Abstract
Background
Lactoferrin, as the main component of milk, can maintain osteoblast formation, which is conducive to the prevention and treatment of osteoporosis. Lactoferrin also serves as an autophagy regulator, especially in osteoblasts. This study aimed to explore the significance of autophagy in osteoblast formation regulated by lactoferrin and the internal mechanism.
Methods and results
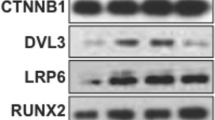
In this study, we firstly explored the roles of lactoferrin in the autophagy activity of primary osteoblasts (LC3 transformation rate, autophagosome formation). Subsequently, we further investigated the effects of lactoferrin on the BCL2 expression and BCL2-Beclin1 complex. Ultimately, the significance of BCL2 overexpression and Beclin1 silencing on lactoferrin-regulated osteoblast autophagy and osteogenic parameters (ALP activity and mRNA expression of PCNA, Col1, BGLAP and OPN) was observed by gene processing, respectively. Our results showed that lactoferrin enhanced the autophagy activity of osteoblasts. Importantly, lactoferrin inhibited BCL2 expression and the co-immunoprecipitation of BCL2 and Beclin1 in osteoblasts. Moreover, lactoferrin-promoted autophagy and osteogenic parameters was reversed by BCL2 overexpression or Beclin1 silencing in osteoblasts.
Conclusions
In conclusion, lactoferrin can inhibit BCL2 expression in osteoblasts, further enhancing Beclin1-dependent autophagy activation.





Similar content being viewed by others
References
Włodarski K (2009) Lactoferrin—a promising bone-growth promoting milk-derived glycoprotein. Chir Narzadow Ruchu Ortop Pol 74(257–259):322–323
Hou JM, Xue Y, Lin QM (2012) Bovine lactoferrin improves bone mass and microstructure in ovariectomized rats via OPG/RANKL/RANK pathway. Acta Pharmacol Sin 33:1277–1284. https://doi.org/10.1038/aps.2012.83
Guo HY, Jiang L, Ibrahim SA, Zhang L, Zhang H, Zhang M, Ren FZ (2009) Orally administered lactoferrin preserves bone mass and microarchitecture in ovariectomized rats. J Nutr 139:958–964. https://doi.org/10.3945/jn.108.100586
Shi P, Fan F, Chen H, Xu Z, Cheng S, Lu W, Du M (2020) A bovine lactoferrin-derived peptide induced osteogenesis via regulation of osteoblast proliferation and differentiation. J Dairy Sci 103:3950–3960. https://doi.org/10.3168/jds.2019-17425
Chen XW, Li YH, Zhang MJ, Chen Z, Ke DS, Xue Y, Hou JM (2019) Lactoferrin ameliorates aging-suppressed osteogenesis via IGF1 signaling. J Mol Endocrinol 63:63–75. https://doi.org/10.1530/JME-19-0003
Li Y, Wang J, Ren F, Zhang W, Zhang H, Zhao L, Zhang M, Cui W, Wang X, Guo H (2018) Lactoferrin promotes osteogenesis through TGF-β receptor II binding in osteoblasts and activation of canonical TGF-β signaling in MC3T3-E1 cells and C57BL/6J mice. J Nutr 148:1285–1292. https://doi.org/10.1093/jn/nxy097
Zhang JL, Han X, Shan YJ, Zhang LW, Du M, Liu M, Yi HX, Ma Y (2018) Effect of bovine lactoferrin and human lactoferrin on the proliferative activity of the osteoblast cell line MC3T3-E1 in vitro. J Dairy Sci 101:1827–1833. https://doi.org/10.3168/jds.2017-13161
