Abstract
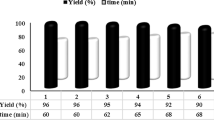
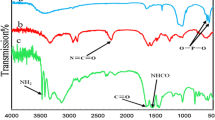
The magnetic nanostructured natural hydroxyapatite (HAP/Fe3O4 NPs) as a novel magnetic nanocatalyst was synthesized and fully characterized. The excellent catalytic activity of HAP/Fe3O4 NPs was investigated in the synthesis of biscoumarin derivatives under mild, green and solvent-free conditions. A series of aromatic (bearing different functional groups), heteroaromatic and aliphatic aldehydes have been converted to biscoumarins with good to excellent isolated yields. The significant advantages offered by the present method in close agreement with green chemistry principles are: catalyst is inexpensive, non-toxic, easy handling and reusable up to six recycle runs, magnetic separation of nanostructured catalyst, simple work-up procedure, short reaction time, high yields of products and using solvent-free conditions.
Graphical abstract











Similar content being viewed by others
References
J.D. Hepworth, C.D. Gabbutt, B.M. Heron, in Comprehensive Heterocyclic Chemistry-II, ed. by A.R. Katritzky, C.W. Rees, E.F.V. Scriven (Pergamon, NewYork, 1996), p. 417
S. Li, X. Qi, B. Huang, Catal. Today 276, 139 (2016)
J.C. Kim, R. Ryoo, M. Opanasenko, M. Shamzhy, J. Cejka, ACS Catal. 5, 2596 (2015)
M.A. Musa, A. Zhou, O.M. Sadik, J. Med. Chem. 7, 112 (2011)
K.M. Khan, Z.S. Saify, M.Z. Khan, C.M.I. Zia-Ullah, S. Atta-ur-Rahman, S. Perveen, Z.H. Chohan, C.T. Supuran, J. Enzyme Inhib. Med. Chem. 19, 373 (2004)
C.A. Kontogiorsis, D.J. Hadjipavloo-Litina, J. Med. Chem. 48, 6400 (2005)
M.E. Riveiro, A. Moglioni, R. Vazquez, N. Gomez, G. Facorro, L. Piehl, E.R. de Celis, C. Shayo, C. Davio, Bioorg. Med. Chem. 16, 2665 (2008)
I. Kostova, S. Bhatia, P. Grigorov, Curr. Med. Chem. 18, 3929 (2011)
N. Hamdi, F. Bouabdallah, A. Romerosa, R. Benhassen, C. R. Chimie. 13, 1261 (2010)
M. Waheed, N. Ahmed, Tetrahedron Lett. 57, 3785 (2016)
W.Y. Lin, L.L. Long, W. Tan, Chem. Commun. 46, 1503 (2010)
L.Q. Yan, Z.N. Kong, W. Shen, W.Q. Du, Y. Zhou, Z.J. Qi, RSC Adv. 6, 5636 (2016)
S. Park, J.E. Kwon, S.H. Kim, J. Seo, K. Chung, S.Y. Park, D.J. Jang, B.M. Medina, J. Gierschner, S.Y. Park, J. Am. Chem. Soc. 131, 14043 (2009)
H.Q. Li, L. Cai, J.X. Li, Y.X. Hu, P.P. Zhou, J.M. Zhang, Dyes Pigment. 91, 309 (2011)
K.D. Seo, H.M. Song, M.J. Lee, M. Pastore, C. Anselmi, F.D. Angelis, M.K. Nazeeruddin, M. Gräetzel, H.K. Kim, Dyes Pigments. 90, 304 (2011)
L. Long, Y. Wu, L. Wang, A. Gong, F. Hu, C. Zhang, Chem. Commun. 51, 10435 (2015)
E.B.B. Ong, N. Watanabe, A. Saito, Y. Futamura, K.H.A.E. Galil, A. Koito, N. Najimudin, H. Osada, J. Biol. Chem. 16, 14049 (2011)
A.K. Shaily, N. Ahmed, Supramol. Chem. 29, 146 (2017)
M. Waheed, N. Ahmed, Tetrahedron Lett. 57, 3785 (2016)
P. Kalita, R. Kumar, Micropor. Mesopor Mat. 149, 1 (2012)
B. Karami, M. Kiani, Catal. Commun. 14, 62 (2011)
