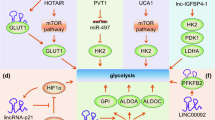
Abstract
Metabolism reprogramming is one of the hallmarks of cancer cells, especially glucose metabolism, to promote their proliferation, metastasis and drug resistance. Cancer cells tend to depend on glycolysis for glucose utilization rather than oxidative phosphorylation, which is called the Warburg effect. Genome instability of oncogenes and tumor-inhibiting factors is the culprits for this anomalous glycolytic fueling, which results in dysregulating metabolism-related enzymes and metabolic signaling pathways. It has been extensively demonstrated that protein-coding genes are involved in this process; therefore, glycolysis-targeted therapy has been widely used in anti-tumor combined therapy via small molecular inhibitors of key enzymes and regulatory molecular. The long non-coding RNA, which is a large class of regulatory RNA with longer than 200 nucleotides, is the novel and significant regulator of various biological processes, including metabolic reprogramming. RNA interference and synthetic antisense oligonucleotide for RNA reduction have developed rapidly these years, which presents potent anti-tumor effects both in vitro and in vivo. However, lncRNA-based glycolysis-targeted cancer therapy, as the highly specific and less toxic approach, is still under the preclinical phase. In this review, we highlight the role of lncRNA in glucose metabolism and dissect the feasibility and limitations of this clinical development, which may provide potential targets for cancer therapy.


Similar content being viewed by others
Data availability
The data of clinical trials was from https://clinicaltrials.gov/.
Abbreviations
- AGPG:
-
Actin gamma 1 pseudogene
- AML:
-
Acute myeloid leukemia
- AMPK:
-
AMP-activated protein kinase
- ANXA2P2:
-
Annexin A2 Pseudogene 2
- ASO:
-
Antisense oligonucleotide
- CHC:
-
α-Cyano-4-hydroxycinnamic acid
- CRC:
-
Colorectal cancer
- ceRNA:
-
Competing endogenous RNA
- CR:
-
Complete remission
- DANCE:
-
Differentiation antagonizing non-coding RNA
- DNMT1:
-
DNA methyltransferase 1
- E2F1:
-
E2F transcription factor 1
- ELF3-AS1:
-
E74 like ETS transcription factor 3 antisense 1
- EGF:
-
Epidermal growth factor
- EC:
-
Esophageal cancer
- EV:
-
Extracellular vesicle
- FCS:
-
Familial chylomicronemia syndrome
- FGFR1:
-
Fibroblast growth factor receptor type1
- FDA:
-
Food and Drug Administration
- FILNC1:
-
FoxO-induced long non-coding RNA 1
- F-1,6-BP:
-
Fructose-1,6-biphosphate
- FBPase:
-
Fructose-2,6-biphosphatase
- F-6-P:
-
Fructose-6-phosphate
- GBS:
-
Gallbladder cancer
- GLUTs:
-
Glucose transports
- G-6-P:
-
Glucose-6-phosphate
- GSH/GSSH:
-
Glutathione/oxidized glutathione
- GADPH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- GLCC1:
-
Glycolysis-associated lncRNA of colorectal cancer
- GPNMB:
-
Glycoprotein NMB
- HCC:
-
Hepatocellular carcinoma
- hnRNP A1:
-
Heterogeneous nuclear ribonucleoprotein A1
- HK:
-
Hexokinase
- HDAC2:
-
Histone deacetylase
- HER:
-
Hypoxia reaction elements
- HIF-1α:
-
Hypoxia-inducible factor 1α
- IGF2BP2:
-
Insulin-like growth factor 2 mRNA-binding protein 2
- IDH:
-
Isocitrate dehydrogenase
- LDH:
-
Lactate dehydrogenase
- LNA:
-
Locked nuclear acid
- LncRNA:
-
Long non-coding RNA
- mTOR:
-
Mammalian target of rapamycin
- MALAT1:
-
Metastasis-associated lung adenocarcinoma transcript 1
- MCT:
-
Monocarboxylate transport family
- MM:
-
Multiple myeloma
- NBR2:
-
Neighbor of BRCA1 gene 2
- NcRNAs:
-
Non-coding RNAs
- NSCLC:
-
Non-small cell lung cancer