Huang L, Yang Z, Liu R, Xiao X, Zhou C, Yin X, Zou S, Chen J (2021) Lactoferrin promotes osteogenesis of MC3T3-E1 cells induced by mechanical strain in an extracellular signal-regulated kinase 1/2-dependent manner. Am J Orthod Dentofac Orthop 159:e113–e121. https://doi.org/10.1016/j.ajodo.2020.08.015
Xu D, Song W, Zhang J, Liu Y, Lu Y, Zhang X, Liu Q, Yuan T, Liu R (2021) Osteogenic effect of polymethyl methacrylate bone cement with surface modification of lactoferrin. J Biosci Bioeng 132:132–139. https://doi.org/10.1016/j.jbiosc.2021.04.006
Liu M, Fan F, Shi P, Tu M, Yu C, Yu C, Du M (2018) Lactoferrin promotes MC3T3-E1 osteoblast cells proliferation via MAPK signaling pathways. Int J Biol Macromol 107:137–143. https://doi.org/10.1016/j.ijbiomac.2017.08.151
Li Y, Huang J, Wang J, Ma M, Lu Y, Wang R, Guo H (2021) Lactoferrin is a potential activator of the vitamin D receptor in its regulation of osteogenic activities in C57BL/6J mice and MC3T3-E1 cells. J Nutr 151:2105–2113. https://doi.org/10.1093/jn/nxab105
Zhang Y, Zhang ZN, Li N, Zhao LJ, Xue Y, Wu HJ, Hou JM (2020) Nbr1-regulated autophagy in lactoferrin-induced osteoblastic differentiation. Biosci Biotechnol Biochem 84:1191–1200. https://doi.org/10.1080/09168451.2020.1737505
Kim SE, Choi S, Hong JY, Shim KS, Kim TH, Park K, Lee SH (2019) Accelerated osteogenic differentiation of MC3T3-E1 cells by lactoferrin-conjugated nanodiamonds through enhanced anti-oxidant and anti-inflammatory effects. Nanomaterials (Basel) 10:50. https://doi.org/10.3390/nano10010050
Weng YM, Ke CR, Kong JZ, Chen H, Hong JJ, Zhou DS (2018) The significant role of ATG5 in the maintenance of normal functions of MC3T3-E1 osteoblast. Eur Rev Med Pharmacol Sci 22:1224–1232. https://doi.org/10.26355/eurrev_201803_14462
Vidoni C, Ferraresi A, Secomandi E, Vallino L, Gardin C, Zavan B, Mortellaro C, Isidoro C (2019) Autophagy drives osteogenic differentiation of human gingival mesenchymal stem cells. Cell Commun Signal 17:98. https://doi.org/10.1186/s12964-019-0414-7
Kang C, Wei L, Song B, Chen L, Liu J, Deng B, Pan X, Shao L (2017) Involvement of autophagy in tantalum nanoparticle-induced osteoblast proliferation. Int J Nanomed 12:4323–4333. https://doi.org/10.2147/IJN.S136281
Lian WS, Ko JY, Chen YS, Ke HC, Wu SL, Kuo CW, Wang FS (2018) Chaperonin 60 sustains osteoblast autophagy and counteracts glucocorticoid aggravation of osteoporosis by chaperoning RPTOR. Cell Death Dis 9:938. https://doi.org/10.1038/s41419-018-0970-6
Yang YH, Li B, Zheng XF, Chen JW, Chen K, Jiang SD, Jiang LS (2014) Oxidative damage to osteoblasts can be alleviated by early autophagy through the endoplasmic reticulum stress pathway–implications for the treatment of osteoporosis. Free Radic Biol Med 77:10–20. https://doi.org/10.1016/j.freeradbiomed.2014.08.028
Huang Z, Wang Q, Zhang T, Fu Y, Wang W (2021) Hyper-activated platelet lysates prevent glucocorticoid-associated femoral head necrosis by regulating autophagy. Biomed Pharmacother 139:111711. https://doi.org/10.1016/j.biopha.2021.111711
Liu P, Cui Y, Liu M, Xiao B, Zhang J, Huang W, Zhang X, Song M, Li Y (2021) Protective effect of mitophagy against aluminum-induced MC3T3-E1 cells dysfunction. Chemosphere 282:131086. https://doi.org/10.1016/j.chemosphere.2021.131086
