Abstract
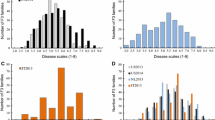
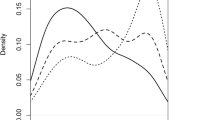
Hop powdery mildew [Podosphaera macularis (Wallr.) U. Braun & S. Takam.] is best controlled via the production of resistant varieties. Recent evidence supports selection against plant susceptibility genes to fungal pathogens as a more durable resistance mechanism than selection for resistance genes. The objective of this study was to identify molecular-based QTLs, their genetic effects and epistasis among QTLs associated with susceptibility to powdery mildew. Parents and offspring from the cross, ‘Perle’ × ‘USDA 19058M’, were clonally replicated and inoculated in a greenhouse using a CRD experimental design in Corvallis, OR. DNA was extracted, purified and analyzed via three different marker systems. Analysis of the resulting markers was based upon the “two-way pseudo-testcross” procedure. QTL mapping using multiple interval mapping and Bayesian interval mapping analyses were performed using WinQTL Cartographer 2.5_003. Comparison amongst mapping analyses identified three persistent QTLs on three linkage groups without significant epistatic effect upon expression. The persistent QTL on linkage group C7 had both additive and dominant effects controlling phenotype expression. The presence or absence of the two AFLP markers bordering the QTL on C7 defined susceptibility in offspring. This is the first report in hop identifying molecular markers linked to QTLs associated with disease susceptibility.


Similar content being viewed by others
References
Asins MJ (2002) Present and future of quantitative trait locus analysis in plant breeding. Plant Breed 121:281–291
Bassil NV, Gilmore B, Oliphant J, Henning J, Hummer K (2005) Genbank derived microsatellite markers in hop. Acta Hort 668:47–52
Beckmann JS, Soller M (1988) Detection of linkage between marker loci and loci affecting quantitative traits in crosses between segregating populations. Theor Appl Genet 76:228–236
Benham J, Jeung J-U, Jasieniuk M, Kanazin V, Blake T (1999) Genographer: a graphical tool for automated fluorescent AFLP and microsatellite analysis. http://wheat.pw.usda.gov/jag/papers99/paper399/1999_P3.html. Accessed 29 Sept 2009
Cerenak A, Satovic Z, Javornik B (2006) Genetic mapping of hop (Humulus lupulus L.) applied to the detection of QTLs for alpha-acid content. Genome 49:485–494
Cerenak A, Kralj D, Javornik B (2009) Compounds of essential oils as markers of hop resistance (Humulus lupulus) to powdery mildew (Podosphaera macularis). Acta Agric Slov [Print] 93(3): 267–273. http://dx.doi.org/10.2478/v10014-009-0015-z
Cerenak A, Satovic Z, Jakse J, Luthar Z, Carovic-Stanko K, Javornik B (2009) Identification of QTLs for alpha acid content and yield in hop (Humulus lupulus L.). Euphytica Published Online, March 31, 2009. doi: 10.1007/s10681-009-9920-9
Doerge R (2002) Mapping and analysis of quantitative trait loci in experimental populations. Nat Rev 3:43–52
Flor HH (1971) Current status of the gene-for-gene concept. Ann Rev Phytopathol 9:275–296
Grattapaglia D, Sederoff R (1994) Genetic linkage maps of Eucalyptus grandis and Eucalyptus urophylla using a pseudo-testcross: mapping strategy and RAPD markers. Genetics 137:1121–1137
Haunold A (1988) Notice of release of USDA 19058M male hop germplasm. USDA-ARS Publications, Beltsville
Haunold A, Likens ST, Nickerson GB, Hampton RO (1984) Registration of Nugget hop. Crop Sci 24:618
Henning JA, Townsend MS, Mahaffee W, Kenny S, Haunold A (2004) Registration of ‘Newport’ hop. Crop Sci 44:1018–1019
Jaccoud D, Peng K, Feinstein D, Kilian A (2001) Diversity arrays: a solid state technology for sequence information independent genotyping. Nucleic Acids Res 29:e25
Jakse J, Bandelj D, Javornik B (2002) Eleven new microsatellites for hop (Humulus lupulus L.). Mol Ecol Notes 2:544–546
Kao CH, Zeng ZB, Teasdale R (1999) Multiple interval mapping for quantitative trait loci. Genetics 140:1111–1127
Koie K, Inaba A, Okada Y, Kaneko T, Ito K (2005) Construction of the genetic linkage map and QTL analysis on hop (Humulus lupulus L.). Acta Hort (ISHS) 668:59–67
Kosambi DD (1944) The estimation of map distance from recombination values. Ann Eugen 12:172–175
