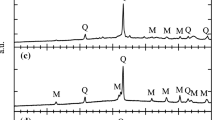
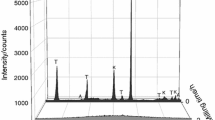
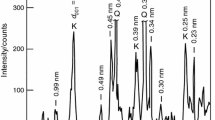
Abstract
The kinetics of the illite dehydroxylation is investigated using data from non-isothermal thermodilatometric experiments. An illitic clay with more than 70 % of illite was thermally treated under several heating rates from 1 to 15 °C min−1 in the dehydroxylation region. Model-free and nonlinear model-fitting methods were used to analyze the data, and their results are discussed. The dehydroxylation of the trans-vacant and cis-vacant illite layers can be described by the Avrami–Erofeev model with the exponent values 1.010 and 2.1, respectively, and the apparent activation energies of 119 and 184 kJ mol−1, respectively. The obtained kinetic parameters were used for the calculation of an optimal heating regime in order to achieve a constant thermal expansion rate in the dehydroxylation region for an illite-based ceramic body.






Similar content being viewed by others
References
Ferrari S, Gualtieri A. The use of illitic clays in the production of stoneware tile ceramics. Appl Clay Sci. 2006;32:73–81.
Gualtieri AF, Ferrari S. Kinetics of illite dehydroxylation. Phys Chem Miner. 2006;33:490–501.
Grim G, Bray R, Bradley W. The mica in argillaceous sediments. Am Mineral. 1937;22:813–29.
Środoń J, Illite ED. Rev Mineral Geochem. 1984;13:495–544.
Gualtieri AF, Ferrari S, Leoni M, Grathoff G, Hugo R, Mouath S, Paglia G, Billinge S. Structural characterization of the clay mineral illite-1M. J Appl Crystallogr. 2008;41:402–15.
Venturelli C, Paganelli M. Sintering behaviour of clays for the production of ceramics. Process Eng. 2007;84:5–8.
Bennour A, Mahmoudi S, Srasra E, Hatira N, Boussen S, Ouaja M, Zargouni F. Identification and traditional ceramic application of clays from the Chouamekh region in south-eastern Tunisia. Appl Clay Sci. 2015;118:212–20.
Antal D, Húlan T, Trník A, Štubňa I, Ondruška J. The influence of texture and firing on thermal and elastic properties of illite-based ceramics. Adv Mater Res. 2015;1126:53–8.
Gemmi M, Merlini M, Pavese A, Curetti N. Thermal expansion and dehydroxylation of phengite micas. Phys Chem Miner. 2008;35:367–79.
Guggenheim S, Chang YH, van Groos KA. Muscovite dehydroxylation: high-temperature studies. Am Mineral. 1987;72:537–50.
Drebushchak VA, Mylnikova LN, Molodin VI. Thermogravimetric investigation of ancient ceramics: metrological analysis of sampling. J Therm Anal Calorim. 2007;90:73–9.
Ptáček P, Frajkorová F, Šoukal F, Opravil T. Kinetics and mechanism of three stages of thermal transformation of kaolinite to metakaolinite. Powder Technol. 2014;264:439–45.
Ptáček P, Kubátová D, Havlica J, Brandštetr J, Šoukal F, Opravil T. The non-isothermal kinetic analysis of the thermal decomposition of kaolinite by thermogravimetric analysis. Powder Technol. 2010;204:222–7.
Ptáček P, Šoukal F, Opravil T, Havlica J, Brandštetr J. The kinetic analysis of the thermal decomposition of kaolinite by DTG technique. Powder Technol. 2011;208:20–5.
Ptáček P, Opravil T, František Š, Wasserbauer J, Másilko J, Baráček J. The influence of structure order on the kinetics of dehydroxylation of kaolinite. J Eur Ceram Soc. 2013;33:2793–9.
Levy JH, Hurst HJ. Kinetics of dehydroxylation, in nitrogen and water wapour, of kaolinite and smectite from Australian Tertiary oil shales. Fuel. 1993;72:873–7.
Ortega A, Manuel M, Gotor FJ. The multistep nature of the kaolinite dehydroxylation: kinetics and mechanism. J Am Ceram Soc. 2010;93:197–203.
Mitra N, Maitra S. Effect of surface-area on the dehydroxylation kinetics of kaolinite mineral. J Indian Chem Soc. 1993;70:629–32.
Hirono T, Tanikawa W. Implications of the thermal properties and kinetic parameters of dehydroxylation of mica minerals for fault weakening, friction heating, and earthquake energetics. Earth Planet Sci Lett. 2011;307:161–72.
Drits VA, Lindgreen H, Salyn AL, Ylagan R, McCarty DK. Semiquantitative determination of trans-vacant and cis-vacant 2:1 layers in illites and illite-smectites by thermal analysis and X-ray diffraction. Am Mineral. 1998;83:1188–98.
Vyazovkin S, Chrissafis K, Di Lorenzo ML, Koga N, Pijolat M, Roduit B, Sbirrazzuoli N, Suñol JJ. ICTAC kinetics committee recommendations for collecting experimental thermal analysis data for kinetic computations. Thermochim Acta. 2014;590:1–23.
Sánchez-Rodríguez D, Eloussifi H, Farjas J, Roura P, Dammak M. Thermal gradients in thermal analysis experiments: criterions to prevent inaccuracies when determining sample temperature and kinetics parameters. Thermochim Acta. 2014;589:37–46.
Podoba R, Podobník Ľ, Trník A. Upgrading of TGA/DTA analyzer derivatograph. Épitőanyag. 2012;64:28–9.
Vyazovkin S, Burnham AK, José CM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520:1–19.
Sbirrazzuoli N. Determination of pre-exponential factors and of the mathematical functions f(α) or G(α) that describe the reaction mechanism in a model-free way. Thermochim Acta. 2013;564:59–69.
Vyazovkin S. Computational aspects of kinetic analysis. Part C. The ICTAC Kinetics Project—the light at the end of the tunnel? Thermochim Acta. 2000;355:155–63.
Arhangelskii I, Dunaev A, Makarenko I, Tikhonov N, Belyaev S, Tarasov A. Non-isothermal kinetic methods: Workbook and laboratory manual. Berlin: Edition Open Access; 2013.
Friedman HL. Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to a phenolic plastic. J Polym Sci. 1964; Part C:183–95.
Perejón A, Sánchez-Jiménez PE, Criado JM, Pérez-Maqueda LA. Kinetic analysis of complex solid-state reactions. A new deconvolution procedure. J Phys Chem B. 2011;115:1780–91.
Málek J. The applicability of Johnson–Mehl–Avrami model in the thermal analysis of the crystallization kinetics of glasses. Thermochim Acta. 1995;267:61–73.
Kingery WD. Factors affecting thermal stress resistance of ceramic materials. J Am Ceram Soc. 1955;38:3–15.
Acknowledgements
This work was supported by the grant VEGA 1/0162/15 from the Ministry of Education of the Slovak Republic, grant VII/5/2016 from Constantine the Philosopher University, and by the Czech Science Foundation under the project No. P105/12/G059. The authors are indebted to J. Biber from Inter-ILI Engineering Office (Hungary) for a supply of illite.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Húlan, T., Trník, A. & Medveď, I. Kinetics of thermal expansion of illite-based ceramics in the dehydroxylation region during heating. J Therm Anal Calorim 127, 291–298 (2017). https://doi.org/10.1007/s10973-016-5873-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5873-0