T, Rifkin DB (1992) Basic fibroblast growth factor, a protein devoid of secretory signal sequence, is released by cells via a pathway independent of the endoplasmic reticulum-Golgi complex. J Cell Physiol 151(1):81–93
Miki T, Bottaro DP, Fleming TP et al (1992) Determination of ligand-binding specificity by alternative splicing: two distinct growth factor receptors encoded by a single gene. Proc Natl Acad Sci USA 89(1):246–250
Gringel S, van Bergeijk J, Haastert K, Grothe C, Claus P (2004) Nuclear fibroblast growth factor-2 interacts specifically with splicing factor SF3a66. Biol Chem 385(12):1203–1208
Tabata H, Silver M, Isner JM (1997) Arterial gene transfer of acidic fibroblast growth factor for therapeutic angiogenesis in vivo: critical role of secretion signal in use of naked DNA. Cardiovasc Res 35(3):470–479
Giordano FJ, Ping P, McKirnan MD et al (1996) Intracoronary gene transfer of fibroblast growth factor-5 increases blood flow and contractile function in an ischemic region of the heart. Nat Med 2(5):534–539
Ito WD, Arras M, Winkler B, Scholz D, Schaper J, Schaper W (1997) Monocyte chemotactic protein-1 increases collateral and peripheral conductance after femoral artery occlusion. Circ Res 80(6):829–837
Suri C, Jones PF, Patan S et al (1996) Requisite role of angiopoietin-1, a ligand for the TIE2 receptor, during embryonic angiogenesis. Cell 87(7):1171–1180
Yamada N, Li W, Ihaya A et al (2006) Platelet-derived endothelial cell growth factor gene therapy for limb ischemia. J Vasc Surg 44(6):1322–1328
Buschmann IR, Hoefer IE, van Royen N et al (2001) GM-CSF: a strong arteriogenic factor acting by amplification of monocyte function. Atherosclerosis 159(2):343–356
Grundmann S, Piek JJ, Pasterkamp G, Hoefer IE (2007) Arteriogenesis: basic mechanisms and therapeutic stimulation. Eur J Clin Invest 37(10):755–766
Heil M, Eitenmuller I, Schmitz-Rixen T, Schaper W (2006) Arteriogenesis versus angiogenesis: similarities and differences. J Cell Mol Med 10(1):45–55
Heil M, Schaper W (2005) Cellular mechanisms of arteriogenesis. EXS (94):181–191
Van Royen N, Piek JJ, Schaper W, Bode C, Buschmann I (2001) Arteriogenesis: mechanisms and modulation of collateral artery development. J Nucl Cardiol 8(6):687–693
Schirmer SH, van Nooijen FC, Piek JJ, van Royen N (2009) Stimulation of collateral artery growth: travelling further down the road to clinical application. Heart 95(3):191–197
Schirmer SH, van Royen N (2004) Stimulation of collateral artery growth: a potential treatment for peripheral artery disease. Expert Rev Cardiovasc Ther 2(4):581–588
Meier P, Gloekler S, Zbinden R et al (2007) Beneficial effect of recruitable collaterals: a 10-year follow-up study in patients with stable coronary artery disease undergoing quantitative collateral measurements. Circulation 116(9):975–983
Fukumura D, Xu L, Chen Y, Gohongi T, Seed B, Jain RK (2001) Hypoxia and acidosis independently up-regulate vascular endothelial growth factor transcription in brain tumors in vivo. Cancer Res 61(16):6020–6024
Semenza G (2002) Signal transduction to hypoxia-inducible factor 1. Biochem Pharmacol 64(5–6):993–998
Gray C, Packham IM, Wurmser F et al (2007) Ischemia is not required for arteriogenesis in zebrafish embryos. Arterioscler Thromb Vasc Biol 27(10):2135–2141
Shyy JY, Chien S (2002) Role of integrins in endothelial mechanosensing of shear stress. Circ Res 91(9):769–775
van Royen N, Piek JJ, Buschmann I, Hoefer I, Voskuil M, Schaper W (2001) Stimulation of arteriogenesis; a new concept for the treatment of arterial occlusive disease. Cardiovasc Res 49(3):543–553
Lopez-Quintero SV, Amaya R, Pahakis M, Tarbell JM (2009) The endothelial glycocalyx mediates shear-induced changes in hydraulic conductivity. Am J Physiol Heart Circ Physiol 296(5):H1451–H1456
Thi MM, Tarbell JM, Weinbaum S, Spray DC (2004) The role of the glycocalyx in reorganization of the actin cytoskeleton under fluid shear stress: a “bumper-car” model. Proc Natl Acad Sci USA 101(47):16483–16488
Reitsma S, Slaaf DW, Vink H, van Zandvoort MA, Oude Egbrink MG (2007) The endothelial glycocalyx: composition, functions, and visualization. Pflugers Arch 454(3):345–359
Eitenmuller I, Volger O, Kluge A et al (2006) The range of adaptation by collateral vessels after femoral artery occlusion. Circ Res 99(6):656–662
Carr AN, Howard BW, Yang HT et al (2006) Efficacy of systemic administration of SDF-1 in a model of vascular insufficiency: support for an endothelium-dependent mechanism. Cardiovasc Res 69(4):925–935
Mitsos S, Katsanos K, Dougeni E, Koletsis EN, Dougenis D (2009) A critical appraisal of open- and closed-chest models of experimental myocardial ischemia. Lab Anim (NY) 38(5):167–177
Banai S, Shweiki D, Pinson A, Chandra M, Lazarovici G, Keshet E (1994) Upregulation of vascular endothelial growth factor expression induced by myocardial ischaemia: implications for coronary angiogenesis. Cardiovasc Res 28(8):1176–1179
Hashimoto E, Ogita T, Nakaoka T, Matsuoka R, Takao A, Kira Y (1994) Rapid induction of vascular endothelial growth factor expression by transient ischemia in rat heart. Am J Physiol 267(5 Pt 2):H1948–H1954
