Abstract
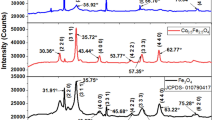
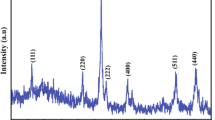
Iron oxide (α-phase) nanoparticles with coercivity larger than 300 Oe have been fabricated at a mild temperature by an environmentally benign method. The economic sodium chloride has been found to effectively serve as a solid spacer to disperse the iron precursor and to prevent the nanoparticles from agglomeration. Higher ratios of sodium chloride to iron nitrate result in smaller nanoparticles (19 nm for 20:1 and 14 nm for 50:1). The presence of polyvinyl alcohol (PVA) limits the particle growth (15 nm for 20:1 and 13 nm for 50:1) and favors nanoparticle dispersion in polymer matrices. Obvious physicochemical property changes have been observed with PVA attached to the nanoparticle surface. With PVA attached to the nanoparticle surface, the nanoparticles are found not only to increase the PVA cross-linking with an increase in melting temperature but also to enhance the thermal stability of the PVA. The nanoparticles are observed to be uniformly dispersed in the polymer matrix. Scanning electron microscopy (SEM) microstructure also shows an intermediate phase with a strong interaction between the nanoparticles and the polymer matrices, arising from the hydrogen bonding between the PVA and hydroxyl groups on the nanoparticle surface. The addition of nanoparticles favors the cross-linkage of the bulk PVA matrices, resulting in a higher melting temperature, and an enhanced thermal stability of the polymer matrix.










Similar content being viewed by others
References
Agrawal SL, Awadhia A (2004) DSC and conductivity studies on PVA based proton conducting gel electrolytes. Bull Mater Sci 27:523–527
Bercoff PG, Bertorello HR, Oliva MI (2007) Memory effect of ball-milled and annealed nanosized hematite. Physica B 398:204–207
Carrillo F, Colom X, Sunol JJ, Saurina J (2004) Structural FTIR analysis and thermal characterisation of lyocell and viscose-type fibers. Eur Polym J 40:2229–2234
Chen JP, Sorensen CM, Klabunde KJ, Hadjipanayis GC (1994) Magnetic properties of nanophase cobalt particles synthesized in inversed micelles. J Appl Phys 76:6316–6318
Chen LX, Liu T, Thurnauer MC, Csencsits R, Rajh T (2002) Fe2O3 nanoparticle structures investigated by X-ray absorption near-edge structure, surface modifications, and model calculations. J Phys Chem B 106:8539–8546
Deschenaux C, Affolter A, Magni D, Hollenstein C, Fayet P (1999) Investigations of CH4, C2H2 and C2H4 dusty RF plasmas by means of FTIR absorption spectroscopy and mass spectrometry. J Phys D 32:1876
Dhage SR, Khollam YB, Potdar HS, Deshpande SB, Bakare PP, Sainkar SR, Date SK (2002) Effect of variation of molar ratio (pH) on the crystallization of iron oxide phases in microwave hydrothermal synthesis. Mater Lett 57:457–462
Dong W, Zhu C (2002) Use of ethylene oxide in the sol-gel synthesis of a-Fe2O3 nanoparticles from Fe(III) salts. J Mater Chem 12:1676–1683
Feldmann C (2001) Preparation of nanoscale pigment particles. Adv Mater 13:1301–1303
Fukazawa M, Matuzaki H, Hara K (1993) Humidity- and gas-sensing properties with an Fe2O3 film sputtered on a porous Al2O3 film. Sens Actuat B14:521–522
Glisenti A (1998) Interaction of formic acid with Fe2O3 powders under different atmospheres: an XPS and FTIR study. J Chem Soc Faraday Trans 94:3671–3676
Guo Z, Henry LL, Palshin V, Podlaha EJ (2006a) Synthesis of poly(methyl methacrylate) stabilized colloidal zero-valence metallic nanoparticles. J Mater Chem 16:1772–1777
Guo Z, Tony P, Oyoung C, Wang Y, Hahn HT (2006b) Surface functionalized alumina nanoparticle filled polymer nanocomposites with enhanced mechanical properties. J Mater Chem 16:2800–2808
