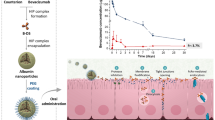
Abstract
The gold(III) complex [AuCl(L1)] has been reported as a lead candidate for Chagas’ disease (CD) treatment. However, in order to be effective, the drug must first reach the target tissue and then be delivered in therapeutic amounts. In this context, nanoparticles (NPs) of poly(lactic-co-glycolic acid) (PLGA) have been developed to be used as delivery systems for [AuCl(L1)]. The [AuCl(L1)]-PLGA-NPs were prepared via the emulsification and solvent evaporation technique and displayed encapsulation efficiency around 90% with the size range of 275 ± 5 nm, polydispersity index (PDI) around 0.115, and a higher value of zeta potential (− 6.51 ± 0.47 mV) compared to blank NPs (without gold complex), which agree with the formation of a cationic species in solution, as indicated by computational calculations. Additionally, high-resolution images obtained by SEM showed the [AuCl(L1)]-PLGA-NPs spherical shape with average sizes close to those analyzed by the DLS technique. Stability studies for the [AuCl(L1)]-PLGA-NPs pointed out that no significant changes in relation to size and PDI occur over almost 2 months of storage at 8 °C. The release of the complex from NPs was about 10% in 24 h, followed by a slow release. By means of the Korsmeyer-Peppas model, it was possible to identify the release mechanism as being through diffusion and relaxation of the polymeric matrix. Overall, it has been shown that the PLGA-NPs are promising carriers for delivery of [AuCl(L1)] and are recommended to be investigated as formulations for parasite treatment in vivo experiments.

Graphical abstract









Similar content being viewed by others
References
Arany I, Safirstein RL (2003) Cisplatin nephrotoxicity. Semin Nephrol 23:460–464. https://doi.org/10.1016/S0270-9295(03)00089-5
Bazana MT, Codevilla CF, Menezes CR (2019) Nanoencapsulation of bioactive compounds: challenges and perspectives. Curr Opin Food Sci 26:47–56. https://doi.org/10.1016/j.cofs.2019.03.005
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652. https://doi.org/10.1063/1.464913
Bohrey S, Chourasiya V, Pandey A (2016) Polymeric nanoparticles containing diazepam: preparation, optimization, characterization, in-vitro drug release and release kinetic study. Nano Converg 3(1):3. https://doi.org/10.1186/s40580-016-0061-2
Borges AP, Carneiro ZA, Prado FS, Souza JR, Furlan e Silva LH, Oliveira CG, Deflon VM, Albuquerque S, Leite NB, Machado AEH, Patrocinio AOT, Maia PIS (2018) Cu(I) complexes with thiosemicarbazides derived from p-toluenesulfohydrazide: structural, luminescence and biological studies. Polyhedron 155:170–179. https://doi.org/10.1016/j.poly.2018.08.013
Browning RJ, Reardon PJT, Parhizkar M, Pedley RB, Edirisinghe M, Knowles JC, Stride E (2017) Drug delivery strategies for platinum-based chemotherapy. ACS Nano 11:8560–8578. https://doi.org/10.1021/acsnano.7b04092
Chatelain E (2015) Chagas disease drug discovery: toward a new era. J Biomol Screen 20:22–35. https://doi.org/10.1177/1087057114550585
Chen G, Wen J (2018) Poly(lactic-co-glycolic acid) based double emulsion nanoparticle as a carrier system to deliver glutathione sublingually. J Biomed 3:50–59. https://doi.org/10.7150/jbm.27148
Danhier F, Ansorena E, Silva JM, Coco R, Breton AL, Préat V (2012) PLGA-based nanoparticles: an overview of biomedical applications. J Control Release 161:505–522. https://doi.org/10.1016/j.jconrel.2012.01.043
Dash S, Murthy PN, Nath L, Chowdhury P (2010) Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm 67:217–223