Sato K, Wu T, Laham RJ et al (2001) Efficacy of intracoronary or intravenous VEGF165 in a pig model of chronic myocardial ischemia. J Am Coll Cardiol 37(2):616–623
Hariawala MD, Horowitz JR, Esakof D et al (1996) VEGF improves myocardial blood flow but produces EDRF-mediated hypotension in porcine hearts. J Surg Res 63(1):77–82
Lopez JJ, Laham RJ, Stamler A et al (1998) VEGF administration in chronic myocardial ischemia in pigs. Cardiovasc Res 40(2):272–281
Harada K, Friedman M, Lopez JJ et al (1996) Vascular endothelial growth factor administration in chronic myocardial ischemia. Am J Physiol 270(5 Pt 2):H1791–H1802
Banai S, Jaklitsch MT, Shou M et al (1994) Angiogenic-induced enhancement of collateral blood flow to ischemic myocardium by vascular endothelial growth factor in dogs. Circulation 89(5):2183–2189
Besse S, Boucher F, Linguet G et al (2010) Intramyocardial protein therapy with vascular endothelial growth factor (VEGF-165) induces functional angiogenesis in rat senescent myocardium. J Physiol Pharmacol 61(6):651–661
Tio RA, Tkebuchava T, Scheuermann TH et al (1999) Intramyocardial gene therapy with naked DNA encoding vascular endothelial growth factor improves collateral flow to ischemic myocardium. Hum Gene Ther 10(18):2953–2960
Lee LY, Patel SR, Hackett NR et al (2000) Focal angiogen therapy using intramyocardial delivery of an adenovirus vector coding for vascular endothelial growth factor 121. Ann Thorac Surg 69(1):14–23 (discussion 23–14)
Zhang D, Gai L, Fan R, Dong W, Wen Y (2002) Efficacy and safety of therapeutic angiogenesis from direct myocardial administration of an adenoviral vector expressing vascular endothelial growth factor 165. Chin Med J (Engl) 115(5):643–648
Mack CA, Patel SR, Schwarz EA et al (1998) Biologic bypass with the use of adenovirus-mediated gene transfer of the complementary deoxyribonucleic acid for vascular endothelial growth factor 121 improves myocardial perfusion and function in the ischemic porcine heart. J Thorac Cardiovasc Surg 115(1):168–176 (discussion 176–167)
Lazarous DF, Shou M, Stiber JA et al (1999) Adenoviral-mediated gene transfer induces sustained pericardial VEGF expression in dogs: effect on myocardial angiogenesis. Cardiovasc Res 44(2):294–302
Vale PR, Losordo DW, Tkebuchava T, Chen D, Milliken CE, Isner JM (1999) Catheter-based myocardial gene transfer utilizing nonfluoroscopic electromechanical left ventricular mapping. J Am Coll Cardiol 34(1):246–254
Unger EF, Banai S, Shou M et al (1994) Basic fibroblast growth factor enhances myocardial collateral flow in a canine model. Am J Physiol 266(4 Pt 2):H1588–H1595
Lazarous DF, Shou M, Scheinowitz M et al (1996) Comparative effects of basic fibroblast growth factor and vascular endothelial growth factor on coronary collateral development and the arterial response to injury. Circulation 94(5):1074–1082
Lazarous DF, Scheinowitz M, Shou M et al (1995) Effects of chronic systemic administration of basic fibroblast growth factor on collateral development in the canine heart. Circulation 91(1):145–153
Shou M, Thirumurti V, Rajanayagam S et al (1997) Effect of basic fibroblast growth factor on myocardial angiogenesis in dogs with mature collateral vessels. J Am Coll Cardiol 29(5):1102–1106
Sato K, Laham RJ, Pearlman JD et al (2000) Efficacy of intracoronary versus intravenous FGF-2 in a pig model of chronic myocardial ischemia. Ann Thorac Surg 70(6):2113–2118
Landau C, Jacobs AK, Haudenschild CC (1995) Intrapericardial basic fibroblast growth factor induces myocardial angiogenesis in a rabbit model of chronic ischemia. Am Heart J 129(5):924–931
Lopez JJ, Edelman ER, Stamler A et al (1998) Angiogenic potential of perivascularly delivered aFGF in a porcine model of chronic myocardial ischemia. Am J Physiol 274(3 Pt 2):H930–H936
Harada K, Grossman W, Friedman M et al (1994) Basic fibroblast growth factor improves myocardial function in chronically ischemic porcine hearts. J Clin Invest 94(2):623–630
Lewis BS, Flugelman MY, Weisz A, Keren-Tal I, Schaper W (1997) Angiogenesis by gene therapy: a new horizon for myocardial revascularization? Cardiovasc Res 35(3):490–497
Bauters C, Asahara T, Zheng LP et al (1995) Site-specific therapeutic angiogenesis after systemic administration of vascular endothelial growth factor. J Vasc Surg 21(2):314–324 (discussion 324–315)
Takeshita S, Zheng LP, Brogi E et al (1994) Therapeutic angiogenesis. A single intraarterial bolus of vascular endothelial growth factor augments revascularization in a rabbit ischemic hind limb model. J Clin Invest 93(2):662–670
Sellke FW, Laham RJ, Edelman ER, Pearlman JD, Simons M (1998) Therapeutic angiogenesis with basic fibroblast growth factor: technique and early results. Ann Thorac Surg 65(6):1540–1544
Schwarz ER, Speakman MT, Patterson M et al (2000) Evaluation of the effects of intramyocardial injection of DNA expressing vascular endothelial growth factor (VEGF) in a myocardial infarction model in the rat-angiogenesis and angioma formation. J Am Coll Cardiol 35(5):1323–1330
Cuevas P, Carceller F, Ortega S, Zazo M, Nieto I, Gimenez-Gallego G (1991) Hypotensive activity of fibroblast growth factor. Science 254(5035):1208–1210
Baumgartner I, Rauh G, Pieczek A et al (2000) Lower-extremity edema associated with gene transfer of naked DNA encoding vascular endothelial growth factor. Ann Intern Med 132(11):880–884