- OSCC:
-
Oral squamous cell carcinoma
- OXPHOS:
-
Oxidative phosphorylation
- TIGAR:
-
P53-induced glycolysis and apoptosis regulator
- PFK-1:
-
6-Phosphofructokinase-1
- PFK-2/PFKFB:
-
6-Phosphofructokinase-2/fructose-2,6-biphosphatase
- PPP:
-
Pentose phosphate pathway
- PI3K:
-
Phosphatidylinositol-3-hydroxykinase
- PEP:
-
Phosphoenolpyruvate
- PVT1:
-
Plasmacytoma variant translocation
- PET:
-
Positron emission tomography scans
- PHD:
-
Proline hydroxylase domain
- PDH:
-
Pyruvate dehydrogenase
- PDK:
-
Pyruvate dehydrogenase kinase
- PK:
-
Pyruvate kinase
- PPARA:
-
Peroxisome proliferator-activated receptor alpha
- ROS:
-
Reactive oxygen species
- RCC:
-
Renal cell carcinoma
- RNAi:
-
RNA interference
- RRM2:
-
M2 subunit of ribonucleotide reductase
- SIRT1:
-
Deacetylases Sirtuin
- siRNA:
-
Small interference RNA
- SGLT:
-
Sodium-dependent glucose transporter family
- SCO2:
-
Synthesis of cytochrome c oxidase 2
- SOX:
-
Cytochrome c oxidase complex
- TF:
-
Transcriptional factors
- TCA:
-
Tricarboxylic acid
- TNBC:
-
Triple-negative breast cancer
- TSC1 and TSC2:
-
Tuberous sclerosis 1,2
- ME:
-
Tumor microenvironment
- TAM:
-
Tumor-associated macrophages
- VHL:
-
Von Hipple-Lindau protein
- XIST:
-
X-inactive specific transcript
- 18F-FDG:
-
18F-labeled 2-fluoro-2-deoxy-d-glucose
- 2-DG:
-
2-Deoxyglucose
- 3-BrPA:
-
3-Bromopyruvate
- α-DG:
-
α-Hydroxyglutarate
- α-KG:
-
α-Ketoglutarate
References
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics. CA 69(1):7–34. https://doi.org/10.3322/caac.21551
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100(1):57–70. https://doi.org/10.1016/s0092-8674(00)81683-9
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. https://doi.org/10.1016/j.cell.2011.02.013
Pavlova NN, Thompson CB (2016) The emerging hallmarks of cancer metabolism. Cell Metab 23(1):27–47. https://doi.org/10.1016/j.cmet.2015.12.006
Warburg O (1956) On the origin of cancer cells. Science (New York, NY) 123(3191):309–314. https://doi.org/10.1126/science.123.3191.309
Warburg O (1928) The chemical constitution of respiration ferment. Science (New York, NY) 68(1767):437–443. https://doi.org/10.1126/science.68.1767.437
Warburg O (1956) On respiratory impairment in cancer cells. Science (New York, NY) 124(3215):269–270
Wang T, Marquardt C, Foker J (1976) Aerobic glycolysis during lymphocyte proliferation. Nature 261(5562):702–705. https://doi.org/10.1038/261702a0
Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science (New York, NY) 324(5930):1029–1033. https://doi.org/10.1126/science.1160809
Peppicelli S, Bianchini F, Calorini L (2014) Extracellular acidity, a “reappreciated” trait of tumor environment driving malignancy: perspectives in diagnosis and therapy. Cancer Metastasis Rev 33(2–3):823–832. https://doi.org/10.1007/s10555-014-9506-4
Shiraishi T, Verdone JE, Huang J, Kahlert UD, Hernandez JR, Torga G, Zarif JC, Epstein T, Gatenby R, McCartney A, Elisseeff JH, Mooney SM, An SS, Pienta KJ (2015) Glycolysis is the primary bioenergetic pathway for cell motility and cytoskeletal remodeling in human prostate and breast cancer cells. Oncotarget 6(1):130–143. https://doi.org/10.18632/oncotarget.2766
Abdel-Wahab AF, Mahmoud W, Al-Harizy RM (2019) Targeting glucose metabolism to suppress cancer progression: prospective of anti-glycolytic cancer therapy. Pharmacol Res 150:104511. https://doi.org/10.1016/j.phrs.2019.104511
