Abstract
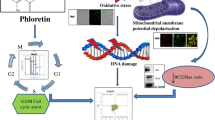
Phosphatidylinositol 3-kinase (PI3-K)/PTEN/Akt signaling is over activated in various tumors including colon cancer. Activation of this pathway regulates multiple biological processes such as apoptosis, metabolism, cell proliferation, and cell growth that underlie the biology of a cancer cell. In the present study, the chemopreventive effects have been observed of Diclofenac, a preferential COX-2 inhibitory non-steroidal anti-inflammatory drugs, and Curcumin, a natural anti-inflammatory agent, in the early stage of colorectal carcinogenesis induced by 1,2-dimethylhydrazine dihydrochloride in rats. The tumor-promoting role of PI3-K/Akt/PTEN signal transduction pathway and its association with anti-apoptotic family of proteins are also observed. Both Diclofenac and Curcumin downregulated the PI3-K and Akt expression while promoting the apoptotic mechanism. Diclofenac and Curcumin administration significantly increased the expression of pro-apoptotic Bcl-2 family members (Bad and Bax) while decreasing the anti-apoptotic Bcl-2 protein. An up-regulation of cysteine protease family apoptosis executioner, such as caspase-3 and -9, is seen. Diclofenac and Curcumin inhibited the Bcl-2 protein by directly interacting at the active site by multiple hydrogen bonding, as also evident by negative glide score of Bcl-2. These drugs stimulated apoptosis by increasing reactive oxygen species (ROS) generation and simultaneously decreasing the mitochondrial membrane potential (ΔΨ M). Diclofenac and Curcumin showed anti-neoplastic effects by downregulating PI3-K/Akt/PTEN pathway, inducing apoptosis, increasing ROS generation, and decreasing ΔΨ M. The anti-neoplastic and apoptotic effects were found enhanced when both Diclofenac and Curcumin were administered together, rather than individually.









Similar content being viewed by others
References
Jacobson MD, Weil M, Raff MC (1997) Programmed cell death in animal development. Cell 88:347–354
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116:205–219
Ferreira CG, Epping M, Kruyt FA, Giaccone G (2002) Apoptosis: target of cancer therapy. Clin Cancer Res 8:2024–2034
Engelman JA (2009) Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer 9:550–562
Manning BD, Cantley LC (2007) AKT/PKB signaling: navigating downstream. Cell 129:1261–1274
Spencer JP, Rice-Evans C, Williams RJ (2003) Modulation of pro-survival Akt/protein kinase B and ERK1/2 signaling cascades by quercetin and its in vivo metabolites underlie their action on neuronal viability. J Biol Chem 2:34783–34793
Vivanco I, Sawyers CL (2002) The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev Cancer 2:489–501
Parsons R (2004) Human cancer, PTEN and the PI-3 kinase pathway. Semin Cell Dev Biol 15:171–176
Franke TF, Hornik CP, Segev L et al (2003) PI3K/Akt and apoptosis: size matters. Oncogene 22:8983–8998
Pugazhenthi S, Nesterova A, Sable C et al (2000) Akt/protein kinase B up-regulates Bcl-2 expression through cAMP-response element-binding protein. J Biol Chem 275:10761–10766
Surh YJ (2003) Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer 3:768–780
Rarnsewak RS, DeWitt DL, Nair MG (2003) Cytotoxicity, antioxidant and anti-inflammatory activities of Curcumins I–III from Curcuma longa. Phytomedicine 7:303–308
Goel A, Kunnumakkara AB, Aggarwal BB (2008) Curcumin as “Curecumin”: from kitchen to clinic. Biochem Pharmacol 75:787–809
Gou M, Men K, Shi HS et al (2011) Curcumin-loaded biodegradable polymeric micelles for colon cancer therapy in vitro and in vivo. Nanoscale 3:1558–1567
Torrance CJ, Jackson PE, Montgomery E et al (2000) Combinatorial chemoprevention of intestinal neoplasia. Nat Med 6:1024–1028
Saini MK, Vaish V, Sanyal SN (2013) Role of cytokines and Jak3/Stat3 signaling in the 1,2-dimethylhydrazine dihydrochloride-induced rat model of colon carcinogenesis: early target in the anticancer strategy. Eur J Cancer Prev 23:215–228
