Abstract
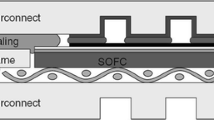
In planar solid oxide fuel and electrolyzer cells, compatibility and thermochemical stability of interconnect-seal glass interface is essential in order to avoid mixing and leakage of different gases and degradation of cell performances. In the present work, interfacial compatibility and thermochemical stability are studied for an alkaline earth silicate based glass (SABS-0) and Crofer 22 APU interconnect system with respect to thermal treatment temperature (700–850 °C) and time (0–100 h). The study has been carried out in argon to avoid complications from oxidation. Even though pore and crack-free interface is obtained and maintained for all the thermal treatment conditions, there are simultaneous diffusion of the Crofer 22 APU and the SABS-0 glass elements, chemical reaction at the Crofer 22 APU/SABS-0 interface, and devitrification of the SABS-0 glass itself.







Similar content being viewed by others
References
EG & G Technical Services (2004) Fuel cell handbook, 7th edn. US Department of Energy, Office of Fossil Energy, National Energy Technological Laboratory, pp 7–12
Singhal SC, Kendall K (2003) High temperature solid oxide fuel cells. Elsevier, Oxford, p 217
Lessing PA (2007) J Mater Sci 42:3465. doi:10.1007/s10853-006-0409-9
Fergus JW (2005) J Power Sources 147:46
Batfalsky P, Haanappel VAC, Malzbender J, Menzler NH, Shemet V, Vinke IC, Steinbrech RW (2006) J Power Sources 155:128
Yang Z, Xia G, Meinhardt KD, Weil KS, Stevenson JW (2004) J Mater Eng Perform 13:327
Yang Z, Meinhardt KD, Stevenson JW (2003) J Electrochem Soc 150:A1095
Haanappel VAC, Shemet V, Vinke IC, Gross SM, Koppitz TH, Menzler NH, Zahid M, Quadakkers WJ (2005) J Mater Sci 40:1583. doi:10.1007/s10853-005-0657-0
Lahl N, Bahadur D, Singh K, Singheiser L, Hilpert K (2002) J Electrochem Soc 149:A607
Chou YS, Stevenson JW, Singh P (2007) J Electrochem Soc 154:B644
Chou YS, Stevenson JW, Gow RN (2007) J Power Sources 170:395
Smeacetto F, Salvo M, Ferraris M, Cho J, Boccaccini AR (2008) J Eur Ceram Soc 28:61
Melling PJ, Vempati CJ, Allmatt AR, Jacobs PWM (1981) Phys Chem Glasses 22:49
Ogasawara K, Kameda H, Matsuzaki Y, Sakurai T, Uehara T, Toji A, Sakai N, Yamaji K, Horita T, Yokokawa H (2007) J Electrochem Soc 154:B657
Lu K, Mahapatra MK (2008) J Appl Phys 104:074910
Mahapatra MK, Lu K, Bodnar RJ (2009) Appl Phys A 95:493
Mahapatra MK, Lu K (2008) J Power Sources 185:993
Cullity BD (1978) Elements of X-ray diffraction, 2nd edn. Addison-Wesley Publishing Company Inc., Philippines, pp 292–294
Smeacetto F, Salvo M, Ferraris M, Casalegno V, Asinari P, Chrysanthou A (2008) J Eur Ceram Soc 28:2521
Lara C, Pascual MJ, Durán A (2007) Phys Chem Glasses Eur J Glass Sci Tech B 48:218
Konysheva E, Laatsch J, Wessel E, Tietz F, Christiansen N, Singheiser L, Hilpert K (2006) Solid State Ionics 177:923
Mahapatra MK, Story C, Lu K, Reynolds WT Jr (2007) Energy: fuel cells: materials, processing, manufacturing and power management technologies. In: Proceedings of materials science and technology, Detroit, MI, USA, September 16–20, pp 371–380. Organizers: Singh P, Azad, Collins DC, Kumta PN, Legzdins C, Manthiram A, Manivannan A, Sundaram SK, Yang ZG
Mahapatra MK, Lu K, Reynolds WT Jr (2008) J Power Sources 179:106
Kumar P, Greenhut VA (1990) Metal-ceramic joining. TMS Minerals, Metals, Materials, Warrendale, PA, pp 3–11
Yang Z, Xia G, Wang CM, Nie Z, Templeton J, Stevenson JW, Singh P (2008) J Power Sources 183:660
Quadadakkers WJ, Shemet V, Singheiser L (2003) US Patent 0,059,335
Beranger G, Armanet F, Lambertin M (1989) In: Lang E (ed) The role of active elements in the oxidation behavior of high temperature metals and alloys. Elsevier Science Publishing Co. Inc., New York, pp 33–51
Bennet MJ, Moon DP (1989) In: Lang E (ed) The role of active elements in the oxidation behavior of high temperature metals and alloys. Elsevier Science Publishing Co. Inc., New York, pp 111–129
Lobnig RE, Schmidt HP, Hennesen K, Grabke HJ (1992) Oxid Met 37:81
Cox MGC, McEnaney B, Scott VD (1972) Philos Mag 2:839
Klug HP, Alexander LE (1974) X-ray diffraction procedures for polycrystalline and amorphous materials, 2nd edn. Wiley, New York
Cromier L, Calas G, Creux S, Gaskell PH, Bouchet-Fabre B, Hannon AC (1999) Phys Rev B 59:13517
Schaller T, Stebbins JF, Wilding MC (1999) J Non-Cryst Solids 243:146
Wilding MC, Navrotsky A (2000) J Non-Cryst Solids 265:238
Jiang N, Silox J (2000) J Appl Phys 87:3768
Volf MB (1984) Chemical approaches to glass: glass science and technology, vol 7. Elsevier, Amsterdam, pp 83–88
Kim YW, Belton GR (1974) Metal Trans 5:1811
Ebbinghaus BB (1993) Combust Flame 93:119
Caplan D, Cohen M (1961) J Electrochem Soc 108:438
Pask JA (1987) In: Moddeman WE, Merten CW, Kramer DP (eds) Technology of glass,ceramic or glass-ceramic to metal sealing. The American Society of Mechanical Engineers, New York, pp 1–7
Acknowledgements
This material is based on work supported by Department of Energy under Award Number DE-FC07-06ID14739. The SEM analysis was done in Nanoscale Characterization and Fabrication Laboratory (NCFL), Virginia Tech. The authors are indebted to Dr. Zhenguo “Gary” Yang, Pacific Northwest National Laboratory, Richland, Washington, for providing Crofer 22 APU alloy for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mahapatra, M.K., Lu, K. Interfacial study of Crofer 22 APU interconnect-SABS-0 seal glass for solid oxide fuel/electrolyzer cells. J Mater Sci 44, 5569–5578 (2009). https://doi.org/10.1007/s10853-009-3781-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-009-3781-4