Abstract
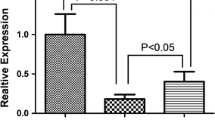
Myasthenia gravis (MG) are T-cell dependent antibody-mediated autoimmune disorders, microRNAs are important regulators of human autoimmune disease pathogenesis. Here, we investigated the miRNAs expression profiles in MG for the first time and found that miR-320a was significantly downregulated in MG patients compared to normal healthy people. Meanwhile, pro-inflammatory cytokins in MG patients were overexpressed. Furthermore, we identified MAPK1 as a direct target of miR-320a. Downregulation of miR-320a induced the overexpression of pro-inflammatory cytokins through promoting COX-2 expression. This process was modulated by ERK/ NF-κB pathways. Taken together, our findings suggested that miR-320a could play a role in modulation of inflammatory cytokins production.





Similar content being viewed by others
Abbreviations
- MG:
-
Myasthenia gravis
- MAPK1:
-
Mitogen-activated protein kinase1
- 3′UTR:
-
3′untranslated region
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- PBMCs:
-
Peripheral blood mononuclear cell
References
Tuzun E, Huda R, Christadoss P. Complement and cytokine based therapeutic strategies in myasthenia gravis. J Autoimmun. 2011;37:136–43.
Yang H, Zhang Y, Wu M, Li J, Zhou W, Li G, et al. Suppression of ongoing experimental autoimmune myasthenia gravis by transfer of RelB-silenced bone marrow dentritic cells is associated with a change from a T helper Th17/Th1 to a Th2 and FoxP3+ regulatory T-cell profile. Inflamm Res. 2010;59:197–205.
Afzali B, Lombardi G, Lechler RI, Lord GM. The role of T helper 17 (Th17) and regulatory T cells (Treg) in human organ transplantation and autoimmune disease. Clin Exp Immunol. 2007;148:32–46.
Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–33.
Pauley KM, Cha S, Chan EK. MicroRNA in autoimmunity and autoimmune diseases. J Autoimmun. 2009;32:189–94.
Nakasa T, Miyaki S, Okubo A, Hashimoto M, Nishida K, Ochi M, et al. Expression of microRNA-146 in rheumatoid arthritis synovial tissue. Arthritis Rheum. 2008;58:1284–92.
Tsitsiou E, Lindsay MA. microRNAs and the immune response. Curr Opin Pharmacol. 2009;9:514–20.
Pan W, Zhu S, Yuan M, Cui H, Wang L, Luo X, et al. MicroRNA-21 and microRNA-148a contribute to DNA hypomethylation in lupus CD4+ T cells by directly and indirectly targeting DNA methyltransferase 1. J Immunol. 2010;184:6773–81.
Jiang L, Cheng Z, Qiu S, Que Z, Bao W, Jiang C, et al. Altered let-7 expression in Myasthenia gravis and let-7c mediated regulation of IL-10 by directly targeting IL-10 in Jurkat cells. Int Immunopharmacol. 2012;14:217–23.
Xia MQ, Bacskai BJ, Knowles RB, Qin SX, Hyman BT. Expression of the chemokine receptor CXCR3 on neurons and the elevated expression of its ligand IP-10 in reactive astrocytes: in vitro ERK1/2 activation and role in Alzheimer’s disease. J Neuroimmunol. 2000;108:227–35.
Cedeno S, Cifarelli DF, Blasini AM, Paris M, Placeres F, Alonso G, et al. Defective activity of ERK-1 and ERK-2 mitogen-activated protein kinases in peripheral blood T lymphocytes from patients with systemic lupus erythematosus: potential role of altered coupling of Ras guanine nucleotide exchange factor hSos to adapter protein Grb2 in lupus T cells. Clin Immunol. 2003;106:41–9.
Bernard MP, Phipps RP. CpG oligodeoxynucleotides induce cyclooxygenase-2 in human B lymphocytes: implications for adjuvant activity and antibody production. Clin Immunol. 2007;125:138–48.
Hirano T, Oka K, Umezawa Y, Hirata M, Oh-i T, Koga M. Individual pharmacodynamics assessed by antilymphocyte action predicts clinical cyclosporine efficacy in psoriasis. Clin Pharmacol Ther. 1998;63:465–70.
Dimitri CA, Dowdle W, MacKeigan JP, Blenis J, Murphy LO. Spatially separate docking sites on ERK2 regulate distinct signaling events in vivo. Curr Biol. 2005;15:1319–24.
