Abstract
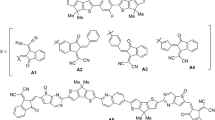
Herein current research work, we designed four new acceptor materials for small solar cell molecules with Naphthalene Di-Imide central unit by employing the wB97xd/6-31 G (d,p) and TD-wB97xd/6-31 G (d,p) level of density functional theories. Absorption properties of designed materials are excellent between the 400 nm to 510 nm with chloroform solvent and 340 nm to 490 nm in gas phase, small reorganization energy values 0.0163–0.0172 eV for electron (λe) and 0.0205–0.0257 eV for hole transfer (λh), large expected open circuit voltages (Voc) from 3.60 to 4.53 eV with respect to [6, 6]-phenyl-C61-butyric acid methyl ester (PCBM), high dipole moment strength ranging from 4.0199 to 8.9647 Debye in excited state and 3.3847 Debye to 7.2632 Debye in ground state which are very helpful for the further construction of organic solar cell (OSC) devices with improved and better power conversion efficiencies (PCEs).










Similar content being viewed by others
References
Ajmal, M., Ali, U., Javed, A., Tariq, A., Arif, Z., Iqbal, J., Shoaib, M., Ahmed, T.: Designing indaceno thiophene-based three new molecules containing non-fullerene acceptors as strong electron withdrawing groups with DFT approaches. J. Mol. Model. 25(10), 311 (2019)
Ali, U., Ans, M., Iqbal, J., Iqbal, M.A., Shoaib, M.: Benchmark study of benzamide derivatives and four novel theoretically designed (L1, L2, L3, and L4) ligands and evaluation of their biological properties by DFT approaches. J. Mol. Model. 25(8), 223 (2019a)
Ali, U., Javed, A., Tallat, A., Iqbal, J., Raza, A.: Molecular designing of four high performance pyrazine-based non-fullerene acceptor materials with naphthalene diimide-based small organic solar cells. J. Mol. Model. 25(2), 50 (2019b)
Cnops, K., Rand, B.P., Cheyns, D., Verreet, B., Empl, M.A., Heremans, P.: 8.4% efficient fullerene-free organic solar cells exploiting long-range exciton energy transfer. Nat. Commun. 5, 3406 (2014)
Deibel, C., Strobel, T., Dyakonov, V.: Role of the charge transfer state in organic donor–acceptor solar cells. Adv. Mater. 22, 4097–4111 (2010)
Dou, L., You, J., Yang, J., Chen, C.-C., He, Y., Murase, S., Moriarty, T., Emery, K., Li, G., Yang, Y.: Tandem polymer solar cells featuring a spectrally matched low-bandgap polymer. Nat. Photonics 6, 180–185 (2012)
Earmme, T., Hwang, Y.-J., Murari, N.M., Subramaniyan, S., Jenekhe, S.A.: All-polymer solar cells with 3.3% efficiency based on naphthalene diimide-selenophene copolymer acceptor. J. Am. Chem. Soc. 135, 14960–14963 (2013)
Gaussian, R.A. Frisch, M.J., Trucks, G.W., Schlegel, H.B., Scuseria, G.E., Robb, M.A., Cheeseman, J.R., Scalmani, G., Barone, V., Mennucci, B., Petersson G.A., et al.: (Gaussian, Inc., Wallingford CT, 2009)
Gélinas, S., Rao, A., Kumar, A., Smith, S.L., Chin, A.W., Clark, J., van der Poll, T.S., Bazan, G.C., Friend, R.H.: Ultrafast long-range charge separation in organic semiconductor photovoltaic diodes. Science 343, 512–516 (2014)
Hickey, A.L., Rowley, C.N.: Benchmarking quantum chemical methods for the calculation of molecular dipole moments and polarizabilities. J. Phys. Chem. A 118, 3678–3687 (2014)
Holliday, S., Ashraf, R.S., Wadsworth, A., Baran, D., Yousaf, S.A., Nielsen, C.B., Tan, C.-H., Dimitrov, S.D., Shang, Z., Gasparini, N.: High-efficiency and air-stable P3HT-based polymer solar cells with a new non-fullerene acceptor. Nat. Commun. 7, 11585 (2016)