Razavi ZS, Tajiknia V, Majidi S, Ghandali M, Mirzaei HR, Rahimian N, Hamblin MR, Mirzaei H (2021) Gynecologic cancers and non-coding RNAs: epigenetic regulators with emerging roles. Crit Rev Oncol Hematol 157:103192. https://doi.org/10.1016/j.critrevonc.2020.103192
Beermann J, Piccoli MT, Viereck J, Thum T (2016) Non-coding RNAs in development and disease: background, mechanisms, and therapeutic approaches. Physiol Rev 96(4):1297–1325. https://doi.org/10.1152/physrev.00041.2015
Vafadar A, Shabaninejad Z, Movahedpour A, Mohammadi S, Fathullahzadeh S, Mirzaei HR, Namdar A, Savardashtaki A, Mirzaei H (2019) Long non-coding RNAs as epigenetic regulators in cancer. Curr Pharm Des 25(33):3563–3577. https://doi.org/10.2174/1381612825666190830161528
Guttman M, Rinn JL (2012) Modular regulatory principles of large non-coding RNAs. Nature 482(7385):339–346. https://doi.org/10.1038/nature10887
Mirzaei H, Hamblin MR (2020) Regulation of glycolysis by non-coding RNAs in cancer: switching on the Warburg effect. Molecular therapy oncolytics 19:218–239. https://doi.org/10.1016/j.omto.2020.10.003
Sun H, Huang Z, Sheng W, Xu MD (2018) Emerging roles of long non-coding RNAs in tumor metabolism. J Hematol Oncol 11(1):106. https://doi.org/10.1186/s13045-018-0648-7
Vincent EE, Coelho PP, Blagih J, Griss T, Viollet B, Jones RG (2015) Differential effects of AMPK agonists on cell growth and metabolism. Oncogene 34(28):3627–3639. https://doi.org/10.1038/onc.2014.301
Szablewski L (2013) Expression of glucose transporters in cancers. Biochim Biophys Acta 1835(2):164–169. https://doi.org/10.1016/j.bbcan.2012.12.004
Yu M, Yongzhi H, Chen S, Luo X, Lin Y, Zhou Y, Jin H, Hou B, Deng Y, Tu L, Jian Z (2017) The prognostic value of GLUT1 in cancers: a systematic review and meta-analysis. Oncotarget 8(26):43356–43367. https://doi.org/10.18632/oncotarget.17445
Phadngam S, Castiglioni A, Ferraresi A, Morani F, Follo C, Isidoro C (2016) PTEN dephosphorylates AKT to prevent the expression of GLUT1 on plasmamembrane and to limit glucose consumption in cancer cells. Oncotarget 7(51):84999–85020. https://doi.org/10.18632/oncotarget.13113
Chu H, Li Z, Gan Z, Yang Z, Wu Z, Rong M (2019) LncRNA ELF3-AS1 is involved in the regulation of oral squamous cell carcinoma cell proliferation by reprogramming glucose metabolism. Onco Targets Ther 12:6857–6863. https://doi.org/10.2147/ott.S217473
Wei S, Fan Q, Yang L, Zhang X, Ma Y, Zong Z, Hua X, Su D, Sun H, Li H, Liu Z (2017) Promotion of glycolysis by HOTAIR through GLUT1 upregulation via mTOR signaling. Oncol Rep 38(3):1902–1908. https://doi.org/10.3892/or.2017.5840
Patra KC, Hay N (2013) Hexokinase 2 as oncotarget. Oncotarget 4(11):1862–1863. https://doi.org/10.18632/oncotarget.1563
Chen J, Yu Y, Li H, Hu Q, Chen X, He Y, Xue C, Ren F, Ren Z, Li J, Liu L, Duan Z, Cui G, Sun R (2019) Long non-coding RNA PVT1 promotes tumor progression by regulating the miR-143/HK2 axis in gallbladder cancer. Mol Cancer 18(1):33. https://doi.org/10.1186/s12943-019-0947-9
Shi H, Li K, Feng J, Liu G, Feng Y, Zhang X (2020) LncRNA-DANCR interferes with miR-125b-5p/HK2 Axis to desensitize colon cancer cells to Cisplatin vis activating anaerobic glycolysis. Front Oncol 10:1034. https://doi.org/10.3389/fonc.2020.01034
Tang D, Yang Z, Long F, Luo L, Yang B, Zhu R, Sang X, Cao G, Wang K (2019) Long noncoding RNA MALAT1 mediates stem cell-like properties in human colorectal cancer cells by regulating miR-20b-5p/Oct4 axis. J Cell Physiol 234(11):20816–20828. https://doi.org/10.1002/jcp.28687
Al Hasawi N, Alkandari MF, Luqmani YA (2014) Phosphofructokinase: a mediator of glycolytic flux in cancer progression. Crit Rev Oncol Hematol 92(3):312–321. https://doi.org/10.1016/j.critrevonc.2014.05.007