Mazue G, Bertolero F, Jacob C, Sarmientos P, Roncucci R (1991) Preclinical and clinical studies with recombinant human basic fibroblast growth factor. Ann NY Acad Sci 638:329–340
Takeshita S, Weir L, Chen D et al (1996) Therapeutic angiogenesis following arterial gene transfer of vascular endothelial growth factor in a rabbit model of hindlimb ischemia. Biochem Biophys Res Commun 227(2):628–635
Gal D, Weir L, Leclerc G, Pickering JG, Hogan J, Isner JM (1993) Direct myocardial transfection in two animal models. Evaluation of parameters affecting gene expression and percutaneous gene delivery. Lab Invest 68(1):18–25
Wells DJ (1993) Improved gene transfer by direct plasmid injection associated with regeneration in mouse skeletal muscle. FEBS Lett 332(1–2):179–182
Wolff JA, Malone RW, Williams P et al (1990) Direct gene transfer into mouse muscle in vivo. Science 247(4949 Pt 1):1465–1468
Tsurumi Y, Takeshita S, Chen D et al (1996) Direct intramuscular gene transfer of naked DNA encoding vascular endothelial growth factor augments collateral development and tissue perfusion. Circulation 94(12):3281–3290
Takeshita S, Isshiki T, Sato T (1996) Increased expression of direct gene transfer into skeletal muscles observed after acute ischemic injury in rats. Lab Invest 74(6):1061–1065
Wolff JA, Ludtke JJ, Acsadi G, Williams P, Jani A (1992) Long-term persistence of plasmid DNA and foreign gene expression in mouse muscle. Hum Mol Genet 1(6):363–369
Takeshita S, Losordo DW, Kearney M, Rossow ST, Isner JM (1994) Time course of recombinant protein secretion after liposome-mediated gene transfer in a rabbit arterial organ culture model. Lab Invest 71(3):387–391
Losordo DW, Pickering JG, Takeshita S et al (1994) Use of the rabbit ear artery to serially assess foreign protein secretion after site-specific arterial gene transfer in vivo. Evidence that anatomic identification of successful gene transfer may underestimate the potential magnitude of transgene expression. Circulation 89(2):785–792
Harats D, Kurihara H, Belloni P et al (1995) Targeting gene expression to the vascular wall in transgenic mice using the murine preproendothelin-1 promoter. J Clin Invest 95(3):1335–1344
Kasahara N, Dozy AM, Kan YW (1994) Tissue-specific targeting of retroviral vectors through ligand-receptor interactions. Science 266(5189):1373–1376
Kornowski R, Fuchs S, Leon MB, Epstein SE (2000) Delivery strategies to achieve therapeutic myocardial angiogenesis. Circulation 101(4):454–458
Webster KA (2005) Therapeutic angiogenesis for coronary artery disease: clinical trials of proteins, plasmids, adenovirus and stem cells. Future Cardiol 1(1):99–109
Freedman SB, Isner JM (2002) Therapeutic angiogenesis for coronary artery disease. Ann Intern Med 136(1):54–71
Henry TD, Annex BH, McKendall GR et al (2003) The VIVA trial: vascular endothelial growth factor in ischemia for vascular angiogenesis. Circulation 107(10):1359–1365
Henry TD, Rocha-Singh K, Isner JM et al (2001) Intracoronary administration of recombinant human vascular endothelial growth factor to patients with coronary artery disease. Am Heart J 142(5):872–880
Udelson JE, Dilsizian V, Laham RJ et al (2000) Therapeutic angiogenesis with recombinant fibroblast growth factor-2 improves stress and rest myocardial perfusion abnormalities in patients with severe symptomatic chronic coronary artery disease. Circulation 102(14):1605–1610
Nabel EG, Yang ZY, Plautz G et al (1993) Recombinant fibroblast growth factor-1 promotes intimal hyperplasia and angiogenesis in arteries in vivo. Nature 362(6423):844–846
Inoue M, Itoh H, Ueda M et al (1998) Vascular endothelial growth factor (VEGF) expression in human coronary atherosclerotic lesions: possible pathophysiological significance of VEGF in progression of atherosclerosis. Circulation 98(20):2108–2116
Svensson EC, Marshall DJ, Woodard K et al (1999) Efficient and stable transduction of cardiomyocytes after intramyocardial injection or intracoronary perfusion with recombinant adeno-associated virus vectors. Circulation 99(2):201–205
Schumacher B, Pecher P, von Specht BU, Stegmann T (1998) Induction of neoangiogenesis in ischemic myocardium by human growth factors: first clinical results of a new treatment of coronary heart disease. Circulation 97(7):645–650
Rosengart TK, Lee LY, Patel SR et al (1999) Angiogenesis gene therapy: phase I assessment of direct intramyocardial administration of an adenovirus vector expressing VEGF121 cDNA to individuals with clinically significant severe coronary artery disease. Circulation 100(5):468–474
Varenne O, Sinnaeve P, Gillijns H et al (2000) Percutaneous gene therapy using recombinant adenoviruses encoding human herpes simplex virus thymidine kinase, human PAI-1, and human NOS3 in balloon-injured porcine coronary arteries. Hum Gene Ther 11(9):1329–1339
March KL, Woody M, Mehdi K, Zipes DP, Brantly M, Trapnell BC (1999) Efficient in vivo catheter-based pericardial gene transfer mediated by adenoviral vectors. Clin Cardiol 22(1 Suppl 1):I23–I29
Uchida Y, Yanagisawa-Miwa A, Nakamura F et al (1995) Angiogenic therapy of acute myocardial infarction by intrapericardial injection of basic fibroblast growth factor and heparin sulfate: an experimental study. Am Heart J 130(6):1182–1188
