Abstract
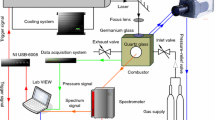
Amorphous boron is usually employed as the most important fuel of boron-based fuel-rich propellants, and NH4ClO4 (AP), cyclotetramethylenetetranitramine (HMX), KClO4 and KNO3 are the solid oxidizers of most used in solid propellants. The mixtures of boron and different solid oxidizers with mass ratio 1:1 were prepared in this paper, and the effect of these oxidizers on the thermal oxidation and combustion performance of amorphous boron was studied by simultaneous thermogravimetry–differential scanning calorimeter–Fourier transform infrared spectroscopy and CO2 laser ignition experiments. The experimental results show that the main reactions during the heating process of B/AP and B/HMX samples are the decomposition of oxidizers, and the decomposition process of oxidizers rather than the decomposition temperature is affected by amorphous boron; boron could react with KClO4 and KNO3 violently with the release of large amounts of heat, and then both of the oxidizers, especially KClO4, have positive effect on the oxidation and combustion performance of amorphous boron.









Similar content being viewed by others
References
Kuo KK, Summerfield M. Fundamentals of solid-propellant combustion. New York: American Institute of Aeronautics and Astronautics; 1984.
Gany A, Timnat YM. Advantages and drawbacks of boron-fueled propulsion. Acta Astronaut. 1993;29(3):181–7. doi:10.1016/0094-5765(93)90047-Z.
Besser HL, Strecker R. Overview of boron ducted rocket development. Int J Energ Mater Chem Propuls. 1993;2(1–6):133–78. doi:10.1615/IntJEnergeticMaterialsChemProp.v2.i1-6.60.
Buchner E, Langel G. Elementary boron as a propellant component in ducted rockets: thermodynamic study. Z Flugwissensch. 1976;24(5):275–8.
Fry RS. A century of ramjet propulsion technology evolution. J Propuls Power. 2004;20(1):27–58. doi:10.2514/1.9178.
Pang WQ, Fan XZ, Zhang W, Xu HX, Li JZ, Li YH, et al. Application of amorphous boron granulated with hydroxyl-terminated polybutadiene in fuel-rich solid propellant. Propellant Explos Pyrotech. 2011;36(4):360–6. doi:10.1002/prep.200900112.
Liu L, He G, Wang Y. Thermal reaction characteristics of the boron used in the fuel-rich propellant. J Therm Anal Calorim. 2013;114(3):1057–68.
Hussmann B, Pfitzner M. Extended combustion model for single boron particles—part I: theory. Combust Flame. 2010;157(4):803–21. doi:10.1016/j.combustflame.2009.12.010.
Hussmann B, Pfitzner M. Extended combustion model for single boron particles—part II: validation. Combust Flame. 2010;157(4):822–33. doi:10.1016/j.combustflame.2009.12.009.
Foelsche RO, Burton RL, Krier H. Boron particle ignition and combustion at 30-150 ATM. Combust Flame. 1999;117(1–2):32–58. doi:10.1016/S0010-2180(98)00080-7.
Yeh CL, Kuo KK. Ignition and combustion of boron particles. Prog Energy Combust. 1996;22(6):511–41. doi:10.1016/S0360-1285(96)00012-3.
Liu L, He G, Wang Y. Effect of oxidizer on the combustion performance of boron-based fuel-rich propellant. J Propuls Power. 2014;30(2):285–9.
Kubota N. Energetics of HMX-based composite modified double-base propellant combustion. J Propuls Power. 1999;15(6):759–62.
Muthiah R, Varghese T, Rao SS, Ninan K, Krishnamurthy V. Realisation of an eco-friendly solid propellant based on HTPB-HMX-AP system for launch vehicle applications. Int J Energ Mater Chem Propul. 1997;4(1–6):134–9.
Sivan J, Haas Y. Spectroscopic characterization of B/KNO3 diode-laser induced combustion. J Phys Chem A. 2013;117(46):11808–14.
Liu L, He G, Wang Y, Liu P. Effect of catocene on the thermal decomposition of ammonium perchlorate and octogen. J Therm Anal Calorim. 2014;117(2):621–8.
Boldyrev V. Thermal decomposition of ammonium perchlorate. Thermochim Acta. 2006;443(1):1–36.
Jacobs P, Pearson G. Mechanism of the decomposition of ammonium perchlorate. Combust Flame. 1969;13(4):419–30.
Lee J-S, Hsu C-K, Jaw K-S. The thermal properties of KClO 4 with different particle size. Thermochim Acta. 2001;367:381–5.
Miyata K, editor. Combustion of boron-pyrotechnics. AIAA/ASME/SAE/ASEE Joint Propulsion Conference and Exhibit, 37th, Salt Lake City, UT; 2001.
El-Awad A. Catalytic effect of some chromites on the thermal decomposition of KClO4. Mechanistic and non-isothermal kinetic studies. J Therm Anal Calorim. 2000;61(1):197–208.
Spalding MJ, Krier H, Burton R. Boron suboxides measured during ignition and combustion of boron in shocked Ar/F/O 2 and Ar/N 2/O 2 mixtures. Combust Flame. 2000;120(1):200–10.
Ao W, Yang W, Wang Y, Zhou J, Liu J, Cen K. Ignition and combustion of boron particles at one to ten standard atmosphere. J Propuls Power. 2014;30(3):760–4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Pj., Liu, Ll. & He, Gq. Effect of solid oxidizers on the thermal oxidation and combustion performance of amorphous boron. J Therm Anal Calorim 124, 1587–1593 (2016). https://doi.org/10.1007/s10973-016-5252-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5252-x