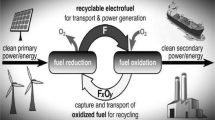
Abstract
Conceptually, thermochemical cycles are heat engines that drive endothermic chemical reactions, e.g., splitting water into hydrogen and oxygen. The two-step metal oxide cycles (typically ferrite-based) are particularly attractive since they are relatively simple, use non-corrosive materials, and involve gas–solid reactions requiring no difficult separations. Additionally, they are potentially the most efficient renewable-energy driven processes for hydrogen production. We are developing a novel concentrating solar power (CSP) driven metal-oxide-based heat engine, the CR5, at the heart of which are rings of a reactive solid that are thermally and chemically cycled to produce oxygen and hydrogen from water in separate and isolated steps. The monolithic ring structures must have high geometric surface area for gas–solid contact and for adsorption of incident solar radiation, and must maintain structural integrity and high reactivity after extensive thermal cycling to temperatures of at least 1,400 °C. We have demonstrated through laboratory and on-sun testing that cobalt ferrite/zirconia mixtures fabricated into monolithic structures suitable for the CR5 are mechanically robust and maintain productivity over tens of cycles. We have also demonstrated that carbon dioxide splitting (CDS) to carbon monoxide and oxygen is a thermodynamically favorable alternative to water splitting that can be conducted with both iron- and cerium-based materials.














Similar content being viewed by others
References
Lewis NS, Nocera DG (2006) Proc Natl Acad Sci USA 103:15729
Dukes JS (2003) Clim Change 61:31
Ivy J (2004) Summary of electrolytic hydrogen production. National Renewable Energy Laboratory report NREL/MP-560-36734
Abanades S, Charvin P, Flamant G, Neveu P (2006) Energy 31:2469
Funk JE, Reinstrom RM (1966) Ind Eng Chem Process Des Develop 5:336; Abraham BA, Schreiner F (1974) Ind Eng Chem Fund 13:305
Nakamura T (1977) Sol Energy 19:467
Kodama T, Kondoh Y, Kiyama A, Shimizu K (2003) In: Thornbloom MD, Jones SA, (eds) Proceedings of the ASME international solar energy conference (ISEC), ASME, Hawaii, p 121
Ehrensberger K, Kuhn P, Shklover V, Oswald HR (1996) Solid State Ionics 90:75
Ehrensberger K, Frei A, Kuhn P, Oswald HR, Hug P (1995) Solid State Ionics 78:151
Tamaura Y, Steinfeld A, Kuhn P, Ehrensberger K (1995) Energy 20:325
Tamaura Y, Kojima M, Sano T, Ueda Y, Hasegawa N, Tsuji M (1998) Int J Hydrogen Energy 23:1185
Ishihara H, Kaneko H, Yokoyama T, Fuse A, Tamaura Y (2006) In: Proceedings of the 2005 international solar energy conference (ISEC), ASME, Orlando, FL, 2005, p 687
Aoki H, Kaneko H, Hasegawa N, Ishihara H, Takahashi Y, Suzuki A, Tamaura Y (2004) In Proceedings of 2004 international solar energy conference (ISEC), ASME, Portland, OR, p 515
Kodama T, Kondoh Y, Yamamoto R, Andou H, Satou N (2005) Sol Energy 78:623
Diver RB, Miller JE, Allendorf MD, Siegel NP, Hogan RE (2006) In: Proceedings of the 2006 international solar energy conference (ISEC), ASME, Denver, CO
Miller JE, Evans LR, Stuecker JN, Allendorf MD, Siegel NP, Diver RB (2006) In: Proceedings of the 2006 international solar energy conference (ISEC), ASME, Denver, CO
James DL, Siegel NP, Diver RB, Boughton BD, Hogan RE (2006) In: Proceedings of the 2006 international solar energy conference (ISEC), ASME, Denver, CO
Allendorf MD, Miller JE, Siegel NP, Diver RB (2006) In: Proceedings of the 2006 international solar energy conference (ISEC), ASME, Denver, CO
Erickson DS, Mason TO (1985) J Solid State Chem 59:42
Allendorf MD, Miller JE, Siegel NP, Diver RB, Energy Fuel (submitted)
Roeb M, Sattler C, Kluser R, Monnerie L, Konstandopoulos AG, Agrafiotis C, Zaspalis VT, Nalbandian L, Steele A, Stobbe P (2006) In: Proceedings of the 2005 international solar energy conference (ISEC), ASME, Orlando, FL, 2005, p 671
Stuecker JN, Miller JE, Ferrizz RE, Mudd JE, Cesarano J III (2004) Ind Eng Chem Res 43:51
Stuecker JN, Cesarano J III, Hirschfeld DA (2003) J Mater Process Technol 142:318; Cesarano J III, Calvert PD (2000) U.S. Patent 6,027,326; Cesarano J III, Segalman R, Calvert P (1998) Ceramic Ind 148:94
Ferrizz RM, Stuecker JN, Cesarano J III, Miller JE (2005) Ind Eng Chem Res 44:302
Diver RB, Siegel NP, Miller JE, Moss TA, Stuecker JN, James DL (2008) In: Proceedings of 2008 14th Biennial CSP solar PACES symposium, Las Vegas, NV
McCarty JG, Gusman M, Lowe DM, Hildenbrand DL, Lau KN (1999) Catal Today 47:5
Abanades S, Flamant G (2006) Sol Energy 80:1611
Sharma S, Hilaire S, Vohs JM, Gorte RJ, Jen H-W (2000) J Catal 190:199
Bernal S, Blanco G, Gatica JM, Larese C, Vidal H (2001) J Catal 200:411
Demoulin O, Navez BM, Mugabo JL, Ruiz P (2007) Appl Catal B 70:284
Acknowledgement
Sandia is a multiprogram laboratory operated by Sandia Corporation, a Lockheed Martin Company, for the United States Department of Energy’s National Nuclear Security Administration under Contract DE-AC04-94AL85000.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miller, J.E., Allendorf, M.D., Diver, R.B. et al. Metal oxide composites and structures for ultra-high temperature solar thermochemical cycles. J Mater Sci 43, 4714–4728 (2008). https://doi.org/10.1007/s10853-007-2354-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-007-2354-7