Laham RJ, Hung D, Simons M (1999) Therapeutic myocardial angiogenesis using percutaneous intrapericardial drug delivery. Clin Cardiol 22(1 Suppl 1):I6–I9
Hendel RC, Henry TD, Rocha-Singh K et al (2000) Effect of intracoronary recombinant human vascular endothelial growth factor on myocardial perfusion: evidence for a dose-dependent effect. Circulation 101(2):118–121
Stegmann TJ, Hoppert T, Schneider A et al (2000) Induction of myocardial neoangiogenesis by human growth factors. A new therapeutic approach in coronary heart disease. Herz 25(6):589–599
Schumacher B, Stegmann T, Pecher P (1998) The stimulation of neoangiogenesis in the ischemic human heart by the growth factor FGF: first clinical results. J Cardiovasc Surg (Torino) 39(6):783–789
Unger EF, Goncalves L, Epstein SE et al (2000) Effects of a single intracoronary injection of basic fibroblast growth factor in stable angina pectoris. Am J Cardiol 85(12):1414–1419
Laham RJ, Chronos NA, Pike M et al (2000) Intracoronary basic fibroblast growth factor (FGF-2) in patients with severe ischemic heart disease: results of a phase I open-label dose escalation study. J Am Coll Cardiol 36(7):2132–2139
Laham RJ, Sellke FW, Edelman ER et al (1999) Local perivascular delivery of basic fibroblast growth factor in patients undergoing coronary bypass surgery: results of a phase I randomized, double-blind, placebo-controlled trial. Circulation 100(18):1865–1871
Simons M, Annex BH, Laham RJ et al (2002) Pharmacological treatment of coronary artery disease with recombinant fibroblast growth factor-2: double-blind, randomized, controlled clinical trial. Circulation 105(7):788–793
Kuethe F, Figulla HR, Voth M et al (2004) Mobilization of stem cells by granulocyte colony-stimulating factor for the regeneration of myocardial tissue after myocardial infarction. Dtsch Med Wochenschr 129(9):424–428
Kang HJ, Kim HS, Koo BK et al (2007) Intracoronary infusion of the mobilized peripheral blood stem cell by G-CSF is better than mobilization alone by G-CSF for improvement of cardiac function and remodeling: 2-year follow-up results of the myocardial regeneration and angiogenesis in myocardial infarction with G-CSF and intra-coronary stem cell infusion (MAGIC Cell) 1 trial. Am Heart J 153(2):237.e1–237.e8
Kang HJ, Kim HS, Zhang SY et al (2004) Effects of intracoronary infusion of peripheral blood stem-cells mobilised with granulocyte-colony stimulating factor on left ventricular systolic function and restenosis after coronary stenting in myocardial infarction: the MAGIC cell randomised clinical trial. Lancet 363(9411):751–756
Valgimigli M, Rigolin GM, Cittanti C et al (2005) Use of granulocyte-colony stimulating factor during acute myocardial infarction to enhance bone marrow stem cell mobilization in humans: clinical and angiographic safety profile. Eur Heart J 26(18):1838–1845
Zohlnhofer D, Ott I, Mehilli J et al (2006) Stem cell mobilization by granulocyte colony-stimulating factor in patients with acute myocardial infarction: a randomized controlled trial. JAMA 295(9):1003–1010
Ripa RS, Jorgensen E, Wang Y et al (2006) Stem cell mobilization induced by subcutaneous granulocyte-colony stimulating factor to improve cardiac regeneration after acute ST-elevation myocardial infarction: result of the double-blind, randomized, placebo-controlled stem cells in myocardial infarction (STEMMI) trial. Circulation 113(16):1983–1992
Losordo DW, Vale PR, Symes JF et al (1998) Gene therapy for myocardial angiogenesis: initial clinical results with direct myocardial injection of phVEGF165 as sole therapy for myocardial ischemia. Circulation 98(25):2800–2804
Symes JF, Losordo DW, Vale PR et al (1999) Gene therapy with vascular endothelial growth factor for inoperable coronary artery disease. Ann Thorac Surg 68(3):830–836 (discussion 836–837)
Vale PR, Losordo DW, Milliken CE et al (2000) Left ventricular electromechanical mapping to assess efficacy of phVEGF(165) gene transfer for therapeutic angiogenesis in chronic myocardial ischemia. Circulation 102(9):965–974
Kastrup J, Jorgensen E, Ruck A et al (2005) Direct intramyocardial plasmid vascular endothelial growth factor-A165 gene therapy in patients with stable severe angina pectoris A randomized double-blind placebo-controlled study: the EUROINJECT ONE trial. J Am Coll Cardiol 45(7):982–988
Gyongyosi M, Khorsand A, Zamini S et al (2005) NOGA-guided analysis of regional myocardial perfusion abnormalities treated with intramyocardial injections of plasmid encoding vascular endothelial growth factor A-165 in patients with chronic myocardial ischemia: subanalysis of the EUROINJECT-ONE multicenter double-blind randomized study. Circulation 112(9 Suppl):I157–I165
Sarkar N, Ruck A, Kallner G et al (2001) Effects of intramyocardial injection of phVEGF-A165 as sole therapy in patients with refractory coronary artery disease—12-month follow-up: angiogenic gene therapy. J Intern Med 250(5):373–381
Stewart DJ, Hilton JD, Arnold JM et al (2006) Angiogenic gene therapy in patients with nonrevascularizable ischemic heart disease: a phase 2 randomized, controlled trial of AdVEGF(121) (AdVEGF121) versus maximum medical treatment. Gene Ther 13(21):1503–1511