Piplani H, Rana C, Vaish V, Sanyal SN (2013) Dolastatin, along with Celecoxib, stimulates apoptosis by a mechanism involving oxidative stress, membrane potential change and PI3-K/AKT pathway down regulation. Biochim Biophys Acta Gen Subj 1830:5142–5156
Rana C, Vaish V, Piplani H, Nehru B, Sanyal SN (2012) Nuclear Factor-κB is expressed in early colon cancer and its down-regulation by Curcumin and Diclofenac is associated with the suppression of proliferation and the induction of apoptosis. Biomed Prev Nutr 2:228–238
Abdelwahab SI, Koko WS, Taha MME et al (2012) In vitro and in vivo anti-inflammatory activities of columbin through the inhibition of cycloxygenase-2 and nitric oxide but not the suppression of NF-κB translocation. Eur J Pharmacol 678:61–70
Winter CA, Risley EA, Nuss GW (1962) Carrageenan-induced oedema in the hind paw of rat as an assay for anti-inflammatory activity. Proc Soc Exp Biol Ther 111:544–547
Piplani H, Rana C, Vaish V, Sanyal SN (2013) Up-regulation of p53 and mitochondrial signalling pathway in apoptosis by a combination of COX-2 inhibitor, Celecoxib and Dolastatin 15, a marine mollusk linear peptide in experimental colon carcinogenesis. Mol Carcinog 52:845–858
Vaish V, Piplani H, Rana C, Sanyal SN (2013) Angiostatic Properties of Sulindac and Celecoxib in the experimentally induced inflammatory colorectal cancer. Cell Biochem Biophys 66:205–227
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Mouille B, Robert V, Blacheir F (2012) Adaptative increase of ornithine production and decrease of ammonia metabolism in rat colonocytes after hyperproteic diet ingestion. J Physiol Gastrointest Liver Physiol 278:G344–G351
Piccagli L, Fabbri E, Borgatti M (2008) Docking of molecules identified in bioactive medicinal plants extracts into the p50 NF-kappaB transcription factor: correlation with inhibition of NF-kappaB/DNA interactions and inhibitory effects on IL-8 gene expression. BMC Struct Biol 8:38
Johnson LV, Walsh ML, Chen LB (1980) Localization of mitochondria in living cells with rhodamine 123. Proc Natl Acad Sci USA 177:990–994
LeBel CP, Ischiropoulos H, Bondy SC (1992) Evaluation of the probe 2′,7′-dichlorofluorescein as an indicator of reactive oxygen species formation and oxidative stress. Chem Res Toxicol 5:227–231
Gupta SC, Kim JH, Prasad S (2010) Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev 29:405–434
Aggarwal BB, Harikumar KB (2009) Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol 41:40–59
Park J, Conteas CN (2010) Anti-carcinogenic properties of curcumin on colorectal cancer. World J Gastrointest Oncol 22:169–176
Cheng AL, Hsu CH, Lin JK et al (2001) Phase I clinical trial of curcumin, a chemopreventive agent, in patients with high-risk or pre-malignant lesions. Anticancer Res 21:2895–2900
Sharma RA, McLelland HR, Hill KA (2001) Pharmacodynamic and pharmacokinetic study of oral Curcuma extract in patients with colorectal cancer. Clin Cancer Res 7:1894–1900
Wakabayashi K (2000) NSAIDs as cancer preventive agents. Asian Pac J Cancer Prev 1:97–113
Sun SY, Hail NJ, Lotan R (2004) Apoptosis as a novel target for cancer chemoprevention. J Natl Cancer Inst 5:662–672
Ahmad A, Biersack B, Li Y et al (2013) Deregulation of PI3K/Akt/mTOR signaling pathways by isoflavones and its implication in cancer treatment. Anticancer Agents Med Chem 13:1014–1024
Benistant C, Chapuis H, Roche S (2000) A specific function for phosphatidylinositol 3-kinase alpha (p85alpha–p110alpha) in cell survival and for phosphatidylinositol 3-kinase beta (p85alphap110beta) in de novo DNA synthesis of human colon carcinoma cells. Oncogene 19:5083–5090
Madhunapantula SV, Mosca PJ, Robertson GP (2011) The Akt signaling pathway: an emerging therapeutic target in malignant melanoma. Cancer Biol Ther 12:1032–1049
Bayascas JR (2010) PDK1: the major transducer of PI 3-kinase actions. Curr Top Microbiol 346:9–29
