Abstract
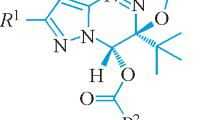
The title compound (NC4H8NCH3)(C6H5)[N(C6H11)2]P{—N–S3N3 crystallizes in the triclinic crystal system with unit cell dimensions: a = 9.7572(2), b = 10.6845(1), c = 16.9381(3) Å, α = 71.75(1)∘, β = 80.65(2)∘, γ = 65.93(1)∘, and Z = 2. The tricoordinated sulfur atom (S1) in the cyclotrithiazene deviates from the mean plane of the other five atoms by 0.716(6) Å, which is the highest among the known phosphiniminocyclotrithiazene structures. In the exocyclic P{—N4–S1 moiety, the two bond distances are short: 1.585(6) and 1.527(6) Å; but the bond angle is large 135.3(4)∘. The S1 displacement is highly correlated with the endocyclic angle at the nitrogen atom (diagonally opposite to S1), but the nature of correlation is bimodal (R = 1.0 + or −1.0) for the cyclic and acyclic substituents (R) in the (R)(C6H5)[N(C6H11)2]P{—N–S3N3 series. On the other hand, the S1–N4 bond distances are negatively correlated (R = −0.94) with the P{—N4–S1 bond angle in the above series.
Similar content being viewed by others
References
Chivers, T.; Oakley, R.T.; Cordes, A.W.; Pennington, W.T. J. Chem. Soc. Chem. Commun. (1981), 1214.
Spang, C.; Edelmann, F.T.; Noltemeyer, M.; Roesky, H.W. Chem. Ber. (1989), 122, 1247.
Elias, A.J.; Rao, M.N.S. Inorg. Chim. Acta. (1989), 164, 45.
Thomas, C.J.; Rao, M.N.S. Heteroatom Chem. (1992), 3, 321.
Mohan, T.; Rao, M.N.S. Heteroatom Chem. (1997), 8, 225.
Holt, E.M.; Holt, S.L. J. Chem. Soc., Dalton Trans. (1974), 18, 1990.
Holt, E.M.; Holt, S.L.; Watson, K.J. J. Chem. Soc., Dalton Trans. (1977), 6, 514.
Liblong, S.W.; Oakley, R.T.; Cordes, A.W.; Noble, M.C. Can. J. Chem. (1983), 61, 2062.
Elias, A.J.; Rao, M.N.S.; Varghese, B. Polyhedron (1990), 9, 1433.
Thomas, C.J.; Bhandary, K.K.; Thomas, L.M.; Senadhi, S.E.; VijayKumar, S. Bull. Chem. Soc. Jpn. (1993), 66, 1830.
Thomas, C.J.; Cea-Olivares, R.; Espinosa-Perez, G.; Turner, R.W. J. Organometal. Chem. (1995), 493, 101.
Janaswamy, S.; Murthy, G.S.; Mohan, T.; Rao, M.N.S. Acta Cryst. (1996), C52, 1250.
Srinivas, J.; Murthy, G.S.; Thomas, C.J.; Rao, M.N.S. J. Chem. Crystallogr. (1996), 26, 403.
Srinivas, J.; Murthy, G.S.; Mohan, T.; Rao, M.N.S. Z. Krist-New Cryst. St. (1997), 212, 323.
Gopalakrishnan, J.; Rao, M.N.S.; Srinivas, J.; Murthy, G.S. Polyhedron (1997), 16, 1089.
Gopalakrishnan, J.; Srinivas, J.; Murthy, G.S.; Rao, M.N.S. P. Indian As-Chem. Sci. (1998), 110, 89.
Gopalakrishnan, J.; Srinivas, J.; Murthy, G.S.; Rao, M.N.S. Indian J. Chem. (1998), A37, 1052.
Srinivas, J.; Murthy, G.S.; Swarnalatha, U.; Rao, M.N.S. J. Chem. Crystallogr. (2001), 31, 267.
Janaswamy, S.; Murthy, G.S.; Mohan, T.; Rao, M.N.S. Kristallografiya (2003), 48, 73 [Crystallogr. Rep. (2003), 48, 68].
Srinivas, J.; Rao, M.V.; Murthy, G.S.; Thomas, C.J.; Rao, M.N.S. J. Chem. Crystallogr. (2004), 34, 19.
Sheldrick, G.M. SHELXS-86, Program for Solution of Crystal Structures, University of Göttingen: Germany, 1985.
Sheldrick, G.M. SHELXL-97, Crystal Structure Refinement-PC Version, University of Göttingen: Germany, 1997.
Johnson, C.K. ORTEPII, Report ORBNL-5138; Oak Ridge National Laboratory: TN, 1976.
Bats, J.W.; Coppens, P.; Koetzle, T.P. Acta Cryst. (1977), B33, 37.
Dean, J. A., Handbook of Organic Chemistry; McGraw-Hill: New York, 1987.
Bojes, J.; Chivers, T.; Laidlaw, W.G.; Trsic, M. J. Am. Chem. Soc. (1979), 101, 4517.
Banister, A.J.; Durrant, J.A. J. Chem. Res. (1978), 5, 150.
Craig, D.P.; Paddock, N.L. J. Chem. Soc. (1962), 4118.
Coulson, C.A. Nature (1969), 221, 1106.
Fluck, E.; Backe-Goehring, M.; Dehoust, G.Z. Z. Anong. Chem. (1961), 312, 60.
Jagg, P.N.; Kelly, P.F.; Rzepa, H.S.; Williams, D.J.; Woollins, J.D.; Wylie, W. J. Chem. Soc. Chem. Commun. (1991), 14, 942.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Janaswamy, S., Murthy, G.S., Mohan, T. et al. Crystal structure analysis of (N-methyl piperazino) (phenyl) (dicyclohexylamino) phosphiniminocyclotrithiazene. J Chem Crystallogr 35, 27–34 (2005). https://doi.org/10.1007/s10870-005-1150-y
Issue Date:
DOI: https://doi.org/10.1007/s10870-005-1150-y