Yalcin A, Clem BF, Imbert-Fernandez Y, Ozcan SC, Peker S, O’Neal J, Klarer AC, Clem AL, Telang S, Chesney J (2014) 6-Phosphofructo-2-kinase (PFKFB3) promotes cell cycle progression and suppresses apoptosis via Cdk1-mediated phosphorylation of p27. Cell Death Dis 5(7):e1337. https://doi.org/10.1038/cddis.2014.292
Liu J, Liu ZX, Wu QN, Lu YX, Wong CW, Miao L, Wang Y, Wang Z, Jin Y, He MM, Ren C, Wang DS, Chen DL, Pu HY, Feng L, Li B, Xie D, Zeng MS, Huang P, Lin A, Lin D, Xu RH, Ju HQ (2020) Long noncoding RNA AGPG regulates PFKFB3-mediated tumor glycolytic reprogramming. Nat Commun 11(1):1507. https://doi.org/10.1038/s41467-020-15112-3
Xing Z, Zhang Y, Liang K, Yan L, Xiang Y, Li C, Hu Q, Jin F, Putluri V, Putluri N, Coarfa C, Sreekumar A, Park PK, Nguyen TK, Wang S, Zhou J, Zhou Y, Marks JR, Hawke DH, Hung MC, Yang L, Han L, Ying H, Lin C (2018) Expression of long noncoding RNA YIYA promotes glycolysis in breast cancer. Cancer Res 78(16):4524–4532. https://doi.org/10.1158/0008-5472.Can-17-0385
Israelsen WJ, Vander Heiden MG (2015) Pyruvate kinase: function, regulation and role in cancer. Semin Cell Dev Biol 43:43–51. https://doi.org/10.1016/j.semcdb.2015.08.004
David CJ, Chen M, Assanah M, Canoll P, Manley JL (2010) HnRNP proteins controlled by c-Myc deregulate pyruvate kinase mRNA splicing in cancer. Nature 463(7279):364–368. https://doi.org/10.1038/nature08697
Chen M, David CJ, Manley JL (2012) Concentration-dependent control of pyruvate kinase M mutually exclusive splicing by hnRNP proteins. Nat Struct Mol Biol 19(3):346–354. https://doi.org/10.1038/nsmb.2219
Huang JZ, Chen M, Chen D, Gao XC, Zhu S, Huang H, Hu M, Zhu H, Yan GR (2017) A peptide encoded by a putative lncRNA HOXB-AS3 suppresses colon cancer growth. Mol Cell 68(1):171-184.e176. https://doi.org/10.1016/j.molcel.2017.09.015
Luo W, Hu H, Chang R, Zhong J, Knabel M, O’Meally R, Cole RN, Pandey A, Semenza GL (2011) Pyruvate kinase M2 is a PHD3-stimulated coactivator for hypoxia-inducible factor 1. Cell 145(5):732–744. https://doi.org/10.1016/j.cell.2011.03.054
Wang C, Li Y, Yan S, Wang H, Shao X, Xiao M, Yang B, Qin G, Kong R, Chen R, Zhang N (2020) Interactome analysis reveals that lncRNA HULC promotes aerobic glycolysis through LDHA and PKM2. Nat Commun 11(1):3162. https://doi.org/10.1038/s41467-020-16966-3
Gao X, Wang H, Yang JJ, Liu X, Liu ZR (2012) Pyruvate kinase M2 regulates gene transcription by acting as a protein kinase. Mol Cell 45(5):598–609. https://doi.org/10.1016/j.molcel.2012.01.001
Bian Z, Zhang J, Li M, Feng Y, Wang X, Zhang J, Yao S, Jin G, Du J, Han W, Yin Y, Huang S, Fei B, Zou J, Huang Z (2018) LncRNA-FEZF1-AS1 promotes tumor proliferation and metastasis in colorectal cancer by regulating PKM2 signaling. Clin Cancer Res 24(19):4808–4819. https://doi.org/10.1158/1078-0432.Ccr-17-2967
Le A, Cooper CR, Gouw AM, Dinavahi R, Maitra A, Deck LM, Royer RE, Vander Jagt DL, Semenza GL, Dang CV (2010) Inhibition of lactate dehydrogenase A induces oxidative stress and inhibits tumor progression. Proc Natl Acad Sci USA 107(5):2037–2042. https://doi.org/10.1073/pnas.0914433107
Zhou Y, Huang Y, Hu K, Zhang Z, Yang J, Wang Z (2020) HIF1A activates the transcription of lncRNA RAET1K to modulate hypoxia-induced glycolysis in hepatocellular carcinoma cells via miR-100-5p. Cell Death Dis 11(3):176. https://doi.org/10.1038/s41419-020-2366-7
