Abstract
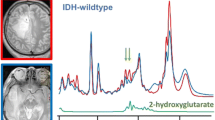
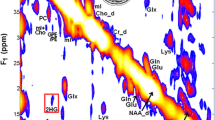
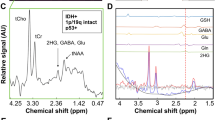
Mutations of the isocitrate dehydrogenase 1 and 2 genes (IDH1 and IDH2) are commonly found in primary brain cancers. We previously reported that a novel enzymatic activity of these mutations results in the production of the putative oncometabolite, R(−)-2-hydroxyglutarate (2-HG). Here we investigated the ability of magnetic resonance spectroscopy (MRS) to detect 2-HG production in order to non-invasively identify patients with IDH1 mutant brain tumors. Patients with intrinsic glial brain tumors (n = 27) underwent structural and spectroscopic magnetic resonance imaging prior to surgery. 2-HG levels from MRS data were quantified using LC-Model software, based upon a simulated spectrum obtained from a GAMMA library added to the existing prior knowledge database. The resected tumors were then analyzed for IDH1 mutational status by genomic DNA sequencing, Ki-67 proliferation index by immunohistochemistry, and concentrations of 2-HG and other metabolites by liquid chromatography–mass spectrometry (LC–MS). MRS detected elevated 2-HG levels in gliomas with IDH1 mutations compared to those with wild-type IDH1 (P = 0.003). The 2-HG levels measured in vivo with MRS were significantly correlated with those measured ex vivo from the corresponding tumor samples using LC–MS (r 2 = 0.56; P = 0.0001). Compared with wild-type tumors, those with IDH1 mutations had elevated choline (P = 0.01) and decreased glutathione (P = 0.03) on MRS. Among the IDH1 mutated gliomas, quantitative 2-HG values were correlated with the Ki-67 proliferation index of the tumors (r 2 = 0.59; P = 0.026). In conclusion, water-suppressed proton (1H) MRS provides a non-invasive measure of 2-HG in gliomas, and may serve as a potential biomarker for patients with IDH1 mutant brain tumors. In addition to 2-HG, alterations in several other metabolites measured by MRS correlate with IDH1 mutation status.





Similar content being viewed by others
References
Balss J, Meyer J, Mueller W, Korshunov A, Hartmann C, von Deimling A (2008) Analysis of the IDH1 codon 132 mutation in brain tumors. Acta Neuropathol 116(6):597–602
Bleeker FE, Lamba S, Leenstra S, Troost D, Hulsebos T, Vandertop WP et al (2009) IDH1 mutations at residue p.R132 (IDH1(R132)) occur frequently in high-grade gliomas but not in other solid tumors. Hum Mutat 30(1):7–11
De Carli E, Wang X, Puget S (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360(21):2248 author reply 9
Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P et al (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321(5897):1807–1812
Yan H, Parsons DW, Jin G, McLendon R, Rasheed BA, Yuan W et al (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360(8):765–773
Watanabe T, Nobusawa S, Kleihues P, Ohgaki H (2009) IDH1 mutations are early events in the development of astrocytomas and oligodendrogliomas. Am J Pathol 174(4):1149–1153
Ducray F, Marie Y, Sanson M (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360(21):2248 author reply 9
Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD et al (2010) Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 17(1):98–110
Dubbink HJ, Taal W, van Marion R, Kros JM, van Heuvel I, Bromberg JE et al (2009) IDH1 mutations in low-grade astrocytomas predict survival but not response to temozolomide. Neurology 73(21):1792–1795
Hartmann C, Meyer J, Balss J, Capper D, Mueller W, Christians A et al (2009) Type and frequency of IDH1 and IDH2 mutations are related to astrocytic and oligodendroglial differentiation and age: a study of 1, 010 diffuse gliomas. Acta Neuropathol 118(4):469–474
Dang L, White DW, Gross S, Bennett BD, Bittinger MA, Driggers EM et al (2009) Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462(7274):739–744
Aghili M, Zahedi F, Rafiee E (2009) Hydroxyglutaric aciduria and malignant brain tumor: a case report and literature review. J Neurooncol 91(2):233–236
Bottomley PA (1987) Spatial localization in NMR spectroscopy in vivo. Ann N Y Acad Sci 508:333–348
Ogg RJ, Kingsley PB, Taylor JS (1994) WET, a T1- and B1-insensitive water-suppression method for in vivo localized 1H NMR spectroscopy. J Magn Reson B 104(1):1–10