Hwang, Y.-J., Earmme, T., Courtright, B.A., Eberle, F.N., Jenekhe, S.A.: n-Type semiconducting naphthalene diimide-perylene diimide copolymers: controlling crystallinity, blend morphology, and compatibility toward high-performance all-polymer solar cells. J. Am. Chem. Soc. 137, 4424–4434 (2015)
Jungsuttiwong, S., Tarsang, R., Sudyoadsuk, T., Promarak, V., Khongpracha, P., Namuangruk, S.: Theoretical study on novel double donor-based dyes used in high efficient dye-sensitized solar cells: the application of TDDFT study to the electron injection process. Org. Electron. 14, 711–722 (2013)
Kaltenbrunner, M., White, M.S., Głowacki, E.D., Sekitani, T., Someya, T., Sariciftci, N.S., Bauer, S.: Ultrathin and lightweight organic solar cells with high flexibility. Nat. Commun. 3, 770 (2012)
Kim, J.H., Moon, K.J., Kim, J.M., Lee, D., Kim, S.H.: Effects of various light-intensity and temperature environments on the photovoltaic performance of dye-sensitized solar cells. Sol. Energy 113, 251–257 (2015)
Kooistra, F.B., Knol, J., Kastenberg, F., Popescu, L.M., Verhees, W.J., Kroon, J.M., Hummelen, J.C.: Increasing the open circuit voltage of bulk-heterojunction solar cells by raising the LUMO level of the acceptor. Org. Lett. 9, 551–554 (2007)
Koster, L., Shaheen, S.E., Hummelen, J.C.: Pathways to a new efficiency regime for organic solar cells. Advanced Energy Materials 2, 1246–1253 (2012)
Li, Y.: Molecular design of photovoltaic materials for polymer solar cells: toward suitable electronic energy levels and broad absorption. Acc. Chem. Res. 45, 723–733 (2012)
Lin, Y., Wang, J., Zhang, Z.G., Bai, H., Li, Y., Zhu, D., Zhan, X.: An electron acceptor challenging fullerenes for efficient polymer solar cells. Adv. Mater. 27, 1170–1174 (2015)
Manzoor, F., Iqbal, J., Zara, Z., Eliasson, B., Mahr, M.S., Ayub, K.: Theoretical calculations of the optical and electronic properties of dithienosilole-and dithiophene-based donor materials for organic solar cells. Chem. Select 3, 1593–1601 (2018)
McNeill, C.R., Greenham, N.C.: Conjugated-polymer blends for optoelectronics. Adv. Mater. 21, 3840–3850 (2009)
Meng, D., Sun, D., Zhong, C., Liu, T., Fan, B., Huo, L., Li, Y., Jiang, W., Choi, H., Kim, T.: High-performance solution-processed non-fullerene organic solar cells based on selenophene-containing perylene bisimide acceptor. J. Am. Chem. Soc. 138, 375–380 (2015)
Meng, G., Shi, Y., Song, X., Ji, M., Xue, Y., Hao, C.: Theoretical insight into the carrier mobility anisotropy of hole transport material Spiro-OMeTAD. Curr. Appl. Phys. 17, 1316–1322 (2017)
Minaev, B., Minaeva, V., Baryshnikov, G., Girtu, M., Agren, H.: Theoretical study of vibration spectra of sensitizing dyes for photoelectrical converters based on ruthenium (II) and iridium (III) complexes. Russ. J. Appl. Chem. 82, 1211–1221 (2009)
Minaev, B.F., Baryshnikov, G.V., Minaeva, V.A.: Electronic structure and spectral properties of the triarylamine-dithienosilole dyes for efficient organic solar cells. Dyes Pigm. 92, 531–536 (2012)
Mishra, A., Bäuerle, P.: Small molecule organic semiconductors on the move: promises for future solar energy technology. Angew. Chem. Int. Ed. 51, 2020–2067 (2012)
Nakano, K., Nakano, M., Xiao, B., Zhou, E., Suzuki, K., Osaka, I., Takimiya, K., Tajima, K.: Naphthodithiophene diimide-based copolymers: ambipolar semiconductors in field-effect transistors and electron acceptors with near-infrared response in polymer blend solar cells. Macromolecules 49, 1752–1760 (2016)
Nazeeruddin, M.K., Baranoff, E., Grätzel, M.: Dye-sensitized solar cells: a brief overview. Sol. Energy 85, 1172–1178 (2011)