Du P, Liao Y, Zhao H, Zhang J, Muyiti K, Mu K (2020) ANXA2P2/miR-9/LDHA axis regulates Warburg effect and affects glioblastoma proliferation and apoptosis. Cell Signal 74:109718. https://doi.org/10.1016/j.cellsig.2020.109718
Payen VL, Mina E, Van Hée VF, Porporato PE, Sonveaux P (2020) Monocarboxylate transporters in cancer. Mol Metab 33:48–66. https://doi.org/10.1016/j.molmet.2019.07.006
Logotheti S, Marquardt S, Gupta SK, Richter C, Edelhäuser BAH, Engelmann D, Brenmoehl J, Söhnchen C, Murr N, Alpers M, Singh KP, Wolkenhauer O, Heckl D, Spitschak A, Pützer BM (2020) LncRNA-SLC16A1-AS1 induces metabolic reprogramming during Bladder Cancer progression as target and co-activator of E2F1. Theranostics 10(21):9620–9643. https://doi.org/10.7150/thno.44176
Woolbright BL, Rajendran G, Harris RA, Taylor JA 3rd (2019) Metabolic flexibility in cancer: targeting the pyruvate dehydrogenase kinase: pyruvate dehydrogenase axis. Mol Cancer Ther 18(10):1673–1681. https://doi.org/10.1158/1535-7163.Mct-19-0079
Xu S, Guo J, Zhang W (2019) lncRNA PCAT19 promotes the proliferation of laryngocarcinoma cells via modulation of the miR-182/PDK4 axis. J Cell Biochem 120(8):12810–12821. https://doi.org/10.1002/jcb.28552
Liu Y, He X, Chen Y, Cao D (2020) Long non-coding RNA LINC00504 regulates the Warburg effect in ovarian cancer through inhibition of miR-1244. Mol Cell Biochem 464(1–2):39–50. https://doi.org/10.1007/s11010-019-03647-z
Kim J, DeBerardinis RJ (2019) Mechanisms and implications of metabolic heterogeneity in cancer. Cell Metab 30(3):434–446. https://doi.org/10.1016/j.cmet.2019.08.013
Dang L, Yen K, Attar EC (2016) IDH mutations in cancer and progress toward development of targeted therapeutics. Ann Oncol 27(4):599–608. https://doi.org/10.1093/annonc/mdw013
Xiang S, Gu H, Jin L, Thorne RF, Zhang XD, Wu M (2018) LncRNA IDH1-AS1 links the functions of c-Myc and HIF1α via IDH1 to regulate the Warburg effect. Proc Natl Acad Sci USA 115(7):E1465-e1474. https://doi.org/10.1073/pnas.1711257115
Dalla-Favera R, Bregni M, Erikson J, Patterson D, Gallo RC, Croce CM (1982) Human c-myc onc gene is located on the region of chromosome 8 that is translocated in Burkitt lymphoma cells. Proc Natl Acad Sci USA 79(24):7824–7827. https://doi.org/10.1073/pnas.79.24.7824
Dang CV (2012) MYC on the path to cancer. Cell 149(1):22–35. https://doi.org/10.1016/j.cell.2012.03.003
Stine ZE, Walton ZE, Altman BJ, Hsieh AL, Dang CV (2015) MYC, metabolism, and cancer. Cancer Discov 5(10):1024–1039. https://doi.org/10.1158/2159-8290.Cd-15-0507
Hua Q, Jin M, Mi B, Xu F, Li T, Zhao L, Liu J, Huang G (2019) LINC01123, a c-Myc-activated long non-coding RNA, promotes proliferation and aerobic glycolysis of non-small cell lung cancer through miR-199a-5p/c-Myc axis. J Hematol Oncol 12(1):91. https://doi.org/10.1186/s13045-019-0773-y
Xiao ZD, Han L, Lee H, Zhuang L, Zhang Y, Baddour J, Nagrath D, Wood CG, Gu J, Wu X, Liang H, Gan B (2017) Energy stress-induced lncRNA FILNC1 represses c-Myc-mediated energy metabolism and inhibits renal tumor development. Nat Commun 8(1):783. https://doi.org/10.1038/s41467-017-00902-z
Tang J, Yan T, Bao Y, Shen C, Yu C, Zhu X, Tian X, Guo F, Liang Q, Liu Q, Zhong M, Chen J, Ge Z, Li X, Chen X, Cui Y, Chen Y, Zou W, Chen H, Hong J, Fang JY (2019) LncRNA GLCC1 promotes colorectal carcinogenesis and glucose metabolism by stabilizing c-Myc. Nat Commun 10(1):3499. https://doi.org/10.1038/s41467-019-11447-8
Wang Y, Lu JH, Wu QN, Jin Y, Wang DS, Chen YX, Liu J, Luo XJ, Meng Q, Pu HY, Wang YN, Hu PS, Liu ZX, Zeng ZL, Zhao Q, Deng R, Zhu XF, Ju HQ, Xu RH (2019) LncRNA LINRIS stabilizes IGF2BP2 and promotes the aerobic glycolysis in colorectal cancer. Mol Cancer 18(1):174. https://doi.org/10.1186/s12943-019-1105-0
