Abstract
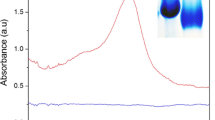
This study is the first report on the separation and reusability of ApoUnaG protein, indicating excellent fluorescence response with high affinity and specificity toward unconjugated bilirubin (UC-BR) molecules, from the UnaG-UC-BR complex structure. The fluorescence properties of the UnaG-UC-BR complex (holo-UnaG) are studied by addition of different metal ions to perform possible interactions with holo-UnaG through absorbance and emission spectra. After addition of metal ions, some changes with respect to the type of metal ions are observed in fluorescence intensity of the holo-UnaG. When compared to metal ions, an excellent quenching response is sighted in the presence of Cu2+ ions by binding with UC-BR in the UnaG-UC-BR complex structure. Obtained non-fluorescence holo-UnaG-Cu2+ complex mixture is passed through Ni–NTA agarose to remove the ingredients such as Cu2+, UC-BR and Cu2+-UC-BR coordination complex from holo-UnaG. From the obtained experiments, it is concluded that Cu2+ ion can be used as an agent for the recovery of ApoUnaG protein via binding with UC-BR molecules.

Recovery and Reusability of ApoUnaG Fluorescence Protein from the Unconjugated Bilirubin Complex Structure.








Similar content being viewed by others
References
Snapp EL (2009) Fluorescent proteins: a cell biologist's user guide. Trends Cell Biol 19(11):649–655. https://doi.org/10.1016/j.tcb.2009.08.002
Keskinates M, Yilmaz B, Ulusu Y, Bayrakci M (2018) Electrospinning of novel calixarene-functionalized PAN and PMMA nanofibers: comparison of fluorescent protein adsorption performance. Mater Chem Phys 205:522–529. https://doi.org/10.1016/j.matchemphys.2017.11.055
Adam V, Berardozzi R, Byrdin M, Bourgeois D (2014) Phototransformable fluorescent proteins: future challenges. Curr Opin Chem Biol 20:92–102. https://doi.org/10.1016/j.cbpa.2014.05.016
Enterina JR, Wu L, Campbell RE (2015) Emerging fluorescent protein technologies. Curr Opin Chem Biol 27:10–17. https://doi.org/10.1016/j.cbpa.2015.05.001
Kumagai A, Ando R, Miyatake H, Greimel P, Kobayashi T, Hirabayashi Y, Miyawaki A (2013) A BR-inducible fluorescent protein from eel muscle. Cell 153(7):1602–1611. https://doi.org/10.1016/j.cell.2013.05.038
Shitashima Y, Shimozawa T, Asahi T, Miyawaki A (2018) A dual-ligand-modulable fluorescent protein based on UnaG and calmodulin. Biochem Biophys Res Commun 496(3):872–879. https://doi.org/10.1016/j.bbrc.2018.01.134
Baydemir G, Andaç M, Bereli N, Say R, Denizli A (2007) Selective removal of bilirubin from human plasma with bilirubin-imprinted particles. Ind Eng Chem Res 46(9):2843–2852. https://doi.org/10.1021/ie0611249
Adhikari S, Joshi R, Gopinathan C (1998) Bilirubin as an anti precipitant against copper mediated denaturation of bovine serum albumin: formation of copper–bilirubin complex. Biochim Biophys Acta Gen Subj 1380(1):109–114. https://doi.org/10.1016/S0304-4165(97)00141-4
Goncharova I, Urbanová M (2009) Vibrational and electronic circular dichroism study of bile pigments: complexes of bilirubin and biliverdin with metals. Anal Biochem 392(1):28–36. https://doi.org/10.1016/j.ab.2009.05.040
Shapiro SM (2005) Definition of the clinical spectrum of kernicterus and bilirubin-induced neurologic dysfunction (BIND). J Perinatol 25(1):54. https://doi.org/10.1038/sj.jp.7211157
Viktorinova A (2017) Current insights on the role of iron and copper dyshomeostasis in the pathogenesis of bilirubin neurotoxicity. Life Sci 191:34. https://doi.org/10.1016/j.lfs.2017.10.013
Suzuki N (1966) On bilirubin-metal complex compounds in relation to black pigments of gallstones. Tohoku J Exp Med 90(2):195–205. https://doi.org/10.1620/tjem.90.195
He J, Lu XD, Zhou X, Yu NT, Chen Z (1995) Surface-enhanced Raman spectroscopy of BR-metal ion complexes. Biospectroscopy 1(2):157–162. https://doi.org/10.1002/bspy.350010210
Van Norman JD, Humans MM (1974) Bilirubin-metal ion complexes. Anal Chem 46(7):926–929. https://doi.org/10.1021/ac60343a033
Watson D (1961) Analytic methods for bilirubin in blood plasma. Clin Chem 7(6):603–625
Puppalwar PV, Goswami K, Dhok A (2012) Review on “evolution of methods of bilirubin estimation”. IOSR J Dent Med Sci 1(3):17–28. https://doi.org/10.9790/0853-0131728
Ulusu Y, Şentürk SB, Kuduğ H, Gökçe İ (2016) Expression, purification, and characterization of bovine chymosin enzyme using an inducible pTolT system. Prep Biochem Biotechnol 46(6):596–601. https://doi.org/10.1080/10826068.2015.1085399
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eczacioglu, N., Yilmaz, B., Ulusu, Y. et al. Recovery and Reusability of ApoUnaG Fluorescence Protein from the Unconjugated Bilirubin Complex Structure. J Fluoresc 30, 497–503 (2020). https://doi.org/10.1007/s10895-020-02519-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-020-02519-w