Kastrup J, Jorgensen E, Fuchs S et al (2011) A randomised, double-blind, placebo-controlled, multicentre study of the safety and efficacy of BIOBYPASS (AdGVVEGF121.10NH) gene therapy in patients with refractory advanced coronary artery disease: the NOVA trial. EuroIntervention 6(7):813–818
Vale PR, Losordo DW, Milliken CE et al (2001) Randomized, single-blind, placebo-controlled pilot study of catheter-based myocardial gene transfer for therapeutic angiogenesis using left ventricular electromechanical mapping in patients with chronic myocardial ischemia. Circulation 103(17):2138–2143
Losordo DW, Vale PR, Hendel RC et al (2002) Phase 1/2 placebo-controlled, double-blind, dose-escalating trial of myocardial vascular endothelial growth factor 2 gene transfer by catheter delivery in patients with chronic myocardial ischemia. Circulation 105(17):2012–2018
Grines CL, Watkins MW, Helmer G et al (2002) Angiogenic gene therapy (AGENT) trial in patients with stable angina pectoris. Circulation 105(11):1291–1297
Grines CL, Watkins MW, Mahmarian JJ et al (2003) A randomized, double-blind, placebo-controlled trial of Ad5FGF-4 gene therapy and its effect on myocardial perfusion in patients with stable angina. J Am Coll Cardiol 42(8):1339–1347
Beohar N, Rapp J, Pandya S, Losordo DW (2010) Rebuilding the damaged heart: the potential of cytokines and growth factors in the treatment of ischemic heart disease. J Am Coll Cardiol 56(16):1287–1297
Lionetti V, Bianchi G, Recchia FA, Ventura C (2010) Control of autocrine and paracrine myocardial signals: an emerging therapeutic strategy in heart failure. Heart Fail Rev 15(6):531–542
Menasche P (2010) Cell therapy for peripheral arterial disease. Curr Opin Mol Ther 12(5):538–545
Kawamoto A, Gwon HC, Iwaguro H et al (2001) Therapeutic potential of ex vivo expanded endothelial progenitor cells for myocardial ischemia. Circulation 103(5):634–637
Kobayashi T, Hamano K, Li TS et al (2000) Enhancement of angiogenesis by the implantation of self bone marrow cells in a rat ischemic heart model. J Surg Res 89(2):189–195
Tomita S, Li RK, Weisel RD et al (1999) Autologous transplantation of bone marrow cells improves damaged heart function. Circulation 100(19 Suppl):II247–II256
Botta R, Gao E, Stassi G et al (2004) Heart infarct in NOD-SCID mice: therapeutic vasculogenesis by transplantation of human CD34+ cells and low dose CD34+ KDR+ cells. FASEB J 18(12):1392–1394
Kocher AA, Schuster MD, Szabolcs MJ et al (2001) Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med 7(4):430–436
Bhakta S, Greco NJ, Finney MR et al (2006) The safety of autologous intracoronary stem cell injections in a porcine model of chronic myocardial ischemia. J Invasive Cardiol 18(5):212–218
Li CJ, Gao RL, Yang YJ et al (2008) Implantation of autologous bone marrow mononuclear cells into ischemic myocardium enhances coronary capillaries and systolic function in miniswine. Chin Med Sci J 23(4):234–238
Kamihata H, Matsubara H, Nishiue T et al (2001) Implantation of bone marrow mononuclear cells into ischemic myocardium enhances collateral perfusion and regional function via side supply of angioblasts, angiogenic ligands, and cytokines. Circulation 104(9):1046–1052
Murohara T, Ikeda H, Duan J et al (2000) Transplanted cord blood-derived endothelial precursor cells augment postnatal neovascularization. J Clin Invest 105(11):1527–1536
Nagaya N, Fujii T, Iwase T et al (2004) Intravenous administration of mesenchymal stem cells improves cardiac function in rats with acute myocardial infarction through angiogenesis and myogenesis. Am J Physiol Heart Circ Physiol 287(6):H2670–H2676
Olivares EL, Ribeiro VP, Werneck de Castro JP et al (2004) Bone marrow stromal cells improve cardiac performance in healed infarcted rat hearts. Am J Physiol Heart Circ Physiol 287(2):H464–H470
Strauer BE, Brehm M, Zeus T et al (2002) Repair of infarcted myocardium by autologous intracoronary mononuclear bone marrow cell transplantation in humans. Circulation 106(15):1913–1918
Wollert KC, Meyer GP, Lotz J et al (2004) Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet 364(9429):141–148
Tse HF, Kwong YL, Chan JK, Lo G, Ho CL, Lau CP (2003) Angiogenesis in ischaemic myocardium by intramyocardial autologous bone marrow mononuclear cell implantation. Lancet 361(9351):47–49
Fuchs S, Baffour R, Zhou YF et al (2001) Transendocardial delivery of autologous bone marrow enhances collateral perfusion and regional function in pigs with chronic experimental myocardial ischemia. J Am Coll Cardiol 37(6):1726–1732
Fernandez-Aviles F, San Roman JA, Garcia-Frade J et al (2004) Experimental and clinical regenerative capability of human bone marrow cells after myocardial infarction. Circ Res 95(7):742–748
Assmus B, Schachinger V, Teupe C et al (2002) Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction (TOPCARE-AMI). Circulation 106(24):3009–3017
Meyer GP, Wollert KC, Lotz J et al (2006) Intracoronary bone marrow cell transfer after myocardial infarction: eighteen months’ follow-up data from the randomized, controlled BOOST (BOne marrOw transfer to enhance ST-elevation infarct regeneration) trial. Circulation 113(10):1287–1294