Nielsen, C.B., Holliday, S., Chen, H.-Y., Cryer, S.J., McCulloch, I.: Non-fullerene electron acceptors for use in organic solar cells. Acc. Chem. Res. 48, 2803–2812 (2015)
O’regan, B., Grätzel, M.: A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353, 737–740 (1991)
Odobel, F., Pellegrin, Y., Gibson, E.A., Hagfeldt, A., Smeigh, A.L., Hammarström, L.: Recent advances and future directions to optimize the performances of p-type dye-sensitized solar cells. Coord. Chem. Rev. 256, 2414–2423 (2012)
Refaely-Abramson, S., Baer, R., Kronik, L.: Fundamental and excitation gaps in molecules of relevance for organic photovoltaics from an optimally tuned range-separated hybrid functional. Phys. Rev. B 84, 075144 (2011)
Scharber, M.C., Sariciftci, N.S.: Efficiency of bulk-heterojunction organic solar cells. Prog. Polym. Sci. 38, 1929–1940 (2013)
Sung, M.J., Huang, M., Moon, S.H., Lee, T.H., Park, S.Y., Kim, J.Y., Kwon, S.-K., Choi, H., Kim, Y.-H.: Naphthalene diimide-based small molecule acceptors for fullerene-free organic solar cells. Sol. Energy 150, 90–95 (2017)
Vandewal, K., Gadisa, A., Oosterbaan, W.D., Bertho, S., Banishoeib, F., Van Severen, I., Lutsen, L., Cleij, T.J., Vanderzande, D., Manca, J.V.: The relation between open-circuit voltage and the onset of photocurrent generation by charge-transfer absorption in polymer: fullerene bulk heterojunction solar cells. Adv. Funct. Mater. 18, 2064–2070 (2008)
Wongcharee, K., Meeyoo, V., Chavadej, S.: Dye-sensitized solar cell using natural dyes extracted from rosella and blue pea flowers. Sol. Energy Mater. Sol. Cells 91, 566–571 (2007)
Wu, Y., Zhu, W.: Organic sensitizers from D–π–A to D-A–π–A: effect of the internal electron-withdrawing units on molecular absorption, energy levels and photovoltaic performances. Chem. Soc. Rev. 42, 2039–2058 (2013)
Xu, J., Wang, L., Liang, G., Bai, Z., Wang, L., Xu, W., Shen, X.: Conjugate spacer effect on molecular structures and absorption spectra of triphenylamine dyes for sensitized solar cells: density functional theory calculations. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 78, 287–293 (2011)
Zhang, S., Ye, L., Hou, J.: Breaking the 10% efficiency barrier in organic photovoltaics: morphology and device optimization of well-known PBDTTT polymers. Adv. Energy Mater. 6, 1502529 (2016)
Zhao, J., Li, Y., Yang, G., Jiang, K., Lin, H., Ade, H., Ma, W., Yan, H.: Efficient organic solar cells processed from hydrocarbon solvents. Nat. Energy 1, 15027 (2016a)
Zhao, W., Qian, D., Zhang, S., Li, S., Inganäs, O., Gao, F., Hou, J.: Fullerene-free polymer solar cells with over 11% efficiency and excellent thermal stability. Adv. Mater. 28, 4734–4739 (2016b)
Zhao, W., Li, S., Yao, H., Zhang, S., Zhang, Y., Yang, B., Hou, J.: Molecular optimization enables over 13% efficiency in organic solar cells. J. Am. Chem. Soc. 139, 7148–7151 (2017)
Acknowledgements
These computations/simulations/[SIMILAR] analysis performed supported by the Punjab Bio-Energy Institute (PBI), Faisalabad, University of Agriculture, Faisalabad (UAF), 38040, Pakistan and Taishan Scholars Project of Shandong Province (ts201712011).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ali, U., Javed, A., Amanullah et al. Designing difluoro substituted benzene ring based fullerene free acceptors for small Naphthalene Di-Imide based molecules with DFT approaches. Opt Quant Electron 51, 332 (2019). https://doi.org/10.1007/s11082-019-2047-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11082-019-2047-x