Abstract
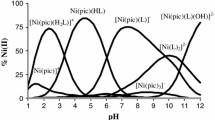
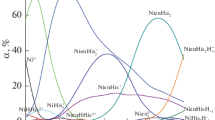
Solution equilibria in the binary systems of Ni(II)–amino acid {aspartic acid (H2asp), glutamic acid (H2glu), cysteine (H2cys) and histidine (Hhis)} and in the ternary systems of Ni(II)–dipicolinic acid (H2dipic)–amino acid {aspartic acid (H2asp), glutamic acid (H2glu), cysteine (H2cys) and histidine (Hhis)} have been studied pH-potentiometrically. The formation constants of the resulting mixed ligand complexes have been determined at 25 °C and ionic strength 1.0 mol·dm−3 NaCl. In the Ni(II)–H2asp system the data indicated the formation of the [NiHL]+, NiL and [NiL2]2− complexes. For the Ni(II)–H2glu system, the formation of the species NiL and [NiL2]2− are indicated. In the Ni(II)–H2cys system the complexes [NiHL]+, NiL and [NiHL2]− were detected, while in the Ni(II)–Hhis system the species [NiH2L]3+ [NiHL]2+, NiL+, [Ni(HL)2]2+, [NiHL2]+ and NiL2 were detected. In the ternary system Ni(II)–H2dipic–H2asp, the species Ni(dipic)(H2L), [Ni(dipic)(HL)]−, [Ni(dipic)(L)]2− and [Ni(dipic)(L)(OH)]3− were detected. In the Ni(II)–H2dipic–H2glu system, the species Ni(dipic)(H2L), [Ni(dipic)(HL)]−, [Ni(dipic)(L)]2− and [Ni(dipic)(L)(OH)]3− were detected. In the Ni(II)–H2dipic–H2cys system, the species Ni(dipic)(H2L), [Ni(dipic)(HL)]− and [Ni(dipic)(L)]2− were detected. Finally, in the Ni(II)–H2dipic–Hhis system, the species [Ni(dipic)(L)]− and [Ni(dipic)(L)(OH)]2− were detected. Species distribution diagrams as a function of pH are briefly discussed.








Similar content being viewed by others
References
Conato, C., Kozłowski, H., Swiatek-Kozłowska, J., Młynarz, P., Remelli, M., Silvestri, S.: Formation equilibria of nickel complexes with glycyl-histidyl-lysine and two synthetic analogues. J. Inorg. Biochem. 98, 153–160 (2004)
Lubes, V.: Vanadium(III) complexes with picolinic acid and dipicolinic acid in aqueous solution. J. Solution Chem. 34, 899–915 (2005)
Yasumatsu, N., Yoshikawa, Y., Adachi, Y., Sakurai, H.: Antidiabetic copper(II)–picolinate: impact of the first transition metal in the metallopicolinate complexes. Bioorg. Med. Chem. 15, 4917–4922 (2007)
Fukui, K., Fujisawa, Y., Ohya-Nishiguchi, H., Kamada, H., Sakurai, H.: In vivo coordination structural changes of a potent insulin-mimetic agent, bis(picolinato)oxovanadium(IV), studied by electron spin-echo envelope modulation spectroscopy. J. Inorg. Biochem. 77, 215–224 (1999)
Martell, A.E., Smith, M., Motekaitis, R.J.: NIST Critical Stability Constants of metal Complexes Database. US Department of Commerce, Gaithersburg, MD (1993)
Powell, K.J., Pettit, L.D.: IUPAC Stability Constants Database. Academic Software, Otley (1997)
Kolthoff, I.M., Stenger, V.A.: Volumetric Analysis, vol. II, p. 94. Interscience Publishers, New York (1947)
Jeffery, G.H., Bassett, J., Mendham, J., Denney, R.C.: Vogel’s Textbook of Quantitative Chemical Analysis, 5th edn, p. 327. Longman Scientific and Technical, UK (1989)
Gran, G.: Determination of the equivalence point in potentiometric titrations. Part II. Anal. 77, 661–671 (1952)
Biedermann, G., Sillén, L.G.: The hydrolysis of metal ions. IV. Liquid-junction potentials and constancy of activity factors in NaClO4–HClO4 ionic medium. Ark. Kemi 5, 425–440 (1953)
Brito, F., Araujo, M.L., Lubes, V., D’Ascoli, A., Mederos, A., Gili, P., Domínguez, S., Chinea, E., Hernández-Molina, R., Armas, M.T., Baran, E.J.: Emf(H) data analysis of weak metallic complexes using reduced formation functions. J. Coord. Chem. 58, 501–512 (2005)
Sillén, L.G., Warnqvist, B.: High-speed computers as a supplement to graphical methods. VI. A strategy for two-level Letagrop adjustment of common and “group” parameters. Features that avoid divergence. Ark. Kemi 31, 315–339 (1969)
Veliz, L., Martínez, J.D., Araujo, M.L., Brito, F., Lubes, G., Rodríguez, M., Lubes, V.: Estudio de la hidrólisis del ion Niquel(II) y de la formación de los complejos de Niquel(II) con los ácidos Picolínico y Dipicolínico en NaCl 1,0 mol·dm–3 a 25°C. Avances Quím. 6, 38 (2011)
Lema J. Estudio de formación de los complejos de níquel(II) con lo aminoácidos: ácido aspártico, ácido glutámico, histidina y cisteína mediante medidas de emf(H) en NaCl 1,0 M a 25 °C. Undergraduate thesis, Escuela de Química, Facultad de Ciencias, Universidad Central de Venezuela (2010)
Lderighi, L., Gans, P., Ienco, A., Peters, D., Sabatini, A., Vacca, A.: Hyperquad simulation and speciation (HySS): a utility program for the investigation of equilibria involving soluble and partially soluble species. Coord. Chem. Rev. 184, 311–318 (1999)
Centeno, M.M., Martínez, J.D., Araujo, M.L., Brito, F., Carpio, E., Hernández, L., Lubes, V.R.: Solution equilibria of ternary systems involving nickel(II) ion, picolinic acid, and the amino acids histidine, cysteine, aspartic and glutamic acids. J. Solution Chem. 43, 1011–1018 (2014)
Acknowledgments
The authors thank Dr. Antonio Barriola for valuable discussions. The authors are grateful for financing provided by the Decanato de Investigación y Desarrollo, from the Simón Bolívar University. Lino Hernández and Edgar Del Carpio thanks Simón Bolívar University for a scholarship.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lema, J., Ortega, J.A., Martínez, J.D. et al. Solution Equilibria and Stabilities of Binary and Ternary Systems of Nickel(II) Complexes with Dipicolinic Acid and the Amino Acids (Histidine, Cysteine, Aspartic and Glutamic Acids). J Solution Chem 44, 2144–2153 (2015). https://doi.org/10.1007/s10953-015-0404-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-015-0404-3