Provencher SW (1993) Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magn Reson Med 30(6):672–679
Smith SA, Levante TO, Meier BH, Ernst RR (1994) Computer simulations in magnetic resonance.An object oriented programming approach. J Magn Reson A106:75–105
Helms G (2008) The principles of quantification applied to in vivo proton MR spectroscopy. Eur J Radiol 67(2):218–229
Cavassila S, Deval S, Huegen C, van Ormondt D, Graveron-Demilly D (2001) Cramer-Rao bounds: an evaluation tool for quantitation. NMR Biomed 14(4):278–283
Reitman ZJ, Jin G, Karoly ED, Spasojevic I, Yang J, Kinzler KW et al (2011) Profiling the effects of isocitrate dehydrogenase 1 and 2 mutations on the cellular metabolome. Proc Natl Acad Sci U S A 108(8):3270–3275
Jin G, Reitman ZJ, Spasojevic I, Batinic-Haberle I, Yang J, Schmidt-Kittler O et al (2011) 2-Hydroxyglutarate production, but not dominant negative function, is conferred by glioma-derived NADP-dependent isocitrate dehydrogenase mutations. PLoS One 6(2):e16812
Chou WC, Hou HA, Chen CY, Tang JL, Yao M, Tsay W et al (2010) Distinct clinical and biologic characteristics in adult acute myeloid leukemia bearing the isocitrate dehydrogenase 1 mutation. Blood 115(14):2749–2754
Gross S, Cairns RA, Minden MD, Driggers EM, Bittinger MA, Jang HG et al (2010) Cancer-associated metabolite 2-hydroxyglutarate accumulates in acute myelogenous leukemia with isocitrate dehydrogenase 1 and 2 mutations. J Exp Med 207(2):339–344
Ward PS, Patel J, Wise DR, Abdel-Wahab O, Bennett BD, Coller HA et al (2010) The common feature of leukemia-associated IDH1 and IDH2 mutations is a neomorphic enzyme activity converting alpha-ketoglutarate to 2-hydroxyglutarate. Cancer Cell 17(3):225–234
Martinez-Bisbal MC, Celda B (2009) Proton magnetic resonance spectroscopy imaging in the study of human brain cancer. Q J Nucl Med Mol Imaging 53(6):618–630
Tedeschi G, Lundbom N, Raman R, Bonavita S, Duyn JH, Alger JR et al (1997) Increased choline signal coinciding with malignant degeneration of cerebral gliomas: a serial proton magnetic resonance spectroscopy imaging study. J Neurosurg 87(4):516–524
Dowling C, Bollen AW, Noworolski SM, McDermott MW, Barbaro NM, Day MR et al (2001) Preoperative proton MR spectroscopic imaging of brain tumors: correlation with histopathologic analysis of resection specimens. AJNR Am J Neuroradiol 22(4):604–612
Labussiere M, Wang XW, Idbaih A, Ducray F, Sanson M (2010) Prognostic markers in gliomas. Future oncology (London, England) 6(5):733–739
Toedt G, Barbus S, Wolter M, Felsberg J, Tews B, Blond F et al (2011) Molecular signatures classify astrocytic gliomas by IDH1 mutation status. Int J Cancer 128(5):1095–1103
van den Bent MJ, Dubbink HJ, Marie Y, Brandes AA, Taphoorn MJ, Wesseling P et al (2010) IDH1 and IDH2 mutations are prognostic but not predictive for outcome in anaplastic oligodendroglial tumors: a report of the European Organization for Research and Treatment of Cancer Brain Tumor Group. Clin Cancer Res 16(5):1597–1604
Dang L, Jin S, Su SM (2010) IDH mutations in glioma and acute myeloid leukemia. Trends Mol Med 16(9):387–397
Wiseman H, Halliwell B (1996) Damage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochem J 313(Pt 1):17–29
Xu W, Yang H, Liu Y, Yang Y, Wang P, Kim S-H et al (2011) Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of [alpha]-ketoglutarate-dependent dioxygenases. Cancer Cell 19(1):17–30
Acknowledgments
Supported in part by grants from the National Cancer Institute (K01-CA111402 and R01-CA123396 to Dr. Prins; R01-CA112358 and R01-CA121131 to Dr. Liau), and research grants from the Neidorf Family Foundation, the Phillip R. Jonsson & Kenneth A. Jonsson Foundations, the Brad Kaminsky Foundation, and the Accelerate Brain Cancer Cure Foundation. Scientific support of Mr. Brian Burns is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Whitney B. Pope, Robert M. Prins, and M. Albert Thomas contributed equally to this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pope, W.B., Prins, R.M., Albert Thomas, M. et al. Non-invasive detection of 2-hydroxyglutarate and other metabolites in IDH1 mutant glioma patients using magnetic resonance spectroscopy. J Neurooncol 107, 197–205 (2012). https://doi.org/10.1007/s11060-011-0737-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-011-0737-8