Abstract
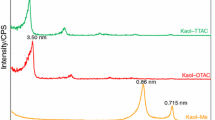
The thermal behavior and decomposition of kaolinite–potassium acetate intercalation complex was investigated through a combination of thermogravimetric analysis and infrared emission spectroscopy. Three main changes were observed at 48, 280, 323, and 460 °C which were attributed to (a) the loss of adsorbed water, (b) loss of the water coordinated to acetate ion in the layer of kaolinite, (c) loss of potassium acetate in the complex, and (d) water through dehydroxylation. It is proposed that the potassium acetate intercalation complex is stability except heating at above 300 °C. The infrared emission spectra clearly show the decomposition and dehydroxylation of the kaolinite intercalation complex when the temperature is raised. The dehydration of the intercalation complex is followed by the loss of intensity of the stretching vibration bands at region 3600–3200 cm−1. Dehydroxylation is followed by the decrease in intensity in the bands between 3695 and 3620 cm−1. Dehydration is completed by 400 °C and partial dehydroxylation by 650 °C. The inner hydroxyl group remained until around 700 °C.






Similar content being viewed by others
References
Franco F, Pérez-Maqueda LA, Pérez-Rodríguez JL. The effect of ultrasound on the particle size and structural disorder of a well-ordered kaolinite. J Colloid Interface Sci. 2004;274:107–17.
Zhang X, Fan D, Xu Z. Rapid preparation for kaolinite/dimethyl sulphoxide intercalated complex induced by microwave. J Tongji Univ (Natural Science). 2005;33:1646–50.
Franco F, Cecila JA, Pérez-Maqueda LA, Pérez-Rodríguez JL, Gomes CSF. Particle-size reduction of dickite by ultrasound treatments: effect on the structure, shape and particle-size distribution. Appl Clay Sci. 2007;35:119–27.
Gardolinski JE, Peralta-Zamora P, Wypych F. Preparation and characterization of a kaolinite-1-methyl-2-pyrrolidone intercalation compound. J Colloid Interface Sci. 1999;211:137–41.
Frost RL, Kristof J, Kloprogge JT, Horvath E. Rehydration of potassium acetate-intercalated kaolinite at 298 K. Langmuir. 2000;16:5402–8.
Frost RL, Kristof J, Horvath E, Kloprogge JT. Deintercalation of dimethylsulphoxide intercalated kaolinites—a DTA/TGA and Raman spectroscopic study. Thermochim Acta. 1999;327:155–66.
Frost RL, Kristof J, Horvath E, Kloprogge JT. Modification of the kaolinite hydroxyl surfaces through the application of pressure and temperature, Part III. J Colloid Interface Sci. 1999;214:380–8.
Horváth E, Kristóf J, Frost RL. Vibrational spectroscopy of intercalated kaolinites. Part I. Appl Spectrosc Rev. 2010;45:130–47.
Gardolinski JEFC, Lagaly G. Grafted organic derivatives of kaolinite: I. Synthesis, chemical and rheological characterization. Clay Miner. 2005;40:537–46.
Zhao S, Wang T, Xu H, Guo Y, Preparation of kaolinite/p-toluene sulfonic acid intercalation compound by melting method. Feijinshukuang 2009;32:37–9.
Frost RL, Kristof J, Paroz GN, Tran TH, Kloprogge JT. The role of water in the intercalation of kaolinite with potassium acetate. J Colloid Interface Sci. 1998;204:227–36.
Koji W. Lattice expansion of kaolin minerals by treatment with potassium acetate. Am Miner. 1961;46:78–91.
Anakli D, Çetinkaya S. Preparation of poly(2-ethyl aniline)/kaolinite composite materials and investigation of their properties. Curr Appl Phys. 2010;10:401–6.
Janek M, Emmerich K, Heissler S, Nuesch R. Thermally induced grafting reactions of ethylene glycol and glycerol intercalates of kaolinite. Chem Mater. 2007;19:684–93.
Letaief S, Elbokl TA, Detellier C. Reactivity of ionic liquids with kaolinite: melt intersalation of ethyl pyridinium chloride in an urea-kaolinite pre-intercalate. J Colloid Interface Sci. 2006;302:254–8.