Lunde K, Solheim S, Aakhus S et al (2006) Intracoronary injection of mononuclear bone marrow cells in acute myocardial infarction. N Engl J Med 355(12):1199–1209
Assmus B, Honold J, Schachinger V et al (2006) Transcoronary transplantation of progenitor cells after myocardial infarction. N Engl J Med 355(12):1222–1232
Schachinger V, Erbs S, Elsasser A et al (2006) Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N Engl J Med 355(12):1210–1221
Bartunek J, Vanderheyden M, Vandekerckhove B et al (2005) Intracoronary injection of CD133-positive enriched bone marrow progenitor cells promotes cardiac recovery after recent myocardial infarction: feasibility and safety. Circulation 112(9 Suppl):I178–I183
Stamm C, Westphal B, Kleine HD et al (2003) Autologous bone-marrow stem-cell transplantation for myocardial regeneration. Lancet 361(9351):45–46
Archundia A, Aceves JL, Lopez-Hernandez M et al (2005) Direct cardiac injection of G-CSF mobilized bone-marrow stem-cells improves ventricular function in old myocardial infarction. Life Sci 78(3):279–283
Losordo DW, Schatz RA, White CJ et al (2007) Intramyocardial transplantation of autologous CD34+ stem cells for intractable angina: a phase I/IIa double-blind, randomized controlled trial. Circulation 115(25):3165–3172
Steinwender C, Hofmann R, Kammler J et al (2006) Effects of peripheral blood stem cell mobilization with granulocyte-colony stimulating factor and their transcoronary transplantation after primary stent implantation for acute myocardial infarction. Am Heart J 151(6):1296.e7–1296.e13
Chen SL, Fang WW, Ye F et al (2004) Effect on left ventricular function of intracoronary transplantation of autologous bone marrow mesenchymal stem cell in patients with acute myocardial infarction. Am J Cardiol 94(1):92–95
Katritsis DG, Sotiropoulou PA, Karvouni E et al (2005) Transcoronary transplantation of autologous mesenchymal stem cells and endothelial progenitors into infarcted human myocardium. Catheter Cardiovasc Interv 65(3):321–329
Shintani S, Kusano K, Ii M et al (2006) Synergistic effect of combined intramyocardial CD34+ cells and VEGF2 gene therapy after MI. Nat Clin Pract Cardiovasc Med 3(Suppl 1):S123–S128
Azarnoush K, Maurel A, Sebbah L et al (2005) Enhancement of the functional benefits of skeletal myoblast transplantation by means of coadministration of hypoxia-inducible factor 1alpha. J Thorac Cardiovasc Surg 130(1):173–179
Wollert KC, Drexler H (2010) Cell therapy for the treatment of coronary heart disease: a critical appraisal. Nat Rev Cardiol 7(4):204–215
Mirhoseini M, Muckerheide M, Cayton MM (1982) Transventricular revascularization by laser. Lasers Surg Med 2(2):187–198
Huikeshoven M, Beek JF, van der Sloot JA, Tukkie R, van der Meulen J, van Gemert MJ (2002) 35 years of experimental research in transmyocardial revascularization: what have we learned? Ann Thorac Surg 74(3):956–970
McNab DC, Schofield PM (2002) Transmyocardial and percutaneous myocardial laser revascularization. Circulation 105(19):e171–e172 (author reply e171–e172)
Burns SM, Sharples LD, Tait S, Caine N, Wallwork J, Schofield PM (1999) The transmyocardial laser revascularization international registry report. Eur Heart J 20(1):31–37
Aaberge L, Nordstrand K, Dragsund M et al (2000) Transmyocardial revascularization with CO2 laser in patients with refractory angina pectoris. Clinical results from the Norwegian randomized trial. J Am Coll Cardiol 35(5):1170–1177
Frazier OH, March RJ, Horvath KA (1999) Transmyocardial revascularization with a carbon dioxide laser in patients with end-stage coronary artery disease. N Engl J Med 341(14):1021–1028
Campbell HE, Tait S, Buxton MJ et al (2001) A UK trial-based cost-utility analysis of transmyocardial laser revascularization compared to continued medical therapy for treatment of refractory angina pectoris. Eur J Cardiothorac Surg 20(2):312–318
Rimoldi O, Burns SM, Rosen SD et al (1999) Measurement of myocardial blood flow with positron emission tomography before and after transmyocardial laser revascularization. Circulation 100(19 Suppl):II134–II138
Al-Sheikh T, Allen KB, Straka SP et al (1999) Cardiac sympathetic denervation after transmyocardial laser revascularization. Circulation 100(2):135–140
Kwong KF, Schuessler RB, Kanellopoulos GK, Saffitz JE, Sundt TM III (1998) Nontransmural laser treatment incompletely denervates canine myocardium. Circulation 98(19 Suppl):II67–II71 (discussion II71–II62)
Hughes GC, Landolfo KP, Lowe JE, Coleman RB, Donovan CL (1999) Diagnosis, incidence, and clinical significance of early postoperative ischemia after transmyocardial laser revascularization. Am Heart J 137(6):1163–1168
Bridges CR (2006) Guidelines for the clinical use of transmyocardial laser revascularization. Semin Thorac Cardiovasc Surg 18(1):68–73
Oesterle SN, Sanborn TA, Ali N et al (2000) Percutaneous transmyocardial laser revascularisation for severe angina: the PACIFIC randomised trial. Potential class improvement from intramyocardial channels. Lancet 356(9243):1705–1710
Lauer B, Junghans U, Stahl F, Kluge R, Oesterle SN, Schuler G (1999) Catheter-based percutaneous myocardial laser revascularization in patients with end-stage coronary artery disease. J Am Coll Cardiol 34(6):1663–1670
