Abstract
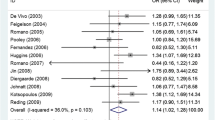
Published data on the association between p21 Ser31Arg polymorphism and breast cancer risk are inconclusive. To derive a more precise estimation of the relationship, a meta-analysis was performed. Crude ORs with 95% CIs were used to assess the strength of association between the p21 Ser31Arg polymorphism and breast cancer risk. The pooled ORs were performed for co-dominant model (Ser/Arg vs. Ser/Ser, Arg/Arg vs. Ser/Ser), dominant model (Arg/Arg + Ser/Arg vs. Ser/Ser), and recessive model (Arg/Arg vs. Ser/Arg + Ser/Ser). A total of 21 studies including 22,109 cases and 29,127 controls were involved in this meta-analysis. Overall, no significant associations were found between p21 Ser31Arg polymorphism and breast cancer risk when all studies pooled into the meta-analysis. In the subgroup analysis by ethnicity, significantly increased risk was found for Caucasians (Arg/Arg vs. Ser/Ser: OR 1.496, 95% CI 1.164–1.924; and recessive model: OR 1.492, 95% CI 1.161–1.919). When stratified by study design, statistically significantly elevated risk was found for population-based studies (Ser/Arg vs. Ser/Ser: OR 1.085, 95% CI 1.019–1.156). In conclusion, this meta-analysis suggests that the p21 Ser31Arg polymorphism may be associated with breast cancer development in Caucasian. However, large sample and representative population-based studies with homogeneous breast cancer patients and well-matched controls are warranted to confirm this finding.
Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics 2002. CA Cancer J Clin 55:74–108
Pharoah PD, Dunning AM, Ponder BA, Easton DF (2004) Association studies for finding cancer-susceptibility genetic variants. Nat Rev Cancer 4:850–860
Qiu LX, Yuan H, Yu KD, Mao C, Chen B, Zhan P, Xue K, Zhang J, Hu XC (2009) Glutathione S-transferase M1 polymorphism and breast cancer susceptibility: a meta-analysis involving 46,281 subjects. Breast Cancer Res Treat. doi:10.1007/s10549-009-0636-2
Qiu LX, Yao L, Yuan H, Mao C, Chen B, Zhan P, Xue K, Zhang J, Hu XC (2010) IGFBP3 A-202C polymorphism and breast cancer susceptibility: a meta-analysis involving 33,557 cases and 45,254 controls. Breast Cancer Res Treat. doi: 10.1007/s10549-010-0739-9
Qiu LX, Chen B, Mao C, Zhan P, Yuan H, Xue K, Li J, Hu XC (2009) STK15 F31I polymorphism is associated with breast cancer risk: a meta-analysis involving 25, 014 subjects. Breast Cancer Res Treat 118:599–603
Gartel AL, Radhakrishnan SK (2005) Lost in transcription: p21 repression, mechanisms, and consequences. Cancer Res 65:3980–3985
Mousses S, Ozcelik H, Lee PD, Malkin D, Bull SB, Andrulis IL (1995) Two variants of the CIP1/WAF1 gene occur together and are associated with human cancer. Hum Mol Genet 4:1089–1092
Lukas J, Groshen S, Saffari B (1997) WAF1/Cip1 gene polymorphism and expression in carcinomas of the breast, ovary, and endometrium. Am J Pathol 150:167–175
The MARIE-GENICA Consortium on Genetic Susceptibility for Menopausal Hormone Therapy Related Breast Cancer Risk (2009) Polymorphisms in the BRCA1 and ABCB1 genes modulate menopausal hormone therapy associated breast cancer risk in postmenopausal women. Breast Cancer Res Treat. doi:10.1007/s10549-009-0489-8
Ma H, Jin G, Hu Z, Zhai X, Chen W, Wang S (2006) Variant genotypes of CDKN1A and CDKN1B are associated with an increased risk of breast cancer in Chinese women. Int J Cancer 119:2173–2178
Tarasov VA, Aslanyan MM, Tsyrendorzhiyeva ES, Litvinov SS, Gar’kavtseva RF, Altukhov YP (2006) Genetically determined subdivision of human populations with respect to the risk of breast cancer in women. Dokl Biol Sci 406:66–69
Keshava C, Frye BL, Wolff MS, McCanlies EC, Weston A (2002) Waf-1 (p21) and p53 polymorphisms in breast cancer. Cancer Epidemiol Biomarkers Prev 11:127–130 (erratum in: Cancer Epidemiol Biomarkers Prev 2004;13:1682)
Cox A, Dunning AM, Garcia-Closas M (2007) A common coding variant in CASP8 is associated with breast cancer risk. Nat Genet 39:352–358
Cochran WG (1954) The combination of estimates from different experiments. Biometrics 10:101–129
Mantel N, Haenszel W (1959) Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst 22:719–748
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clini Trials 7:177–188
Tobias A (1999) Assessing the influence of a single study in the meta-analysis estimate. Stata Tech Bull 8:15–17
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Hirschhorn JN, Lohmueller K, Byrne E (2002) A comprehensive review of genetic association studies. Genet Med 4:45–61
Wacholder S, Chanock S, Garcia-Closas M (2004) Assessing the probability that a positive report is false: an approach for molecular epidemiology studies. J Natl Cancer Inst 96:434–442
Author information
Authors and Affiliations
Corresponding authors
Additional information
Li-Xin Qiu, Jian Zhang, and Xiao-Dong Zhu contributed equally to this work and are considered as co-first authors.
Rights and permissions
About this article
Cite this article
Qiu, LX., Zhang, J., Zhu, XD. et al. The p21 Ser31Arg polymorphism and breast cancer risk: a meta-analysis involving 51,236 subjects. Breast Cancer Res Treat 124, 475–479 (2010). https://doi.org/10.1007/s10549-010-0858-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-010-0858-3