Guo Z, Liang X, Pereira T, Scaffaro R, Hahn HT (2007) CuO nanoparticle reinforced vinyl-ester resin nanocomposites: fabrication, characterization and property analysis. Compos Sci Technol 67:2036–2044
Guo Z, Lei K, Li Y, Ng HW, Hahn HT (2008a) Fabrication and characterization of iron oxide nanoparticle reinforced vinyl-ester resin nanocomposites. Compos Sci Technol 68:1513–1520
Guo Z, Lin H, Karki AB, Young DP, Hahn HT (2008b) Facile monomer stabilization approach to fabricate iron/vinyl ester resin nanocomposites. Compos Sci Technol 68:2551–2556
Guo Z, Shin K, Karki AB, Young DP, Hahn HT (2009a) Fabrication and characterization of iron oxide nanoparticles reinforced polypyrrole nanocomposites. J Nanopart Res 11:1441–1453
Guo Z, Lee SE, Kim H, Park S, Hahn HT, Karki AB, Young DP (2009b) Fabrication, characterization and microwave properties of polyurethane nanocomposites reinforced with iron oxide and barium titanate nanoparticles. Acta Mater 57:267–277
Huo L, Li Q, Zhao H, Yu L, Gao S, Zhao J (2005) Sol–gel route to pseudocubic shaped α-Fe2O3 alcohol sensor: preparation and characterization. Sens Actuat B 107:915–920
Kay A, Cesar I, Gratzel M (2006) New benchmark for water photooxidation by nanostructured α-Fe2O3 films. J Am Chem Soc 128:15714–15721
Kim G-M, Asran AS, Michler GH, Simon P, Kim J-S (2008) Electrospun PVA/HAp nanocomposite nanofibers: biomimetics of mineralized hard tissues at a lower level of complexity. Bioinspir Biomim 3:046003
Li Y, Liao H, Qian Y (1998) Hydrothermal synthesis of ultrafine α-Fe2O3 and Fe3O4 powders. Mater Res Bull 33:841–844
Li L, Chu Y, Liu Y, Dong L (2007) Template-free synthesis and photocatalytic properties of novel Fe2O3 hollow spheres. J Phys Chem C 111:2123–2127
Mansur HS, Orefice RL, Mansur AAP (2004) Characterization of poly(vinyl alcohol)/poly(ethylene glycol) hydrogels and PVA-derived hybrids by small-angle X-ray scattering and FTIR spectroscopy. Polymer 45:7193–7202
Matveev S, Portnyagin M, Ballhaus C, Brooker R, Geiger CA (2005) FTIR spectrum of phenocryst olivine as an indicator of silica saturation magmas. J Petrol 46:603–614
Mbhele ZH, Salemane MG, van Sittert CGCE, Nedeljkovic JM, Djokovic V, Luyt AS (2003) Fabrication and characterization of silver–polyvinyl alcohol nanocomposites. Chem Mater 15:5019–5024
Nassar N, Husein M (2006) Preparation of iron oxide nanoparticles from FeCl3 solid powder using microemulsions. Phys Status Solidi A 203:1324–1328
Neri G, Bonavita A, Galvagno S, Siciliano P, Capone S (2002) CO and NO2 sensing properties of doped-Fe2O3 thin films prepared by LPD. Sens Actuat B 82:40–47
Peppas NA, Merrill EW (1976) Differential scanning calorimetry of crystallized PVA hydrogels. J Appl Polym Sci 20:1457–1465
Ohmori T, Takahashi H, Mametsuka H, Suzuki E (2000) Photocatalytic oxygen evolution on α-Fe2O3 films using Fe3+ ion as a sacrificial oxidizing agent. Phys Chem Chem Phys 2:3519–3522
Okuno D, Lwase T, Shinzawa-Itoh K, Yoshikawa S, Kitagawa T (2003) FTIR detection of protonation/deprotonation of key carboxyl side chains caused by redox change of the CuA-Heme a moiety and ligand dissociation from the Heme a3-CuB center of bovine heart cytochrome c oxidase. J Am Chem Soc 125:7209–7218
Ping ZH, Nguyen QT, Chen SM, Zhou JQ, Ding YD (2001) States of water in different hydrophilic polymers: DSC and FTIR studies. Polymer 42:8461–8467
Ren Y, Ma Z, Qian L, Dai S, He H, Bruce PG (2009) Ordered crystalline mesoporous oxides as catalysts for CO oxidation. Catal Lett 131:146–154
