Abstract
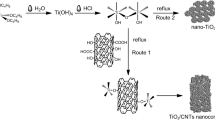
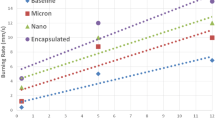
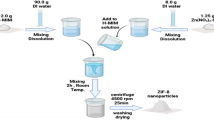
Ammonium perchlorate (APC) is the most common oxidizer for highly energetic systems. The initial decomposition of APC is an endothermic process. This behavior withstands high activation energy and could render high burning rate. We report on the sustainable fabrication of TiO2 nanoparticles; a novel catalyzing agent for APC. Mono-dispersed TiO2 particles of 10 nm particle size were fabricated using hydrothermal processing. XRD diffractogram demonstrated highly crystalline structure. The synthesized colloidal TiO2 particles were effectively integrated into APC particles via co-precipitation technique. The impact of TiO2 particles (1 wt%) on APC thermal behavior was investigated using DSC and TGA. APC demonstrated an initial endothermic decomposition (with absorption heat of 102.5 J/g) at 242 °C, and two subsequent exothermic decomposition stages at 297.8 and 452.8 °C respectively. TiO2 offered a decrease in APC early endothermic decomposition by 80%. The two main exothermic decomposition stages were merged into one single stage with an increase in total heat release by 18%. These novel features could inherit titanium oxide particles unique catalyzing ability for advanced highly energetic systems.














Similar content being viewed by others
Change history
31 March 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10904-020-01826-w
References
S. Jain et al., Size and shape of ammonium perchlorate and their influence on properties of composite propellant. Defence Science Journal 59, 294 (2009)
Kumari et al., Nano-ammonium perchlorate: preparation, characterization, and evaluation in composite propellant formulation. J. Energ. Mater. 31, 192–202 (2013)
G.P. Li et al., The preparation and properties of AP-based nano-limit growth energetic materials. Adv. Mater. Res. 924, 105–109 (2014)
M. Zou et al., Nano or micro? A mechanism on thermal decomposition of ammonium perchlorate catalyzed by cobalt oxalate. J. Hazard. Mater. 225, 124–130 (2012)
J.A. Conkling, C. Mocella, Chemistry of Pyrotechnics: Basic Principles and Theory (CRC Press, Boca Raton, 2010)
G.P. Sutton, O. Biblarz, Rocket Propulsion Elements (Wiley, New York, 2001)
N. Kubota, Propellants and Explosives: Thermochemical Aspects of Combustion (Wiley, Weinheim, 2015)
R.A. Chandru et al., Exceptional activity of mesoporous β-MnO2 in the catalytic thermal sensitization of ammonium perchlorate. J. Mater. Chem. 22, 6536–6538 (2012)
S. Chaturvedi, P.N. Dave, Nano-metal oxide: potential catalyst on thermal decomposition of ammonium perchlorate. J. Exp. Nanosci. 7, 205–231 (2012)
M.J. Turner, Rocket and spacecraft propulsion: principles, practice and new developments (Springer, Berlin, 2008)
N.R. Council, Advanced Energetic Materials (National Academies Press, Washington, DC, 2004)
S.G. Hosseini et al., Pure CuCr2O4 nanoparticles: synthesis, characterization and their morphological and size effects on the catalytic thermal decomposition of ammonium perchlorate. Solid State Sci. 37, 72–79 (2014)
S. Wang et al., An investigation into the fabrication and combustion performance of porous silicon nanoenergetic array chips. Nanotechnology 23, 435701 (2012)
P.R. Patil et al., Differential scanning calorimetric study of HTPB based composite propellants in presence of nano ferric oxide. Propellants Explos. Pyrotech. vol 31, 442–446 (2006)
S.-M. Shen et al., The thermal decomposition of ammonium perchlorate (AP) containing a burning-rate modifier. Thermochim. Acta 223, 135–143 (1993)
Nema et al., Mechanistic aspect of thermal decomposition and burn rate of binder and oxidiser of AP/HTPB composite propellants comprising HYASIS-CAT. Int. J. Plastics Technol. 8, 344–354 (2004)
L. Liu et al., Effects of nanometer Ni, Cu, Al and NiCu powders on the thermal decomposition of ammonium perchlorate. Propellants Explos. Pyrotech. 29, 34–38 (2004)
R. Rastogi et al., Burning rate catalysts for composite solid propellants. Combust. Flame 30, 117–124 (1977)
N. Li et al., Well-dispersed ultrafine Mn3O4 nanoparticles on graphene as a promising catalyst for the thermal decomposition of ammonium perchlorate. Carbon 54, 124–132, 2013
S.R. Chakravarthy et al., Mechanism of burning rate enhancement of composite solid propellants by ferric oxide. J. Propuls. Power 13, 471–480 (1997)
W. Pang et al., Effects of different nano-sized metal oxide catalysts on the properties of composite solid propellants. Combust. Sci. Technol. 188, 315–328 (2016)
P.W.M. Jacobs, H. Whitehead, Decomposition and combustion of ammonium perchlorate. Chem. Rev. 69, 551–590 (1969)