Stone GW, Teirstein PS, Rubenstein R et al (2002) A prospective, multicenter, randomized trial of percutaneous transmyocardial laser revascularization in patients with nonrecanalizable chronic total occlusions. J Am Coll Cardiol 39(10):1581–1587
Horowitz JR, Rivard A, van der Zee R et al (1997) Vascular endothelial growth factor/vascular permeability factor produces nitric oxide-dependent hypotension. Evidence for a maintenance role in quiescent adult endothelium. Arterioscler Thromb Vasc Biol 17(11):2793–2800
Lopez JJ, Laham RJ, Carrozza JP et al (1997) Hemodynamic effects of intracoronary VEGF delivery: evidence of tachyphylaxis and NO dependence of response. Am J Physiol 273(3 Pt 2):H1317–H1323
Rajagopalan S, Mohler ER III, Lederman RJ et al (2003) Regional angiogenesis with vascular endothelial growth factor in peripheral arterial disease: a phase II randomized, double-blind, controlled study of adenoviral delivery of vascular endothelial growth factor 121 in patients with disabling intermittent claudication. Circulation 108(16):1933–1938
Springer ML, Chen AS, Kraft PE, Bednarski M, Blau HM (1998) VEGF gene delivery to muscle: potential role for vasculogenesis in adults. Mol Cell 2(5):549–558
Lee RJ, Springer ML, Blanco-Bose WE, Shaw R, Ursell PC, Blau HM (2000) VEGF gene delivery to myocardium: deleterious effects of unregulated expression. Circulation 102(8):898–901
Kloner RA, Dow J, Chung G, Kedes LH (2000) Intramyocardial injection of DNA encoding vascular endothelial growth factor in a myocardial infarction model. J Thromb Thrombolysis 10(3):285–289
Couffinhal T, Silver M, Kearney M et al (1999) Impaired collateral vessel development associated with reduced expression of vascular endothelial growth factor in ApoE−/− mice. Circulation 99(24):3188–3198
Folkman J (1995) Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1(1):27–31
Ferrara N, Davis-Smyth T (1997) The biology of vascular endothelial growth factor. Endocr Rev 18(1):4–25
Laitinen M, Hartikainen J, Hiltunen MO et al (2000) Catheter-mediated vascular endothelial growth factor gene transfer to human coronary arteries after angioplasty. Hum Gene Ther 11(2):263–270
Boyd SR, Tan D, Bunce C et al (2002) Vascular endothelial growth factor is elevated in ocular fluids of eyes harbouring uveal melanoma: identification of a potential therapeutic window. Br J Ophthalmol 86(4):448–452
Adamis AP, Miller JW, Bernal MT et al (1994) Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am J Ophthalmol 118(4):445–450
Moulton KS, Heller E, Konerding MA, Flynn E, Palinski W, Folkman J (1999) Angiogenesis inhibitors endostatin or TNP-470 reduce intimal neovascularization and plaque growth in apolipoprotein E-deficient mice. Circulation 99(13):1726–1732
Sueishi K, Kumamoto M, Sakuda H, Tanaka K (1993) Angiogenic processes in the pathogenesis of human coronary atherosclerosis. Curr Top Pathol 87:47–58
Ignatescu MC, Gharehbaghi-Schnell E, Hassan A et al (1999) Expression of the angiogenic protein, platelet-derived endothelial cell growth factor, in coronary atherosclerotic plaques: in vivo correlation of lesional microvessel density and constrictive vascular remodeling. Arterioscler Thromb Vasc Biol 19(10):2340–2347
Libby P, Sukhova G, Lee RT, Galis ZS (1995) Cytokines regulate vascular functions related to stability of the atherosclerotic plaque. J Cardiovasc Pharmacol 25(Suppl 2):S9–S12
Flugelman MY, Virmani R, Correa R et al (1993) Smooth muscle cell abundance and fibroblast growth factors in coronary lesions of patients with nonfatal unstable angina. A clue to the mechanism of transformation from the stable to the unstable clinical state. Circulation 88(6):2493–2500
Van Belle E, Tio FO, Chen D, Maillard L, Kearney M, Isner JM (1997) Passivation of metallic stents after arterial gene transfer of phVEGF165 inhibits thrombus formation and intimal thickening. J Am Coll Cardiol 29(6):1371–1379
Asahara T, Bauters C, Pastore C et al (1995) Local delivery of vascular endothelial growth factor accelerates reendothelialization and attenuates intimal hyperplasia in balloon-injured rat carotid artery. Circulation 91(11):2793–2801
Isner JM (1998) Arterial gene transfer of naked DNA for therapeutic angiogenesis: early clinical results. Adv Drug Deliv Rev 30(1–3):185–197
Dewey RA, Morrissey G, Cowsill CM et al (1999) Chronic brain inflammation and persistent herpes simplex virus 1 thymidine kinase expression in survivors of syngeneic glioma treated by adenovirus-mediated gene therapy: implications for clinical trials. Nat Med 5(11):1256–1263
Wersto RP, Rosenthal ER, Seth PK, Eissa NT, Donahue RE (1998) Recombinant, replication-defective adenovirus gene transfer vectors induce cell cycle dysregulation and inappropriate expression of cyclin proteins. J Virol 72(12):9491–9502
Hollon T (2000) Researchers and regulators reflect on first gene therapy death. Nat Med 6(1):6
Seiler C, Pohl T, Wustmann K et al (2001) Promotion of collateral growth by granulocyte-macrophage colony-stimulating factor in patients with coronary artery disease: a randomized, double-blind, placebo-controlled study. Circulation 104(17):2012–2017