Lander ES, Botstein D (1989) Mapping Mendelian factors underlying quantitative traits using RFLP linkage maps. Genetics 121:185–199
Luo ZW, Kearsey MJ (1989) Maximum likelihood estimation of linkage between a marker gene and a quantitative trait locus. Heredity 63:401–408
Mackay TFC (2001) Quantitative trait loci in Drosophila. Nat Rev Genet 2:11–20
Mahaffee WF, Engelhard B, Gent DH, Groves GG (2009) Powdery mildew. In: Mahaffee WF, Pethybridge SJ, Gent DH (eds) Compendium of hop diseases and pests. American Phytopathological Society Press, St. Paul
Mehlenbacher SA, Brown RN, Nouhra ER, Gökirmak T, Bassil NV, Kubisiak TL (2006) A genetic linkage map for hazelnut (Corylus avellana L.) based on RAPD and SSR markers. Genome 49:122–133
Neve RA (1991) Hops. Chapman and Hall Publishers, London 266
Pavan S, Jacobsen E, Visser R, Bai Y (2010) Loss of susceptibility as a novel breeding strategy for durable and broad-spectrum resistance. Mol Breed 25:1–12
Satagopan JM, Yandell BS, Newton MA, Osborn TC (1996) A Bayesian approach to detect quantitative trait loci using Markov Chain Monte Carlo. Genetics 144:805–816
Seefelder S, Ehrmaier H, Schweizer G, Seigner E (2000) Male and female genetic linkage map of hops, Humulus lupulus. Plant Breed 119:249–255
Seefelder S, Lutz A, Seigner E (2006) Development of molecular markers for powdery mildew resistance support breeding for high quality hops. Monatsschrift für Brauwissenschaft 5(6):100–102
Seigner E, Lutz A, Radic-Miehle H, Seefelder S (2005) Breeding for powdery mildew resistance in hop (Humulus L.): strategies at the Hop Research Center, Huell, Germany. Acta Hort 668:19–29
Townsend MS, Henning JA (2005) Potential heterotic groups in hop as determined by AFLP analysis. Crop Sci 45:1907–1910
Townsend MS, Henning JA, Moore DL (2000) AFLP analysis of DNA from dried hop cones. Crop Sci 40:1383–1386
Van Ooijen JW, Voorrips RE (2001) Joinmap® 3.0, software for the calculation of genetic linkage maps. Plant Research International, Wageningen
Wang S, Basten CJ, Zeng Z-B (2010) Windows QTL Cartographer 2.5. Department of Statistics, North Carolina State University, Raleigh. http://statgen.ncsu.edu/qtlcart/WQTLCart.htm. Accessed 9 Aug 2010
Whittock S, Leggett G, Jakše J, Javornik B, Carling J, Kilian A, Matthews PD, Probasco G, Henning JA, Darby P, Čerenak A, Koutoulis A (2009) Use of diversity array technology (DArT) for genotyping of Humulus lupulus L. Acta hort (ISHS) 848:59–64
Wittenberg AH, Van Der Lee T, Cayla C, Kilian A, Visser RG, Schouten HJ (2005) Validation of the high-throughput marker technology DArT using the model plant Arabidopsis thaliana. Mol Gen Genet 274:30–39
Xia L, Peng K, Yang S, Wenzl P, Vicente MC, Fregene M, Kilian A (2005) DArT for high-throughput genotyping of cassava (Manihot esculenta) and its wild relatives. Theor Appl Genet 110:1092–1098
Zeng ZB (1993) Theoretical basis of precision mapping of quantitative trait loci. Proc Nat Acad Sci 90:10972–10976
Zeng ZB (1994) Precision mapping of quantitative trait loci. Genetics 136:1457–1468
Zeng ZB, Kao CH, Basteen CJ (1999) Estimating the genetic architecture of quantitative traits. Genet Res 74:279–289
Acknowledgments
The authors wish to acknowledge support from the International Hop DArT Consortium (Members listed alphabetically by first name: Andreja Cerenak; Anthony Koutoulis; Branka Javornik; Claudia Wiedow; Emily Buck; Erin Howard; Gene Probasco; Jernej Jakse; John Henning; Jules Freeman; Paul Matthews; Peter Darby; Rene Vaillancourt; Ron Beatson and Simon Whittock) along with their respective institutions for providing information and support to screen the USDA-ARS mapping population used in this study with diversity array technology (DArT) markers (www.diversityarray.com/). We would also like to thank Andrzej Kilian and Diversity Array Technology Pty Ltd for providing the DArT marker data for QTL mapping analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Henning, J.A., Townsend, M.S., Gent, D.H. et al. QTL mapping of powdery mildew susceptibility in hop (Humulus lupulus L.). Euphytica 180, 411–420 (2011). https://doi.org/10.1007/s10681-011-0403-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-011-0403-4