Ma F, Liu X, Zhou S, Li W, Liu C, Chadwick M, Qian C (2019) Long non-coding RNA FGF13-AS1 inhibits glycolysis and stemness properties of breast cancer cells through FGF13-AS1/IGF2BPs/Myc feedback loop. Cancer Lett 450:63–75. https://doi.org/10.1016/j.canlet.2019.02.008
Semenza GL (2011) Hypoxia-inducible factor 1: regulator of mitochondrial metabolism and mediator of ischemic preconditioning. Biochim Biophys Acta 1813(7):1263–1268. https://doi.org/10.1016/j.bbamcr.2010.08.006
Semenza GL (2009) Regulation of cancer cell metabolism by hypoxia-inducible factor 1. Semin Cancer Biol 19(1):12–16. https://doi.org/10.1016/j.semcancer.2008.11.009
Kaelin WG Jr, Ratcliffe PJ (2008) Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol Cell 30(4):393–402. https://doi.org/10.1016/j.molcel.2008.04.009
Courtnay R, Ngo DC, Malik N, Ververis K, Tortorella SM, Karagiannis TC (2015) Cancer metabolism and the Warburg effect: the role of HIF-1 and PI3K. Mol Biol Rep 42(4):841–851. https://doi.org/10.1007/s11033-015-3858-x
Yang F, Zhang H, Mei Y, Wu M (2014) Reciprocal regulation of HIF-1α and lincRNA-p21 modulates the Warburg effect. Mol Cell 53(1):88–100. https://doi.org/10.1016/j.molcel.2013.11.004
Lin A, Li C, Xing Z, Hu Q, Liang K, Han L, Wang C, Hawke DH, Wang S, Zhang Y, Wei Y, Ma G, Park PK, Zhou J, Zhou Y, Hu Z, Zhou Y, Marks JR, Liang H, Hung MC, Lin C, Yang L (2016) The LINK-A lncRNA activates normoxic HIF1α signalling in triple-negative breast cancer. Nat Cell Biol 18(2):213–224. https://doi.org/10.1038/ncb3295
Chen P, Zuo H, Xiong H, Kolar MJ, Chu Q, Saghatelian A, Siegwart DJ, Wan Y (2017) Gpr132 sensing of lactate mediates tumor-macrophage interplay to promote breast cancer metastasis. Proc Natl Acad Sci USA 114(3):580–585. https://doi.org/10.1073/pnas.1614035114
Chen F, Chen J, Yang L, Liu J, Zhang X, Zhang Y, Tu Q, Yin D, Lin D, Wong PP, Huang D, Xing Y, Zhao J, Li M, Liu Q, Su F, Su S, Song E (2019) Extracellular vesicle-packaged HIF-1α-stabilizing lncRNA from tumour-associated macrophages regulates aerobic glycolysis of breast cancer cells. Nat Cell Biol 21(4):498–510. https://doi.org/10.1038/s41556-019-0299-0
Vousden KH, Ryan KM (2009) p53 and metabolism. Nat Rev Cancer 9(10):691–700. https://doi.org/10.1038/nrc2715
Khan MR, Xiang S, Song Z, Wu M (2017) The p53-inducible long noncoding RNA TRINGS protects cancer cells from necrosis under glucose starvation. Embo J 36(23):3483–3500. https://doi.org/10.15252/embj.201696239
Bensaad K, Tsuruta A, Selak MA, Vidal MN, Nakano K, Bartrons R, Gottlieb E, Vousden KH (2006) TIGAR, a p53-inducible regulator of glycolysis and apoptosis. Cell 126(1):107–120. https://doi.org/10.1016/j.cell.2006.05.036
Du W, Amarachintha S, Wilson AF, Pang Q (2016) SCO2 Mediates oxidative stress-induced glycolysis to oxidative phosphorylation switch in hematopoietic stem cells. Stem Cells (Dayton, Ohio) 34(4):960–971. https://doi.org/10.1002/stem.2260
Chen R, Liu Y, Zhuang H, Yang B, Hei K, Xiao M, Hou C, Gao H, Zhang X, Jia C, Li L, Li Y, Zhang N (2017) Quantitative proteomics reveals that long non-coding RNA MALAT1 interacts with DBC1 to regulate p53 acetylation. Nucleic Acids Res 45(17):9947–9959. https://doi.org/10.1093/nar/gkx600
Liao M, Liao W, Xu N, Li B, Liu F, Zhang S, Wang Y, Wang S, Zhu Y, Chen D, Xie W, Jiang Y, Cao L, Yang BB, Zhang Y (2019) LncRNA EPB41L4A-AS1 regulates glycolysis and glutaminolysis by mediating nucleolar translocation of HDAC2. EBioMedicine 41:200–213. https://doi.org/10.1016/j.ebiom.2019.01.035