Dennington RD, Keith TA, Millam JM (2009) GaussView 5.0.8, Gaussian, Inc., Wallingford, CT, USA
Dourado Junior SM, Nunes ES, Marques RP, Rossino LS, Quites FJ, Siqueira Júnior JR, Moreto JÁ (2017) Controlled release behavior of sulfentrazone herbicide encapsulated in Ca-ALG microparticles: preparation, characterization, mathematical modeling and release tests in field trial weed control. J Mater Sci 52:9491–9507. https://doi.org/10.1007/s10853-017-1103-9
Duan X, He C, Kron SJ, Lin W (2016) Nanoparticle formulations of cisplatin for cancer therapy. Wiley Interdiscip Rev Nanomed Nanobiotechnol 8:776–791. https://doi.org/10.1002/wnan.1390
Fredenberg S, Wahlgren M, Reslow M, Axelsson A (2011) The mechanisms of drug release in poly(lactic-co-glycolic acid)-based drug delivery systems--a review. Int J Pharm 415:34–52. https://doi.org/10.1016/j.ijpharm.2011.05.049
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2015) Gaussian 09, Revision E.01. Gaussian, Inc., Wallingford
Gaglio SC, Rosa CD, Piccinelli F, Romeo A, Perduca M (2019) Complexes of rare earth ions embedded in poly(lactic-co-glycolic acid) (PLGA) nanoparticles: characterization and spectroscopic study. Opt Mater 94:249–256. https://doi.org/10.1016/j.optmat.2019.05.034
Gonçalves ACR, Carneiro ZA, Oliveira CG, Danuello A, Guerra W, Oliveira RJ, Ferreira FB, Veloso-Silva LLW, Batista FAH, Borges JC, Albuquerque S, Deflon VM, Maia PIS (2017) PtII, PdII and AuIII complexes with a thiosemicarbazone derived from diacethylmonooxime: structural analysis, trypanocidal activity, cytotoxicity and first insight into the antiparasitic mechanism of action. Eur J Med Chem 141:615–631. https://doi.org/10.1016/j.ejmech.2017.10.013
Haraguchi H (2017) Metallomics the history in the last decade and a future outlook. Metallomics 9:1001–1013. https://doi.org/10.1039/C7MT00023E
Imberti C, Sadler PJ (2020) 150 years of the periodic table: new medicines and diagnostic agents. Adv Inorg Chem 75:3–56. https://doi.org/10.1016/bs.adioch.2019.11.001
Johnstone TC, Suntharalingam K, Lippard SJ (2016) The next generation of platinum drugs: targeted Pt(II) agents, nanoparticle delivery, and Pt(IV) prodrugs. Chem Rev 116:3436–3486. https://doi.org/10.1021/acs.chemrev.5b00597
Kang X, Xiao HH, Song HQ, Jing XB, Yan LS, Qi RG (2015) Advances in drug delivery system for platinum agents based combination therapy. Cancer Biol Med 12:362–374. https://doi.org/10.7497/j.issn.2095-3941.2015.0063
Khalil NM, Nascimento TCF, Casa DM, Dalmolin LF, Mattos AC, Hoss I, Romano MA, Mainardes RM (2013) Pharmacokinetics of curcumin-loaded PLGA and PLGA–PEG blend nanoparticles after oral administration in rats. Colloids Surf B Biointerfaces 101:353–360. https://doi.org/10.1016/j.colsurfb.2012.06.024
Korsmeyer RW, Gurny R, Doelker EM, Buri P, Peppas NA (1983) Mechanism of solute release from porous hydrophilic polymers. Int J Pharm 15(1):25–35. https://doi.org/10.1016/0378-5173(83)90064-9
Lindner GR, Santos DB, Colle D, Moreira ELG, Prediger RD, Farina M, Khalil NM, Mainardes MR (2015) Improved neuroprotective effects of resveratrol-loaded polysorbate 80-coated poly(lactide) nanoparticles in MPTP-induced Parkinsonism. Nanomedicine 10:1127–1138. https://doi.org/10.2217/nnm.14.165
Lopes CD, Gaspari APS, Oliveira RJ, Abram U, Almeida JPA, Maia PIS, Albuquerque S, Carneiro ZA (2019) Organometallic gold(III) complex [Au(Hdamp)(L14)]+ (L1 = SNS-donating thiosemicarbazone) as a candidate to new formulations against Chagas disease. ACS Infect Dis 5(10):1698–1707. https://doi.org/10.1021/acsinfecdis.8b00284
Maia PIS, Deflon VM, Abram U (2014) Gold(III) complexes in medicinal chemistry. Future Med Chem 6:1515–1536. https://doi.org/10.4155/fmc.14.87
