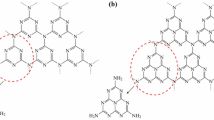
Abstract
Synthesis of a novel magnetic 1-naphthalenesulfonic acid-grafted graphene oxide (Fe3O4-GO-naphthalene-SO3H) via a three-step procedure has been described. The structure of this newly synthesized nanographene oxide was fully characterized by X-ray diffraction, energy dispersive X‐ray, vibrating sample magnetometer, scanning electron microscopy, Fourier-transform infrared, Raman spectroscopy, and thermo-gravimetric analytical techniques. The catalytic efficiency of these nanoparticles as recyclable nanocatalyst was explored in one-pot three-component reaction between aldehydes, malononitrile, and 4-hydroxyquinolin-2(1H)-one or 4-hydroxycoumarin under green conditions in water for the synthesis of 2-amino-3-cyano-1,4,5,6-tetrahydropyrano[3,2-c]quinolin-5-ones and 5-oxo-dihydropyrano[3,2-c]chromenes (coumarins) respectively. High yields of the products, low reaction times, easy preparation of the catalyst, and use of water as green solvent are the main advantages of this protocol. In addition, the catalyst can be easily separated simply by using a magnet and reused for six fresh runs without significant loss of activity.
Graphical abstract














Similar content being viewed by others
References
R. Wohlgemuth, New Biotechnol. 25, 204 (2009)
R. Ballini, F. Bigi, M.L. Conforti, D.D. Santis, R. Maggi, G. Oppici, G. Sartori, Catal. Today 60, 305 (2000)
A. Dömling, I. Ugi, Angew. Chem. Int. Ed. 39, 3168 (2000)
S.Y. Wei, Q. Wang, J.H. Zhu, L.Y. Sun, H.F. Lin, Z.H. Guo, Nanoscale. 3, 4474 (2011)
C.Y. Li, C. Ma, F. Wang, Z.J. Xi, Z.F. Wang, Y. Deng, N.Y. He, J. Nanosci. Nanotechnol. 12, 2964 (2012)
G. Li, Z. Tang, Nanoscale. 6, 3995 (2014)
R.G. Chaudhuri, S. Paria, Chem. Rev. 112, 2373 (2012)
M.J. Yao, Z. Guan, Y.H. He, Synth. Commun. 43, 2073 (2013)
D.R. Dreyer, H.P. Jia, C.W. Bielawski, Angew. Chem. Int. Ed. 49, 6813 (2010)
D.R. Dreyer, S. Park, C.W. Bielawski, R.S. Ruoff, Chem. Soc. Rev. 39, 228 (2010)
J.M. Khurana, K. Vij, Tetrahedron Lett. 52, 3666 (2011)
B. Karami, K. Eskandari, S. Khodabakhshi, Arkivoc Ix, 76 (2012)
A. Rashidi, M. Khaleghi Abbasabadi, S. Khodabakhshi, J. Nat. Gas Sci. Eng. 36, 13 (2016)
A. Rashidi, Z. Tavakoli, Y. Tarak, S. Khodabakhshi, M.K. Abbasabadi, J. Chin. Chem. Soc. 63, 399 (2016)
M.K. Abbasabadi, A. Rashidi, J. Safaei-Ghomi, S. Khodabakhshi, R. Rahighi, J. Sulf. Chem. 36, 660 (2015)
S. Stankovich, D.A. Dikin, G.H.B. Dommett, K.M. Kohlhaas, E.J. Zimney, E.A. Stach, R.D. Piner, S.T. Nguyen, R.S. Ruoff, Nature 442, 282 (2006)
D.W. Wang, F. Li, J.P. Zhao, W.C. Ren, Z.G. Chen, J. Tan, Z.S. Wu, I. Gentle, G.Q. Lu, H.M. Cheng, ACS Nano 3, 1745 (2009)
I.V. Lightcap, T.H. Kosel, P.V. Kamat, Nano Lett. 10, 577 (2010)