Elstrom RL, Bauer DE, Buzzai M, Karnauskas R, Harris MH, Plas DR, Zhuang H, Cinalli RM, Alavi A, Rudin CM, Thompson CB (2004) Akt stimulates aerobic glycolysis in cancer cells. Cancer Res 64(11):3892–3899. https://doi.org/10.1158/0008-5472.Can-03-2904
Li W, Huang K, Wen F, Cui G, Guo H, He Z, Zhao S (2019) LINC00184 silencing inhibits glycolysis and restores mitochondrial oxidative phosphorylation in esophageal cancer through demethylation of PTEN. EBioMedicine 44:298–310. https://doi.org/10.1016/j.ebiom.2019.05.055
Zheng YL, Li L, Jia YX, Zhang BZ, Li JC, Zhu YH, Li MQ, He JZ, Zeng TT, Ban XJ, Yuan YF, Li Y, Guan XY (2019) LINC01554-mediated glucose metabolism reprogramming suppresses tumorigenicity in hepatocellular carcinoma via downregulating PKM2 expression and inhibiting Akt/mTOR signaling pathway. Theranostics 9(3):796–810. https://doi.org/10.7150/thno.28992
Cheng Z, Luo C, Guo Z (2020) LncRNA-XIST/microRNA-126 sponge mediates cell proliferation and glucose metabolism through the IRS1/PI3K/Akt pathway in glioma. J Cell Biochem 121(3):2170–2183. https://doi.org/10.1002/jcb.29440
Hardie DG (2015) Molecular pathways: is AMPK a friend or a foe in cancer? Clin Cancer Res 21(17):3836–3840. https://doi.org/10.1158/1078-0432.Ccr-14-3300
Liu X, Xiao ZD, Han L, Zhang J, Lee SW, Wang W, Lee H, Zhuang L, Chen J, Lin HK, Wang J, Liang H, Gan B (2016) LncRNA NBR2 engages a metabolic checkpoint by regulating AMPK under energy stress. Nat Cell Biol 18(4):431–442. https://doi.org/10.1038/ncb3328
Liu X, Gan B (2016) lncRNA NBR2 modulates cancer cell sensitivity to phenformin through GLUT1. Cell Cycle 15(24):3471–3481. https://doi.org/10.1080/15384101.2016.1249545
Sun LY, Li XJ, Sun YM, Huang W, Fang K, Han C, Chen ZH, Luo XQ, Chen YQ, Wang WT (2018) LncRNA ANRIL regulates AML development through modulating the glucose metabolism pathway of AdipoR1/AMPK/SIRT1. Mol Cancer 17(1):127. https://doi.org/10.1186/s12943-018-0879-9
Zhao Y, Liu Y, Lin L, Huang Q, He W, Zhang S, Dong S, Wen Z, Rao J, Liao W, Shi M (2018) The lncRNA MACC1-AS1 promotes gastric cancer cell metabolic plasticity via AMPK/Lin28 mediated mRNA stability of MACC1. Mol Cancer 17(1):69. https://doi.org/10.1186/s12943-018-0820-2
Bomanji JB, Costa DC, Ell PJ (2001) Clinical role of positron emission tomography in oncology. Lancet Oncol 2(3):157–164. https://doi.org/10.1016/s1470-2045(00)00257-6
Dalva-Aydemir S, Bajpai R, Martinez M, Adekola KU, Kandela I, Wei C, Singhal S, Koblinski JE, Raje NS, Rosen ST, Shanmugam M (2015) Targeting the metabolic plasticity of multiple myeloma with FDA-approved ritonavir and metformin. Clin Cancer Res 21(5):1161–1171. https://doi.org/10.1158/1078-0432.Ccr-14-1088
Sheng Y, Jiang Q, Dong X, Liu J, Liu L, Wang H, Wang L, Li H, Yang X, Dong J (2020) 3-Bromopyruvate inhibits the malignant phenotype of malignantly transformed macrophages and dendritic cells induced by glioma stem cells in the glioma microenvironment via miR-449a/MCT1. Biomed Pharmacother 121:109610. https://doi.org/10.1016/j.biopha.2019.109610
Icard P, Shulman S, Farhat D, Steyaert JM, Alifano M, Lincet H (2018) How the Warburg effect supports aggressiveness and drug resistance of cancer cells? Drug Resist Updates 38:1–11. https://doi.org/10.1016/j.drup.2018.03.001
Vasan N, Baselga J, Hyman DM (2019) A view on drug resistance in cancer. Nature 575(7782):299–309. https://doi.org/10.1038/s41586-019-1730-1