Maia PIS, Carneiro ZA, Lopes CD, Oliveira CG, Silva JS, Albuquerque S, Hagenbach A, Gust R, Deflon VM, Abram U (2017) Organometallic gold(III) complexes with hybrid SNS-donating thiosemicarbazone ligands: cytotoxicity and anti-Trypanosoma cruzi activity. Dalton Trans 46:2559–2571. https://doi.org/10.1039/c6dt04307k
Mainardes RM, Khalil NM, Gremião MPD (2010) Intranasal delivery of zidovudine by PLA and PLA–PEG blend nanoparticles. Int J Pharm 395:266–271. https://doi.org/10.1016/j.ijpharm.2010.05.020
Marini A, Muñoz-Losa A, Biancardi A, Mennucci B (2010) What is Solvatochromism? J Phys Chem B 114(51):17128–17135. https://doi.org/10.1021/jp1097487
Marquele-Oliveira F, Santana DCA, Taveira SF, Vermeulen DM, Oliveira ARM, Silva RS, Lopez RFV (2010) Development of nitrosyl ruthenium complex-loaded lipid carriers for topical administration: improvement in skin stability and in nitric oxide release by visible light irradiation. J Pharm Biomed Anal 53:843–851. https://doi.org/10.1016/j.jpba.2010.06.007
Mirakabad FST, Nejati-Koshki K, Akbarzadeh A, Yamchi MR, Milani M, Zarghami N, Zeighamian V, Rahimzadeh A, Alimohammadi S, Hanifehpour Y, Joo SW (2014) PLGA-based nanoparticles as cancer drug delivery systems. Asian Pac J Cancer Prev 15:517–535. https://doi.org/10.7314/APJCP.2014.15.2.517
Nascimento TCF, Casa DM, Dalmolin LF, Mattos AC, Khalil NM, Mainardes RM (2012) Development and validation of an HPLC method using fluorescence detection for the quantitative determination of curcumin in PLGA and PLGA-PEG nanoparticles. Curr Pharm Anal 8:324–333. https://doi.org/10.2174/157341212803341654
Oliveira CG, Maia PIS, Souza PC, Pavan FR, Leite CQF, Viana RB, Batista AA, Nascimento OR, Deflon VM (2014) Manganese(II) complexes with thiosemicarbazones as potential anti-Mycobacterium tuberculosis agents. J Inorg Biochem 132:21–29. https://doi.org/10.1016/j.jinorgbio.2013.10.011
Oliveira CG, Romero-Canelón I, Silva MM, Coverdale JPC, Maia PIS, Batista AA, Castelli S, Desideri A, Sadler PJ, Deflon VM (2019) Palladium(II) complexes with thiosemicarbazones derived from pyrene as topoisomerase IB inhibitors. Dalton Trans 48:16509–16517. https://doi.org/10.1039/c9dt02570g
Oliveira ALCSL, Santos-Silva AM, Silva-Júnior AA, Garcia VB, Araújo AA, de Geus-Oei L-F, Chan AB, Cruz LJ, Júnior RFA (2020) Cholesterol-functionalized carvedilol-loaded PLGA nanoparticles: anti-inflammatory, antioxidant, and antitumor effects. J Nanopart Res 22:115. https://doi.org/10.1007/s11051-020-04832-8
Pelosi G, Bisceglie F, Bignami F, Ronzi P, Schiavone P, Re MC, Casoli C, Pilotti E (2010) Antiretroviral activity of thiosemicarbazone metal complexes. J Med Chem 53:8765–8769. https://doi.org/10.1021/jm1007616
Prayag K, Surve DH, Paul AT, Kumar S, Jindal AB (2020) Nanotechnological interventions for treatment of trypanosomiasis in humans and animals. Drug Deliv Transl Res 10:945–961. https://doi.org/10.1007/s13346-020-00764-x
Rappoport D, Furche F (2010) Property-optimized Gaussian basis sets for molecular response calculations. J Chem Phys 133:134105. https://doi.org/10.1063/1.3484283
Renfrew AK (2014) Transition metal complexes with bioactive ligands: mechanisms for selective ligand release and applications for drug delivery. Metallomics 6:1324–1335. https://doi.org/10.1039/c4mt00069b
Rettondin AR, Carneiro ZA, Gonçalves ACR, Ferreira VF, Oliveira CG, Lima AN, Oliveira RJ, Albuquerque S, Deflon VM, Maia PIS (2016) Gold(III) complexes with ONS-Tridentate thiosemicarbazones: toward selective trypanocidal drugs. Eur J Med Chem 120:217–226. https://doi.org/10.1016/j.ejmech.2016.05.003