K.S. Novoselov, Z. Jiang, Y. Zhang, S.V. Morozov, H.L. Stormer, U. Zeitler, J.C. Maan, G.S. Boebinger, P. Kim, A.K. Geim, Science 315, 1379 (2007)
K.S. Novoselov, A.K. Geim, S.V. Morozov, D. Jiang, M.I. Katsnelson, I.V. Grigorieva, S.V. Dubonos, A.A. Firsov, Nature 438, 200 (2005)
S. Khodabakhshi, F. Marahel, A. Rashidi, M.K. Abbasabadi, J. Chin. Chem. Soc. 62, 389 (2015)
F. Hu, M. Patel, F. Luo, C. Flach, R. Mendelsohn, E. Garfunkel, H. He, M. Szostak, J. Am. Chem. Soc. 137, 14473 (2015)
B. Majumdar, D. Sarma, T. Bhattacharya, T.K. Sarma, A.C.S. Sustain, Chem. Eng. 5, 9286 (2017)
H. Hea, J. Klinowskia, M. Forsterb, A. Lerfb, Chem. Phys. Lett. 287, 53 (1998)
I.S. Chen, I.W. Tsai, C.M. Teng, J.J. Chen, Y.L. Chang, F.N. Ko, M.C. Lu, J.M. Pezzuto, Phytochemistry 46, 525 (1997)
H.K. Wabo, P. Tane, J.D. Connolly, C.C. Okunji, B.M. Schuster, M.M. Iwu, Nat. Prod. Res. 19, 591 (2005)
J.P. Michael, Nat. Prod. Rep. 20, 476 (2003)
M.J. Yao, Z. Guan, Y.H. He, Synth. Commun. 43, 2073 (2013)
M.M. Abdou, Arab. J. Chem. 10, 3324 (2017)
U. Emde, K. Schiemann, T. Schlueter, C. Saal, M. Maiwald. WO Patent. 147480A2 (2007)
E. Abbaspour-Gilandeh, M. Aghaei-Hashjinb, A. Yahyazadeh, H. Salemi, RSC Adv. 6, 55444 (2016)
F. Asghari-Haji, K. Rad-Moghadam, N.O. Mahmoodi, T. Tonekaboni, N. Rahimi, Appl. Organomet. Chem. 31, e3891 (2017)
X.S. Wang, Z.S. Zeng, D.Q. Shi, X.Y. Wei, Z.M. Zong, Synth. Commun. 34, 3021 (2004)
M. Lei, L. Ma, L.H. Hu, Tetrahedron Lett. 52, 2597 (2011)
Z. Tashrifi, K. Rad-Moghadam, M. Mehrdad, J. Mol. Liq. 248, 278 (2017)
D. Azarifar, Y. Abbasi, O. Badalkhani, Appl. Organomet. Chem. 32, e3949 (2017)
B. Karami, S. Khodabakhshi, K. Eskandari, Chem. Pap. 67, 1474 (2013)
J.C. Capraa, M.P. Cunhaa, D.G. Machadoa, A.D.E. Zomkowskia, B.G. Mendesc, A.R.S. Santos, M.G. Pizzolattic, S.A.L. Rodrigues, Eur. J. Pharmacol. 643, 232 (2010)
B. Karami, M. Kiani, J. Chin. Chem. Soc. 61, 213 (2014)
A. Witaicenis, L.N. Seito, A. da Silveira Chagas, L. Dominguesde Almeida, A.C. Uchini, P. Rodrigues-Orsi, S.H. Cestari, L.C.D. Stasi, Phytomedicine 21, 240 (2014)
X. Ni, Y. Guo, H. Bu, J. An, D. En, J. Chin. Chem. Soc. 59, 1439 (2012)
H. Nagabhushana, S.S. Saundalkar, L. Muralidhar, B.M. Nagab-hushana, C.R. Girija, D. Nagaraja, M.A. Pasha, V.P. Jayashankara, Chin. Chem. Lett. 22, 143 (2011)
M. Seifi, H. Sheibani, Catal. Lett. 126, 275 (2008)
T.S. Jin, L.B. Liu, Y. Zhao, T.S. Li, Synth. Commun. 35, 1859 (2005)
B.N. Seshu, N. Pasha, R.K.T. Venkateswara, P.P.S. Sai, N. Lingaiah, Tetrahedron Lett. 49, 2730 (2008)
S. Balalaie, S. Abdolmohammadi, Tetrahedron Lett. 48, 3299 (2007)
S. Banerjee, A. Horn, H. Khatri, G. Sereda, Tetrahedron Lett. 52, 1878 (2011)
M.M. Heravi, B. Alimadadi Jani, F. Derikvand, F.F. Bamoharram, H.A. Oskooie, Catal. Commun 10, 272 (2008)
Y. Peng, G. Song, Catal. Commun. 8, 111 (2007)
W.S.J. Hummers, R.E. Offeman, J. Am. Chem. Soc. 80, 1339 (1958)