T. Daou et al., Hydrothermal synthesis of monodisperse magnetite nanoparticles. Chem. Mater. 18, 4399–4404 (2006)
X. Wang, Y. Li, Selected-control hydrothermal synthesis of α-and β-MnO2 single crystal nanowires. J. Am. Chem. Soc. 124, 2880–2881 (2002)
Cabanas, M. Poliakoff, The continuous hydrothermal synthesis of nano-particulate ferrites in near critical and supercritical water. J. Mater. Chem. 11, 1408–1416 (2001)
J.W. Lee et al., A facile and template-free hydrothermal synthesis of Mn3O4 nanorods on graphene sheets for supercapacitor electrodes with long cycle stability. Chem. Mater. 24, 1158–1164 (2012)
J. Li, Engineering nanoparticles in near-critical and supercritical water, PhD, University of Nottingham, Nottingham, 2008
M. Yoshimura, K. Byrappa, Hydrothermal processing of materials: past, present and future. J. Mater. Sci. 43, 2085–2103 (2008)
K. Byrappa, M. Yoshimura (eds.), Handbook of Hydrothermal Technology (William Andrew, Norwich, 2001)
P. Savage et al., Reactions at supercritical conditions: applications and fundamentals. Am. Inst. Chem. Eng. (AIChE) J. 41, 1723–1778 (1995)
K.S. Morley et al., Clean preparation on nanoparticulate metals in porous supports: a supercritical route. J. Chem. Mater. 12, 1898–1905 (2002)
H. Hobbs, Biocatalysis in ‘green solvents, PhD, Chemistry, University of Nottingham, Notttingham, 2006
J.A. Darr, M. Poliakoff, New directions in inorganic and metal-organic coordination chemistry in supercritical fluids. Chem. Rev. 99, 495–541 (1999)
T. Adschiri et al., Hydrothermal synthesis of metal oxide fine particles at supercritical conditions. Ind. Eng. Chem. Res. 39, 4901–4907 (2000)
T. Adschiri et al., Rapid and continuous hydrothermal synthesis of boehmite particles in subcritical and supercritical water. Am. Ceram. Soc. 75, 2615–2618 (1992)
S. Elbasuney, Dispersion characteristics of dry and colloidal nano-titania into epoxy resin. Powder Technol. 268, 158–164 (2014)
S. Elbasuney, Surface engineering of layered double hydroxide (LDH) nanoparticles for polymer flame retardancy. Powder Technol. 277, 63–73 (2015)
S. Elbasuney, Continuous hydrothermal synthesis of AlO(OH) nanorods as a clean flame retardant agent, Particuology, 22, 66–71 (2015)
S. Elbasuney, Sustainable steric stabilization of colloidal titania nanoparticles. Appl. Surf. Sci. 409, 438–447 (2017)
S. Elbasuney, Novel multi-component flame retardant system based on nanoscopic aluminium-trihydroxide (ATH). Powder Technol. 305, 538–545 (2017)
S. Elbasuney, Novel colloidal molybdenum hydrogen bronze (MHB) for instant detection and neutralization of hazardous peroxides. TrAC Trends Anal. Chem. 102, 272–279 (2018)
S. Elbasuney, Novel colloidal nanothermite particles (MnO2/Al) for advanced highly energetic systems. J. Inorg. Organomet. Polym. Mater. 28, 1793–1800 (2018)
S. Elbasuney et al., Infrared signature of novel super-thermite (Fe2O3/Mg) fluorocarbon nanocomposite for effective countermeasures of infrared seekers. J. Inorg. Organomet. Polym. Mater. 28, 1718–1727 (2018)
S. Elbasuney et al., Super-thermite (Al/Fe2O3) fluorocarbon nanocomposite with stimulated infrared thermal signature via extended primary combustion zones for effective countermeasures of infrared seekers. J. Inorgan. Organomet. Polym. Mater. 28, 2231–2240 (2018)
S. Elbasuney, H.E. Mostafa, “Synthesis and surface modification of nanophosphorous-based flame retardant agent by continuous flow hydrothermal synthesis. Particuology 22, 82–88 (2015)
S. Elbasuney, S.F. Mostafa, Continuous flow formulation and functionalization of magnesium di-hydroxide nanorods as a clean nano-fire extinguisher. Powder Technol. 278, 72–83 (2015)
T. Tillotson et al., Sol–gel processing of energetic materials. J. Non-Cryst. Solids 225, 358–363 (1998)
M. Mahinroosta, Catalytic effect of commercial nano-CuO and nano-Fe2O3 on thermal decomposition of ammonium perchlorate. J. Nanostruct. Chem. 3, 47 (2013)
V. Boldyrev, Thermal decomposition of ammonium perchlorate. Thermochim. Acta 443, 1–36 (2006)
L. Li et al., One step fabrication of Mn3O4/carbonated bacterial cellulose with excellent catalytic performance upon ammonium perchlorate decomposition. Mater. Res. Bull. 60, 802–807 (2014)
Acknowledgements
This work has been conducted at Nanotechnology Center, Military Technical College, Cairo, Egypt.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elbasuney, S., Yehia, M. Ammonium Perchlorate Encapsulated with TiO2 Nanocomposite for Catalyzed Combustion Reactions. J Inorg Organomet Polym 29, 1349–1357 (2019). https://doi.org/10.1007/s10904-019-01099-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-019-01099-y