Siepmann J, Peppas NA (2001) Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Adv Drug Deliv Rev 48:139–157. https://doi.org/10.1016/S0169-409X(01)00112-0
Slade GG, Oliveira RJ, Moreto JA (2019) Developing a mathematical model for the controlled release over time of sulfentrazone herbicide from biodegradable polymer. Mater Res 22(5):e20190306. https://doi.org/10.1590/1980-5373-mr-2019-0306
Souza JG, Dias K, Pereira TA, Bernardi DS, Lopez RFV (2013) Topical delivery of ocular therapeutics: carrier systems and physical methods. J Pharm Pharmacol 66:507–530. https://doi.org/10.1111/jphp.12132
Stephens PJ, Devlin JF, Chabalowski CF, Frisch MJ (1994) Ab initio calculation of vibrational absorption and circular Dichroism spectra using density functional force fields. J Chem Phys 98:11623–11627. https://doi.org/10.1021/j100096a001
Tomasi J, Mennucci B, Cancès E (1999) The IEF version of the PCM solvation method: an overview of a new method addressed to study molecular solutes at the QM ab initio level. J Mol Struct 464:211–226. https://doi.org/10.1016/S0166-1280(98)00553-3
Weigend F, Ahlrichs R (2005) Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys Chem Chem Phys 7:3297–3305. https://doi.org/10.1039/b508541a
Xian TS, Onn WJ, Misran M, Ali HM (2016) Encapsulation of platinum complex of indole-7-carbaldehyde thiosemicarbazone, Pt(L)(PPh3) into nanolipid carrier for sustain released anti-cancer treatment. Mater Today 3:635–639. https://doi.org/10.1016/j.matpr.2016.01.102
Yanai T, Tew DP, Handy NC (2004) A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem Phys Lett 393:51–57. https://doi.org/10.1016/j.cplett.2004.06.011
Zheng YR, Suntharalingam K, Johnstone TC, Yoo H, Lin W, Brooks JG, Lippard SJ (2014) Pt(IV) prodrugs designed to bind non-covalently to human serum albumin for drug delivery. J Am Chem Soc 136:8790–8798. https://doi.org/10.1021/ja5038269
Zhong Y, Jia C, Zhang X, Liao X, Yang B, Cong Y, Pu S, Gao C (2020) Targeting drug delivery system for platinum(IV)-based antitumor complexes. Eur J Med Chem 194:112229. https://doi.org/10.1016/j.ejmech.2020.112229
Funding
The authors received financial support through the grants from the National Counsel of Technological and Scientific Development (CNPq) (Grants: 402142/2016-0, 303659/2019-0, 305432/2017-6, and 424095/2018-1), FAPEMIG (Grants: APQ-03174-18, APQ-02276-18, and CEX APQ-03017-16), and FAPESP.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• The behavior of the gold(III) thiosemicarbazonate complex [AuCl(L1)] in solution was studied both experimentally and by computational methods;
• [AuCl(L1)] was efficiently encapsulated into PLGA nanoparticles with a high drug loading efficiency;
• [AuCl(L1)]-PLGA-NPs obtained showed suitable physicochemical properties such as small size, a narrow size distribution, and physical stability;
• Scanning electron microscope micrographs confirmed the formation of spherical nanoparticles and their homogeneity;
• The in vitro release study showed a slow and sustained release of [AuCl(L1)]-PLGA-NPs governed by diffusion and relaxation of the polymeric matrix.
Electronic supplementary material
ESM 1
(DOCX 1614 kb).
Rights and permissions
About this article
Cite this article
Silva, R.T.C., Dalmolin, L.F., Moreto, J.A. et al. Development of gold(III) thiosemicarbazonate complex–loaded PLGA nanoparticles: characterization and sustained release studies. J Nanopart Res 22, 339 (2020). https://doi.org/10.1007/s11051-020-05064-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-020-05064-6