Noël G, Langouo Fontsa M, Willard-Gallo K (2018) The impact of tumor cell metabolism on T cell-mediated immune responses and immuno-metabolic biomarkers in cancer. Semin Cancer Biol 52(Pt 2):66–74. https://doi.org/10.1016/j.semcancer.2018.03.003
Ramapriyan R, Caetano MS, Barsoumian HB, Mafra ACP, Zambalde EP, Menon H, Tsouko E, Welsh JW, Cortez MA (2019) Altered cancer metabolism in mechanisms of immunotherapy resistance. Pharmacol Ther 195:162–171. https://doi.org/10.1016/j.pharmthera.2018.11.004
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391(6669):806–811. https://doi.org/10.1038/35888
Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T (2001) Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411(6836):494–498. https://doi.org/10.1038/35078107
Davis ME, Zuckerman JE, Choi CH, Seligson D, Tolcher A, Alabi CA, Yen Y, Heidel JD, Ribas A (2010) Evidence of RNAi in humans from systemically administered siRNA via targeted nanoparticles. Nature 464(7291):1067–1070. https://doi.org/10.1038/nature08956
Golan T, Khvalevsky EZ, Hubert A, Gabai RM, Hen N, Segal A, Domb A, Harari G, David EB, Raskin S, Goldes Y, Goldin E, Eliakim R, Lahav M, Kopleman Y, Dancour A, Shemi A, Galun E (2015) RNAi therapy targeting KRAS in combination with chemotherapy for locally advanced pancreatic cancer patients. Oncotarget 6(27):24560–24570. https://doi.org/10.18632/oncotarget.4183
Xue VW, Wong SCC, Song G, Cho WCS (2020) Promising RNA-based cancer gene therapy using extracellular vesicles for drug delivery. Expert Opin Biol Ther 20(7):767–777. https://doi.org/10.1080/14712598.2020.1738377
Das M, Musetti S, Huang L (2019) RNA Interference-Based Cancer Drugs: The Roadblocks, and the “Delivery” of the Promise. Nucleic acid therapeutics 29(2):61–66. https://doi.org/10.1089/nat.2018.0762
Mainini F, Eccles MR (2020) Lipid and polymer-based nanoparticle siRNA delivery systems for cancer therapy. Molecules (Basel, Switzerland). https://doi.org/10.3390/molecules25112692
Maruyama R, Yokota T (2020) Knocking down long noncoding RNAs using antisense oligonucleotide gapmers. Methods Mol Biol 2176:49–56. https://doi.org/10.1007/978-1-0716-0771-8_3
Post N, Yu R, Greenlee S, Gaus H, Hurh E, Matson J, Wang Y (2019) Metabolism and disposition of volanesorsen, a 2’-O-(2 methoxyethyl) antisense oligonucleotide, across species. Drug Metabol Disposit 47(10):1164–1173. https://doi.org/10.1124/dmd.119.087395
Amodio N, Stamato MA, Juli G, Morelli E, Fulciniti M, Manzoni M, Taiana E, Agnelli L, Cantafio MEG, Romeo E, Raimondi L, Caracciolo D, Zuccalà V, Rossi M, Neri A, Munshi NC, Tagliaferri P, Tassone P (2018) Drugging the lncRNA MALAT1 via LNA gapmeR ASO inhibits gene expression of proteasome subunits and triggers anti-multiple myeloma activity. Leukemia 32(9):1948–1957. https://doi.org/10.1038/s41375-018-0067-3
Fathi Dizaji B (2020) Strategies to target long non-coding RNAs in cancer treatment: progress and challenges. Egypt J Med Hum Genet 21:41. https://doi.org/10.1186/s43042-020-00074-4
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. RL performed the literature search and analysis. RL prepared tables and figures, and drafted the manuscript. AH, YS and XW revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, R., Wang, X., Shen, Y. et al. Long non-coding RNA-based glycolysis-targeted cancer therapy: feasibility, progression and limitations. Mol Biol Rep 48, 2713–2727 (2021). https://doi.org/10.1007/s11033-021-06247-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06247-7