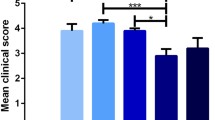
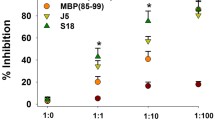
Objective. To study the efficacy of release-active forms of antibody to S100 protein in an animal model of multiple sclerosis. Materials and methods. The study was performed on 60 female rats aged 12 weeks. Experimental allergic encephalomyelitis was induced by administration of spinal cord homogenate at the base of the tail. Female rats then received intragastric solution of release-active antibodies to S100 protein (Tenoten) at a dose of 2.5 ml/kg/day or distilled water for 30 days. Positive controls received i.m. injections of glatiramer acetate at a dose of 4 mg/kg/day (Copaxone). Results and discussion. Administration of release-active forms of antibody to S100 increased the latent period of disease onset, decreased in peak disease intensity, and compensated for weight loss in the animals, as compared with controls. The effect of the agents was comparable with that of the Copaxone group.
Similar content being viewed by others
References
R. Aharoni, “New findings and old controversies in the research of multiple sclerosis and its model experimental autoimmune encephalomyelitis,” Expert Rev. Clin. Immunol., 9, No. 5, 423–440 (2013), doi: 10.1586/eci.13.21.
E. G. Gilerovich, E. A. Fedorova, I. N. Abdurasulova, et al., “Analysis of the morphological signs of inflammatory reactions in the spinal cord of Wistar rats in an experimental model,” Morfologiya, 138, No. 5, 16–20 (2010).
S. Sinha, S. Subramanian, L. Miller, et al., “Cytokine switch and bystander suppression of autoimmune responses to multiple antigens in experimental autoimmune encephalomyelitis by a single recombinant T-cell receptor ligand,” Neuroscience, 29, No. 1, 3816–3823 (2009), doi: 10.1523/jneurosci.5812-08.2009.
A. V. Pozdnyakov, I. N. Abdurasulova, and L. N. Prakhova, “Proton magnetic resonance spectroscopy in studies of the metabolic mechanisms of demyelinating process in experimental allergic encephalomyelitis,” Zh. Luch. Diagnost. Ter., 2, No. 1, 30–36 (2010).
R. Donato, “Intracellular and extracellular roles of S100 proteins,” Microsc. Res. Tech., 60, No. 6, 540–551 (2003), doi: 10.1002/jemt.10296.
D. B. Zimmer, E. H. Cornwall, A. Landar, and W. Song, “The S100 protein family: history, function and expression,” Brain Res. Bull., 37, 417–429 (1995); 10.1016/0361-9230(95)00040-2.
D. F. de Souza, K. Wartchow, F. Hansen, et al., “Interleukin-6-induced S100B secretion is inhibited by haloperidol and risperidone,” Prog. Neuropsychopharmacol. Biol. Psychiatry, 43,13–22 (2013), doi: 10.1016/j.pnpbp.2012.12.001.
C. Adami, G. Sorci, E. Blasi, et al., “S100B expression in and effects on microglia,” Glia, 33, No. 2, 131–142 (2001), doi: 10.1002/1098-1136 (200102)33:2<131::aid-glia1012>3.3.co;2-4.
G. Esposito, D. DeFilippis, C. Cirillo, et al., “The astroglial-derived S100beta protein stimulates the expression of nitric oxide synthase in rodent macrophages through p38 MAP kinase activation,” Life Sci., 78, No. 23, 2707–2715 (2006), doi: 10.1016/j.lfs.2005.10.023.
J. Hu and L. J. Can Elkik, “Glial-derived proteins activate cultured astrocytes and enhance beta amyloid-induced glial activation,” Brain Res., 842, No. 1, 46–54 (1999), doi: 10.1016/s0006-8993(99) 01804-1.
U. Missler, K. P. Wandinger, M. Wiesmann, et al., “Acute exacerbation of multiple sclerosis increases plasma levels of S-100 protein,” Acta Neurol. Scand., 96, 142–144 (1997), doi: 10.1111/j.1600-0404. 1997.tb00256.x.
K. Mitosek-Szewczyk, W. Gordon-Krajcer, D. Flis, and Z. Stelmasiak, “Some markers of neuronal damage in cerebrospinal fluid of multiple sclerosis patients in relapse,” Folia Neuropathol., 49, No. 3, 191–196 (2011).