Ricciardi R, Auriemma F, De Rosa C, Laupretre F (2004) X-ray diffraction analysis of Poly(vinyl alcohol) hydrogels, obtained by freezing and thawing techniques. Macromolecules 37:1921–1927
Rofer-Depoorter CK (1981) A comprehensive mechanism for the Fischer-Tropsch synthesis. Chem Rev 81:447–474
Rudel R, Zite-Ferenczy F (1971) Interpretation of Light Diffraction By cross-striated muscle as Bragg reflexion of light by the lattice of contractile proteins. J Physiol 290:317–330
Santra S, Tapec R, Theodoropoulou N, Dobson J, Hebard A, Tan W (2001) Synthesis and characterization of silica-coated iron oxide nanoparticles in microemulsion: the effect of nonionic surfactants. Langmuir 17:2900–2906
Talapin DV, Haubold S, Rogach AL, Kornowski A, Haase M, Weller H (2001a) A novel organometallic synthesis of highly luminescent CdTe nanocrystals. J Phys Chem B 105:2260–2263
Talapin DV, Rogach AL, Haase M, Weller H (2001b) Evolution of an ensemble of nanoparticles in a colloidal solution: theoretical Study. J Phys Chem B 105:12278–12285
Wang L-L, Jiang J-S (2007) Preparation of α-Fe2O3 nanoparticles by high-energy ball milling. Physica B 390:23–27
Wang X, Chen X, Ma X, Zheng H, Ji M, Zhang Z (2004) Low-temperature synthesis of α-Fe2O3 nanoparticles with a closed cage structure. Chem Phys Lett 384:391–393
Wang Y, Cao J, Wang S, Guo X, Zhang J, Xia H, Zhang S, Wu S (2008) Facile synthesis of porous α-Fe2O3 nanorods and their application in ethanol sensors. J Phys Chem C 112:17804–17808
Wieczorek-Ciurowa K, Kozak AJ (1999) The thermal decomposition of Fe(NO3)3·9H2O. J Therm Anal Calorim 58:647–651
Wu C, Yin P, Zhu X, OuYang C, Xie Y (2006) Synthesis of hematite (α-Fe2O3) nanorods: diameter-size and shape effects on their applications in magnetism, lithium ion battery, and gas sensors. J Phys Chem B 110:17806–17812
Wu X-L, Guo Y-G, Wan L-J, Hu C-W (2008) A-Fe2O3 nanostructures: inorganic salt-controlled synthesis and their electrochemical performance toward lithium storage. J Phys Chem C 112:16824–26829
Xie X, Li Y, Liu Z-Q, Haruta M, Shen W (2009) Low-temperature oxidation of CO catalysed by Co3O4 nanorods. Nature 458:746–749
Yu B, Zhu C, Gan F (2000) Large nonlinear optical properties of Fe2O3 nanoparticles. Physica E 8:360–364
Zhang D, Karki AB, Rutman D, Young DP, Wang A, Cocke D, Ho TH, Guo Z (2009) Electrospun polyacrylonitrile nanocomposite fibers reinforced with Fe3O4 nanoparticles: fabrication and property analysis. Polymer 50:4189–4198
Zhang Y, Chu Y, Dong L (1989) One-step synthesis and properties of urchin-like PS/α-Fe2O3 composite hollow microspheres. Nanotechnology 18:435608
Zheng Y, Cheng Y, Wang Y, Bao F, Zhou L, Wei X, Zhang Y, Zheng Q (2007) Quasicubic a-Fe2O3 nanoparticles with excellent catalytic performance. J Phys Chem B 110:3093–3097
Zhu L-P, Xiao H-M, Fu S-Y (2007) Template-free synthesis of monodispersed and single-crystalline cantaloupe-like Fe2O3 superstructures. Cryst Growth Des 7:177–182
Acknowledgments
The project is partially supported by the start-up grant and research enhancement grant from Lamar University. The authors kindly acknowledge the support from Northrop–Grumman Corporation. DPY acknowledges support from the NSF under Grant No. DMR 04-49022. FT-IR analysis done by Dr. Y. Mou from Department of Chemistry and Physics at LU and financial support from the Welch Foundation (V-1103) are kindly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, Z., Zhang, D., Wei, S. et al. Effects of iron oxide nanoparticles on polyvinyl alcohol: interfacial layer and bulk nanocomposites thin film. J Nanopart Res 12, 2415–2426 (2010). https://doi.org/10.1007/s11051-009-9802-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9802-z