Letaief S, Detellier C. Interlayer grafting of glycidol (2, 3-epoxy-1-propanol) on kaolinite. Can J Chem. 2008;86:1–6.
Frost RL, Bahfenne S, Graham J. Infrared and infrared emission spectroscopic study of selected magnesium carbonate minerals containing ferric iron—Implications for the geosequestration of greenhouse gases. Spectrochim Acta A Mol Biomol Spectrosc. 2008;71:1610–6.
Frost RL, Kloprogge JT. Infrared emission spectroscopic study of brucite. Spectrochim Acta A Mol Biomol Spectrosc. 1999;55:2195–205.
Frost RL, Weier ML. Thermal treatment of weddellite—a Raman and infrared emission spectroscopic study. Thermochim Acta. 2003;406:221–32.
Frost RL, Cash GA, Kloprogge JT. ‘Rocky Mountain leather’, sepiolite and attapulgite—an infrared emission spectroscopic study. Vib Spectrosc. 1998;16:173–84.
Frost RL, Wain D. A thermogravimetric and infrared emission spectroscopic study of alunite. J Therm Anal Calorim. 2008;91:267–74.
Arizaga GGC, Gardolinski JEFDC, Schreiner WH, Wypych F. Intercalation of an oxalatooxoniobate complex into layered double hydroxide and layered zinc hydroxide nitrate. J Colloid Interface Sci. 2009;330:352–8.
Zhang B, Li Y, Pan X, Jia X, Wang X. Intercalation of acrylic acid and sodium acrylate into kaolinite and their in situ polymerization. J Phys Chem Solids. 2007;68:135–42.
Franco F, Ruiz Cruz MD. Factors influencing the intercalation degree (‘reactivity’) of kaolin minerals with potassium acetate, formamide, dimethylsulphoxide and hydrazine. Clay Miner. 2004;39:193–205.
Frost RL, Kristof J, Horvath E, Kloprogge JT. Modification of kaolinite surfaces through intercalation with potassium acetate, II. J Colloid Interface Sci. 1999;214:109–17.
Cheng H, Liu Q, Yang J, Zhang Q, Frost RL. Thermal behavior and decomposition of kaolinite-potassium acetate intercalation composite. Thermochim Acta. 2010;503–504:16–20.
Frost RL, Kristof J, Horvath E, Kloprogge JT. Effect of water on the formamide-intercalation of kaolinite. Spectrochim Acta A Mol Biomol Spectrosc. 2000;56:1711–29.
Frost RL, Kristof J, Mako E, Horvath E. A DRIFT spectroscopic study of potassium acetate intercalated mechanochemically activated kaolinite. Spectrochim Acta A Mol Biomol Spectrosc. 2003;59:1183–94.
Frost RL, Kristof J, Paroz GN, Kloprogge JT. Role of water in the intercalation of kaolinite with hydrazine. J Colloid Interface Sci. 1998;208:216–25.
Franco F, Ruiz Cruz M. Thermal behaviour of dickite-dimethylsulfoxide intercalation complex. J Therm Anal Calorim. 2003;73:151–65.
Gorb LG, Aksenenko EV, Adams JW, Larson SL, Weiss CA, Leszczynska D, Leszczynski J. Computational design of clay minerals: hydration of Mg-exchange cation located in ditrigonal cavity. J Mol Struct. 1998;425:129–35.
Hess AC, Saunders VR. Periodic ab initio Hartree-Fock calculations of the low-symmetry mineral kaolinite. J Phys Chem. 1992;96:4367–74.
Kristof J, Frost RL, Felinger A, Mink J. FTIR spectroscopic study of intercalated kaolinite. J Mol Struct. 1997;410–411:119–22.
Acknowledgements
The authors gratefully acknowledge the financial support provided by the National “863” project of China (2008AA06Z109) and infra-structure support of the Queensland University of Technology Chemistry Discipline, Faculty of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheng, H., Yang, J., Frost, R.L. et al. Thermal analysis and Infrared emission spectroscopic study of kaolinite–potassium acetate intercalate complex. J Therm Anal Calorim 103, 507–513 (2011). https://doi.org/10.1007/s10973-010-0917-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-010-0917-3