E. I. Gusev, I. A. Belyaeva, V. P. Chekhonin, and T. L. Demina, “Clinical-immunochemical characteristics of the remitting course of multiple sclerosis,” Vestn. Ros. Akad. Med. Nauk., No. 7, 40–45 (1999).
K. Kojima, H. Wekerle, H. Lassmann, et al., “Induction of experimental autoimmune encephalomyelitis by CD4+ T cells specific for an astrocyte protein, S100 beta,” J. Neural Transm. Suppl., 9, 43–51 (1997), doi: 10.1007/978-3-7091-6844-8_5.
O. I. Epshtein, “The phenomenon of release-activity and the ‘spatial homeostasis hypothesis’,” Usp. Fiziol. Nauk., 44, No. 3, 54–76 (2013).
Yu. Yu. Orlov, V. M. Alifirova, N. V. Cherdyntseva, et al., “Results of a three-year clinical-immunological observations of patients with multiple sclerosis receiving Copaxone,” Zh. Nevrol. Psikhiat., 105, No. 5, 23–27 (2005).
T. E. Shmidt, I. G. Churkina, I. A. Elagina, and N. N. Yakhno, “The course of remitting multiple sclerosis on the background of treatment with Copaxone and after its withdrawal,” Nevrol. Zh., 10, No. 3, 23–28 (2005).
I. N. Abdurasulova, Yu. L. Zhitnukhin, E. A. Tarasova, and V. M. Klimenko, “Expression of cytokine mRNAs in the spleen and spinal cord in rats with EAE of different severities,” Med. Immunol., 6, No. 1–2, 37–46 (2004).
Yu. L. Zhitnukhin, I. N. Abdurasulova, E. A. Tarasova, et al., “Features of the dynamics of circulating and expressible cytokines in the induction of experimental allergic encephalomyelitis,” Med. Immunol., 10, No. 2–3, 193–202 (2008).
N. B. Serebryanaya, M. N. Karpenko, Yu. L. Zhitnukhin, and I. N. Abdurasulova, “Studies of the protective action of Ferrovir in acute experimental allergic encephalomyelitis,” Zh. Tsitok Vospal., 9, No. 1, 33–39 (2010).
M. D. Carruthers, “ Update on disease-modifying treatments for multiple sclerosis,” Clin. Ther., 36, No. 12, 1938–1945 (2014), doi: 10.1016/j.clinthera.2014.08.006.
J. Steiner, N. Marquardt, I. Pauls, et al., “Human CD8+ T cells and NK cells express and secrete S100B upon stimulation,” Brain Behav. Immun., 25, 1233–1241 (2011), doi: 10.1016/j.bbi.2011.03.015.
T. Petrova, J. Hu, and L. J. Van Eldik, “Modulation of glial activation by astrocyte-derived protein S100B: differential responses of astrocyte and microglial cultures,” Brain Res., 853, 74–80 (2000), doi: 10.1016/s0006-8993(99)02251-9.
C. Adami, R. Bianchi, G. Pula, and R. Donato, “S100B-stimulated NO production by BV-2 microglia is independent of RAGE transducing activity but dependent on RAGE extracellular domain,” Biochim. Biophys. Acta, 1742, 169–177 (2004), doi: 10.1016/j.bbamcr.2004.09.008.
R. Donato, G. Sorci, F. Riuzzi, et al., “S100’s double life: intracellular regulator and extracellular signal,” Biochim. Biophys. Acta, 1793, 1008–1022 (2009), doi: 10.1016/j.bbamcr.2008.11.009.
C. Reali, R. Pillai, F. Saba, et al., “S100B modulates growth factors and costimulatory molecules expression in cultured human astrocytes,” J. Neuroimmunol., 243, 95–99 (2012), doi: 10.1016/j.jneuroim.2011.11.011.
D. S. Goncalves, G. Lenz, J. Karl, et al., “Extracellular S100B protein modulates ERK in astrocyte cultures,” Neuroreport, 11, No. 4, 807–809 (2000), doi: 10.1097/00001756-200003200-00030.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Nevrologii i Psikhiatrii imeni S. S. Korsakova, Vol. 115, No. 6, Iss. 1, pp. 78–82, June, 2015.
Rights and permissions
About this article
Cite this article
Ganina, K.K., Dugina, Y.L., Zhavbert, K.S. et al. Release-Active Antibodies to S100 Protein Can Correct the Course of Experimental Allergic Encephalomyelitis. Neurosci Behav Physi 47, 163–167 (2017). https://doi.org/10.1007/s11055-016-0380-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-016-0380-0