Zhang GX, Navikas V, Link H. Cytokines and the pathogenesis of myasthenia gravis. Muscle Nerve. 1997;20:543–51.
Williams LM, Lali F, Willetts K, Balague C, Godessart N, Brennan F, et al. Rac mediates TNF-induced cytokine production via modulation of NF-kappaB. Mol Immunol. 2008;45:2446–54.
Qi H, Li J, Allman W, Saini SS, Tuzun E, Wu X, et al. Genetic deficiency of estrogen receptor alpha fails to influence experimental autoimmune myasthenia gravis pathogenesis. J Neuroimmunol. 2011;234:165–7.
Vrolix K, Niks EH, Le Panse R, van Ostaijen-Ten Dam MM, Muris AH, der Zijde CM J-v, et al. Reduced thymic expression of ErbB receptors without auto-antibodies against synaptic ErbB in myasthenia gravis. J Neuroimmunol. 2011;232:158–65.
Sepramaniam S, Armugam A, Lim KY, Karolina DS, Swaminathan P, Tan JR, et al. MicroRNA 320a functions as a novel endogenous modulator of aquaporins 1 and 4 as well as a potential therapeutic target in cerebral ischemia. J Biol Chem. 2010;285:29223–30.
Diakos C, Zhong S, Xiao Y, Zhou M, Vasconcelos GM, Krapf G, et al. TEL-AML1 regulation of survivin and apoptosis via miRNA-494 and miRNA-320a. Blood. 2010;116:4885–93.
Sun JY, Huang Y, Li JP, Zhang X, Wang L, Meng YL, et al. MicroRNA-320a suppresses human colon cancer cell proliferation by directly targeting beta-catenin. Biochem Biophys Res Commun. 2012;420:787–92.
Mu L, Zhang Y, Sun B, Wang J, Xie X, Li N, et al. Activation of the receptor for advanced glycation end products (RAGE) exacerbates experimental autoimmune myasthenia gravis symptoms. Clin Immunol. 2011;141:36–48.
Wang HB, Shi FD, Li H, van der Meide PH, Ljunggren HG, Link H. Role for interferon-gamma in rat strains with different susceptibility to experimental autoimmune myasthenia gravis. Clin Immunol. 2000;95:156–62.
Conti-Fine BM, Milani M, Wang W. CD4+ T cells and cytokines in the pathogenesis of acquired myasthenia gravis. Ann N Y Acad Sci. 2008;1132:193–209.
Mu L, Sun B, Kong Q, Wang J, Wang G, Zhang S, et al. Disequilibrium of T helper type 1, 2 and 17 cells and regulatory T cells during the development of experimental autoimmune myasthenia gravis. Immunology. 2009;128:e826–36.
Shandley S, Martinez S, Krolick K. IL-4 receptor as a bridge between the immune system and muscle in experimental myasthenia gravis I: up-regulation of muscle IL-15 by IL-4. Clin Immunol. 2009;132:246–56.
Khan AQ, Khan R, Qamar W, Lateef A, Ali F, Tahir M, et al. Caffeic acid attenuates 12-O-tetradecanoyl-phorbol-13-acetate (TPA)-induced NF-kappaB and COX-2 expression in mouse skin: abrogation of oxidative stress, inflammatory responses and proinflammatory cytokine production. Food Chem Toxicol. 2012;50:175–83.
Aruna BV, Ben-David H, Sela M, Mozes E. A dual altered peptide ligand down-regulates myasthenogenic T cell responses and reverses experimental autoimmune myasthenia gravis via up-regulation of Fas-FasL-mediated apoptosis. Immunology. 2006;118:413–24.
Acknowledgments
This work was supported by Shanghai Science and Technology Commission (No. 10JC1414500) and the Shanghai Committee of Science and Technology [grant 11DZ2260600].
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
MiR-486-5p (A) and miR-363(B) expression differentiate 34 MG patients from 10 normal control by real-time PCR (JPEG 24 kb)
Online Resource 2
The bioinformatic analysis data by targetscan database (available at http://www.targetscan.org/) (JPEG 25 kb)
Rights and permissions
About this article
Cite this article
Cheng, Z., Qiu, S., Jiang, L. et al. MiR-320a is Downregulated in Patients with Myasthenia Gravis and Modulates Inflammatory Cytokines Production by Targeting Mitogen-activated Protein Kinase 1. J Clin Immunol 33, 567–576 (2013). https://doi.org/10.1007/s10875-012-9834-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-012-9834-5