Raimondi MF (2011) Targeting PDK1 in cancer. Curr Med Chem 18:2763–2769
Yamada KM, Araki M (2001) Tumor suppressor PTEN, Modulator of cell signalling, growth, migration, and apoptosis. J Cell Sci 114:2375–2382
Van Kanegan MJ, Adams DJ, Wadzinski BE et al (2005) Distinct protein phosphatase 2A heterotrimers modulate growth factor signaling to extracellular signal-regulated kinases and AKT. J Biol Chem 280:36029–36036
Brooijmans N, Kuntz ID (2003) Molecular recognition and docking algorithms. Annu Rev Biophys Biomol Struct 32:335–373
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407:770–776
Liu Y, Borchert GL, Surazynski A et al (2006) Proline oxidase activates both intrinsic and extrinsic pathways for apoptosis: the role of ROS/superoxides, NFAT and MEK/ERK signalling. Oncogene 25:5640–5647
Herman-Antosiewicz A, Powolny AA, Singh SV (2007) Molecular targets of cancer chemoprevention by garlic-derived organosulfides. Acta Pharmacol Sin 28:1355–1364
Adams JM, Cory S (2007) The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene 26:1324–1337
Fletcher JI, Meusburger S, Hawkins CJ et al (2008) Apoptosis is triggered when prosurvival Bcl-2 proteins cannot restrain Bax. Proc Natl Acad Sci USA 2008(105):18081–18087
Maddika S, Ande SR, Panigrahi S et al (2007) Cell survival, cell death and cell cycle pathways are interconnected: implications for cancer therapy. Drug Resist Update 10:13–29
Asnaghi L, Calastretti A, Bevilacqua A (2004) Bcl-2 phosphorylation and apoptosis activated by damaged microtubules require mTOR and are regulated by Akt. Oncogene 23:5781–5791
Rathmell JC, Fox CJ, Plas DR et al (2003) Akt-directed glucose metabolism can prevent Bax conformation change and promote growth factor-independent survival. Mol Cell Biol 23:7315–7328
Kandasamy K, Srinivasula SM, Alnemri ES et al (2003) Involvement of proapoptotic molecules Bax and Bak in tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced mitochondrial disruption and apoptosis: differential regulation of cytochrome c and Smac/DIABLO release. Cancer Res 63:1712–1721
Kroemer G, Galluzz L, Brenner C (2007) Mitochondrial membrane permeabilization in cell death. Physiol Rev 87:99–163
Zou H, Li Y, Liu X et al (1997) An APAF-1.cytochrome c multimeric complex is a functional apoptosome that activates procaspase-9. J Biol Chem 23:11549–11597
Varghese J, Khandre NS, Sarin A (2008) Caspase-3 activation is an early event and initiates apoptotic damage in a human leukemia cell line. Apoptosis 8:363–370
Torkin R, Lavoi JF, Kaplan DR et al (2005) Induction of caspase-dependent, p53-mediated apoptosis by apigenin in human neuroblastoma. Mol Cancer Ther 4:1–11
Circu ML, Aw TW (2010) Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med 8:749–762
Miki H, Funato Y (2012) Regulation of intracellular signalling through cysteine oxidation by reactive oxygen species. J Biochem 151:255–261
Ying C, Geert A, Martens SA et al (2007) Increased oxygen radical formation and mitochondrial dysfunction mediate beta cell apoptosis under conditions of AMP-activated protein kinase stimulation. Free Radic Biol Med 42:64–78
Acknowledgments
Financial assistance from the Department of Atomic Energy (DAE) and Board of Research in Nuclear Physics (BRNS), Govt. of India (2009/37/45/BRNS) is gratefully acknowledged. We also acknowledge the contributions of Dr. Ravi Kumar Muttineni, Application Specialist, Schrodinger, India for helping us in performing the docking studies.
Conflict of interest
Authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rana, C., Piplani, H., Vaish, V. et al. Downregulation of PI3-K/Akt/PTEN pathway and activation of mitochondrial intrinsic apoptosis by Diclofenac and Curcumin in colon cancer. Mol Cell Biochem 402, 225–241 (2015). https://doi.org/10.1007/s11010-015-2330-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2330-5