Wang N, Xu P, Wu R, Wang X, Wang Y, Shou D, Zhang Y (2021) Timosaponin BII improved osteoporosis caused by hyperglycemia through promoting autophagy of osteoblasts via suppressing the mTOR/NFκB signaling pathway. Free Radic Biol Med 171:112–123. https://doi.org/10.1016/j.freeradbiomed.2021.05.014
Huang L, Chen R, Liu L, Zhou Y, Chen Z (2021) Lactoferrin ameliorates pathological cardiac hypertrophy related to mitochondrial quality control in aged mice. Food Funct 12:7514–7526. https://doi.org/10.1039/d0fo03346d
Guo C, Xue H, Guo T, Zhang W, Xuan WQ, Ren YT, Wang D, Chen YH, Meng YH, Gao HL, Zhao P (2020) Recombinant human lactoferrin attenuates the progression of hepatosteatosis and hepatocellular death by regulating iron and lipid homeostasis in ob/ob mice. Food Funct 11:7183–7196. https://doi.org/10.1039/d0fo00910e
Hsu YH, Chiu IJ, Lin YF, Chen YJ, Lee YH, Chiu HW (2020) Lactoferrin contributes a renoprotective effect in acute kidney injury and early renal fibrosis. Pharmaceutics 12:434. https://doi.org/10.3390/pharmaceutics12050434
Pattingre S, Tassa A, Qu X, Garuti R, Liang XH, Mizushima N, Packer M, Schneider MD, Levine B (2005) Bcl-2 anti-apoptotic proteins inhibit Beclin1-dependent autophagy. Cell 122:927–939. https://doi.org/10.1016/j.cell.2005.07.002
Wei Y, Pattingre S, Sinha S, Bassik M, Levine B (2008) JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Mol Cell 30:678–688. https://doi.org/10.1016/j.molcel.2008.06.001
Ke D, Ji L, Wang Y, Fu X, Chen J, Wang F, Zhao D, Xue Y, Lan X, Hou J (2019) JNK1 regulates RANKL-induced osteoclastogenesis via activation of a novel Bcl-2-Beclin1-autophagy pathway. FASEB J 33:11082–11095. https://doi.org/10.1096/fj.201802597RR
Yang P, Song R, Li N, Sun K, Shi F, Liu H, Shen F, Jiang S, Zhang L, Jin Y (2020) Silica dust exposure induces autophagy in alveolar macrophages through switching Beclin1 affinity from Bcl-2 to PIK3C3. Environ Toxicol 35:758–767. https://doi.org/10.1002/tox.22910
Guo QQ, Wang SS, Zhang SS et al (2020) ATM-CHK2-Beclin 1 axis promotes autophagy to maintain ROS homeostasis under oxidative stress. EMBO J 39:e103111. https://doi.org/10.15252/embj.2019103111
Li S, Lin Z, Zheng W, Zheng L, Chen X, Yan Z, Cheng Z, Yan H, Zheng C, Guo P (2019) IL-17A inhibits autophagic activity of HCC cells by inhibiting the degradation of Bcl2. Biochem Biophys Res Commun 509:194–200. https://doi.org/10.1016/j.bbrc.2018.12.103
Funding
This work was supported by Jiangmen Science and Technology Program in Basic and Theoretical Science Research (2020-159) and The Postdoctoral Science Foundation of China (2020M672731).
Author information
Authors and Affiliations
Contributions
DK, YL and SW conceived and designed the overall experiments; DK and SW carried out experiments, analyzed data, and prepared figures; XW assisted in experimental preparation and data analysis; DKand SW wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
None of the authors disclosed potential conflicts of interest.
Research involving human and/or animal participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ke, D., Wang, X., Lin, Y. et al. Lactoferrin promotes the autophagy activity during osteoblast formation via BCL2-Beclin1 signaling. Mol Biol Rep 49, 259–266 (2022). https://doi.org/10.1007/s11033-021-06866-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06866-0