Zbinden S, Zbinden R, Meier P, Windecker S, Seiler C (2005) Safety and efficacy of subcutaneous-only granulocyte-macrophage colony-stimulating factor for collateral growth promotion in patients with coronary artery disease. J Am Coll Cardiol 46(9):1636–1642
Belardinelli R, Belardinelli L, Shryock JC (2001) Effects of dipyridamole on coronary collateralization and myocardial perfusion in patients with ischaemic cardiomyopathy. Eur Heart J 22(14):1205–1213
Henry TD, Grines CL, Watkins MW et al (2007) Effects of Ad5FGF-4 in patients with angina: an analysis of pooled data from the AGENT-3 and AGENT-4 trials. J Am Coll Cardiol 50(11):1038–1046
Hedman M, Hartikainen J, Syvanne M et al (2003) Safety and feasibility of catheter-based local intracoronary vascular endothelial growth factor gene transfer in the prevention of postangioplasty and in-stent restenosis and in the treatment of chronic myocardial ischemia: phase II results of the Kuopio Angiogenesis Trial (KAT). Circulation 107(21):2677–2683
Stewart DJ, Kutryk MJ, Fitchett D et al (2009) VEGF gene therapy fails to improve perfusion of ischemic myocardium in patients with advanced coronary disease: results of the NORTHERN trial. Mol Ther 17(6):1109–1115
Kukula K, Chojnowska L, Dabrowski M et al (2011) Intramyocardial plasmid-encoding human vascular endothelial growth factor A165/basic fibroblast growth factor therapy using percutaneous transcatheter approach in patients with refractory coronary artery disease (VIF-CAD). Am Heart J 161(3):581–589
Ruel M, Beanlands RS, Lortie M et al (2008) Concomitant treatment with oral l-arginine improves the efficacy of surgical angiogenesis in patients with severe diffuse coronary artery disease: the endothelial modulation in angiogenic therapy randomized controlled trial. J Thorac Cardiovasc Surg 135(4):762–770
Zhao Q, Sun Y, Xia L, Chen A, Wang Z (2008) Randomized study of mononuclear bone marrow cell transplantation in patients with coronary surgery. Ann Thorac Surg 86(6):1833–1840
Tse HF, Thambar S, Kwong YL et al (2007) Prospective randomized trial of direct endomyocardial implantation of bone marrow cells for treatment of severe coronary artery diseases (PROTECT-CAD trial). Eur Heart J 28(24):2998–3005
Menasche P, Alfieri O, Janssens S et al (2008) The myoblast autologous grafting in ischemic cardiomyopathy (MAGIC) trial: first randomized placebo-controlled study of myoblast transplantation. Circulation 117(9):1189–1200
Schachinger V, Assmus B, Britten MB et al (2004) Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction: final one-year results of the TOPCARE-AMI trial. J Am Coll Cardiol 44(8):1690–1699
Beitnes JO, Gjesdal O, Lunde K et al (2011) Left ventricular systolic and diastolic function improve after acute myocardial infarction treated with acute percutaneous coronary intervention, but are not influenced by intracoronary injection of autologous mononuclear bone marrow cells: a 3 year serial echocardiographic sub-study of the randomized-controlled ASTAMI study. Eur J Echocardiogr 12(2):98–106
Egeland T, Brinchmann JE (2007) The REPAIR-AMI and ASTAMI trials: cell isolation procedures. Eur Heart J 28(17):2174–2175 (author reply 2175)
Mills JS, Rao SV (2007) REPAIR-AMI: stem cells for acute myocardial infarction. Future Cardiol 3(2):137–140
Osterziel KJ (2007) Improved clinical outcome after intracoronary administration of bone-marrow-derived progenitor cells in acute myocardial infarction: final 1 year results of the REPAIR-AMI trial. Eur Heart J 28(5):638
Schachinger V, Assmus B, Erbs S et al (2009) Intracoronary infusion of bone marrow-derived mononuclear cells abrogates adverse left ventricular remodelling post-acute myocardial infarction: insights from the reinfusion of enriched progenitor cells and infarct remodelling in acute myocardial infarction (REPAIR-AMI) trial. Eur J Heart Fail 11(10):973–979
Ripa RS, Kastrup J (2009) Stem cells: REGENT trial-the end of cell therapy for MI? Nat Rev Cardiol 6(9):567–568
Tendera M, Wojakowski W, Ruzyllo W et al (2009) Intracoronary infusion of bone marrow-derived selected CD34+ CXCR4+ cells and non-selected mononuclear cells in patients with acute STEMI and reduced left ventricular ejection fraction: results of randomized, multicentre myocardial regeneration by intracoronary infusion of selected population of stem cells in acute myocardial infarction (REGENT) trial. Eur Heart J 30(11):1313–1321
Gyongyosi M, Lang I, Dettke M et al (2009) Combined delivery approach of bone marrow mononuclear stem cells early and late after myocardial infarction: the MYSTAR prospective, randomized study. Nat Clin Pract Cardiovasc Med 6(1):70–81
Acknowledgments
This work was supported in part by a University of Patras, Research Programme “K. Karatheodori”. Sofoclis Mitsos was also supported by a scholarship for doctoral studies of Alexander S. Onassis Public Benefit Foundation, Greece.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mitsos, S., Katsanos, K., Koletsis, E. et al. Therapeutic angiogenesis for myocardial ischemia revisited: basic biological concepts and focus on latest clinical trials. Angiogenesis 15, 1–22 (2012). https://doi.org/10.1007/s10456-011-9240-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-011-9240-2