M.K. Abbasabadi, A. Rashidi, S. Khodabakhshi, J. Nat. Gas Sci. Eng. 28, 87 (2016)
M.Z. Kassaee, E. Motamedi, M. Majdi, Chem. Eng. J. 172, 540 (2011)
C. Nethravathi, M. Rajamathi, Carbon 46, 1994 (2008)
G. Chen, S. Zhai, Y. Zhai, K. Zhang, Q. Yue, L. Wang, J. Zhao, H. Wang, J. Liu, J. Jia, Biosens. Bioelectron. 26, 3136 (2011)
J.S. Wang, R.T. Peng, J.H. Yang, Y.C. Liu, X.J. Hu, Carbohydr. Polym. 84, 1169 (2011)
X.J. Hu, Y.G. Liu, H. Wang, A.W. Chen, G.M. Zeng, S.M. Liu et al., Sep. Purif. Technol. 108, 189 (2013)
L.Z. Bai, D.L. Zhao, Y. Xu, J.M. Zhang, Y.L. Gao, L.Y. Zhao, J.T. Tang, Mater. Lett. 68, 399 (2012)
X. Huo, J. Liu, B. Wang, H. Zhang, Z. Yang, X. Sheb, P. Xia, J. Mater. Chem. A. 1, 656 (2013)
S. Khodabakhshi, B. Karami, New J. Chem. 38, 3586 (2014)
H. Kim, K.Y. Park, J. Hong, K. Kang, Sci. Rep. 4, 5278 (2014)
C. Hou, Q. Zhang, M. Zhu, Y. Li, H. Wang, Carbon 49, 47 (2011)
E. Rafiee, M. Khodayari, RSC Adv. 6, 36433 (2016)
M. Rohaniyan, A. Davoodnia, A. Nakhaei, Appl. Organomet. Chem. 30, 626 (2016)
A. Kumar, L. Rout, L.S. Kumar Achary, S.D. Rajendra, P. Dash, Sci. Rep. 7, 42975 (2017)
M. Mirza-Aghayan, M. Molaee Tavana, R. Boukherroub, Ultrason. Sonochem. 29, 371 (2016)
P. Li, Y. Gao, Z. Sun, D. Chang, G. Gao, A. Dong, Molecules 22, 12 (2017)
K. Tabatabaeian, H. Heidari, M. Mamaghani, N.O. Mahmoodi, Appl. Organomet. Chem. 26, 56 (2012)
M. Khoobi, L. Ma’mani, F. Rezazadehb, Z. Zareieb, A. Foroumadia, A. Ramazanib, J. Mol. Catal. A Chem 359, 74 (2012)
D. Azarifar, O. Badalkhani, Y. Abbasi, M. Hasanabadi, J. Iran. Chem. Soc. 14, 403 (2017)
A. Montaghami, N. Montazeri, Orient. J. Chem. 30, 1361 (2014)
R. Ghahremanzadeh, G.H. Imani Shakibaei, A. Bazgir, Synlett 8, 1129 (2008)
S. Balalaie, S. Abdolmohammadi, Tetrahedron Lett. 48, 3299 (2007)
H.J. Wang, J. Lu, Z.H. Zhang, Monatshefte Chem. 141, 1107 (2010)
D. Azarifar, M. Ghaemi, Appl. Organomet. Chem. 31, e3834 (2017)
D. Azarifar, O. Badalkhani, Y. Abbasi, J. Sulf. Chem. 37, 656 (2016)
D. Azarifar, M. Khaleghi-Abbasabadi, Res. Chem. Intermed. (2018)
Acknowledgements
The authors wish to thank the Research Council of Bu‐Ali Sina University for financial support to carry out this research. Islamic Azad University, North Tehran Branch, is also greatly acknowledged for technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khaleghi-Abbasabadi, M., Azarifar, D. Magnetic Fe3O4-supported sulfonic acid-functionalized graphene oxide (Fe3O4@GO-naphthalene-SO3H): a novel and recyclable nanocatalyst for green one-pot synthesis of 5-oxo-dihydropyrano[3,2-c]chromenes and 2-amino-3-cyano-1,4,5,6-tetrahydropyrano[3,2-c]quinolin-5-ones. Res Chem Intermed 45, 2095–2118 (2019). https://doi.org/10.1007/s11164-018-03722-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-03722-y