A. Mobaraki, S. Yasham, B. Movassagh, Synlett 26, 1263 (2015)
D. Khan, S. Mukhtar, M.A. Alsharif, M.I. Alahmdi, N. Ahmed, Tetrahedron Lett. 58, 3183 (2017)
S. Pakdel, B. Akhlaghinia, A. Mohammadinezhad, Chem. Afr. (2019)
J.C. Jung, J.H. Lee, S. Oh, J.G. Lee, O.S. Park, Bioorg. Med. Chem. Lett. 14, 5527 (2004)
S. Stanchev, G. Momekov, F. Jensen, I. Manolov, Eur. J. Med. Chem. 43, 694 (2008)
I. Kostava, I. Manolov, I. Nicolova, S. Konstantonov, M. Karaivanova, Eur. J. Med. Chem. 36, 339 (2001)
Z.H. Chohan, A.U. Shaikh, A. Rauf, C.T. Supuran, J. Enzyme Inhib. Med. Chem. 21, 741 (2006)
J.W. Hinman, H. Hoeksema, E.L. Caron, W.G. Jackson, J. Am. Chem. Soc. 78, 1072 (1956)
J.H. Lee, H.B. Bang, S.Y. Han, J.G. Jun, Tetrahedron Lett. 48, 2889 (2007)
R.D.R.S. Manian, J. Jayashankaran, R. Raghunathan, Tetrahedron Lett. 48, 1385 (2007)
H. Zhao, N. Neamati, H. Hong, A. Mazumder, S. Wang, S. Sunder, G.W. Milne, Y. Pommier, T.R. Burke, J. Med. Chem. 40, 242 (1997)
C. Montagner, S.M. De Souza, C. Groposo, F. Delle Monache, E.F.A. Smania, A. Smania Jr., Z. Naturforsch C Bio. Sci. 63, 21 (2008)
Y.A. Al-Soud, H.H. Al-Sa’doni, H.A. Amajaour, K.S. Salih, M.S. Mubarak, N.A. Al-Masoudic, Z. Naturforsch., B: Chem. Sci. 63, 83 (2008)
K.M. Khan, S. Iqbal, M.A. Lodhi, G.M. Maharvi, M.I. Choudhary, S. Perveen, Bioorg. Med. Chem. 12, 1963 (2004)
V.D. Kancheva, P.V. Boranova, J.T. Nechev, I.I. Monolov, Biochimie 92, 1138 (2010)
A.N. Bhat, B.D. Jain, Talanta 5, 271 (1960)
J.S. Yadav, V.S. Reddy, A.K. Basak, B. Visali, A.V. Narsaiah, K. Nagaiah, Eur. J. Org. Chem. 2004, 546 (2004)
M.M. Heravi, F. Nahavandi, S. Sadjadi, H.A. Oskooie, F.F. Bamoharram, Synth. Commun. 40, 498 (2010)
J. Albadi, A. Mansournezhad, S. Salehnasab, Res. Chem. Intermed. 41, 5713 (2015)
A. Zhu, M. Wang, L. Li, J. Wang, RSC Adv. 5, 73974 (2015)
K.P. Boroujeni, P. Ghasemi, Z. Rafienia, Monatsh. Chem. 145, 1023 (2014)
R. Rezaei, F. Moezzi, M.M. Doroodmand, Chin. Chem. Lett. 25, 183 (2014)
B. Sadeghi, J. Chem. Res. 37, 171 (2013)
K. Tabataeian, H. Heidari, A. Khorshidi, M. Managhani, N.O. Mahmoodi, J. Serb. Chem. Soc. 77, 407 (2012)
W. Li, Y. Wang, Z. Wang, L. Dai, Y. Wang, Catal. Lett. 141, 1651 (2011)
H. Mehrabi, H. Abusaidi, J. Iran. Chem. Soc. 7, 890 (2010)
L. Shastri, S. Kalegowda, M. Kulkarni, Tetrahedron Lett. 48, 7215 (2007)
N. Montazeri, V. Vahabi, Bulg. Chem. Commun. 47, 136 (2015)
M.A. Zolfigol, R. Nasrabadi, S. Baghery, Appl. Organomet. Chem. 30, 500 (2016)
M. Sedighi, N. Montazeri, Adv. Stud. Biol. 7, 89 (2015)
K.P. Boroujeni, S. Hadizadeh, S. Hasani, A. Fadavi, M. Shahrokh, Acta Chim. Slov. 64, 692 (2017)
B. Karmakara, A. Nayakb, J. Banerji, Tetrahedron Lett. 53, 4343 (2012)
F. Shirini, A. Fallah-Shojaei, L. Samavi, M. Abedini, RSC Adv. 6, 48469 (2016)
S.K. Patil, D.V. Awale, M.M. Vadiyar, S.A. Patil, S.C. Bhise, S.S. Kolekar, Res. Chem. Intermed. 43, 5365 (2017)
J. Safaei-Ghomi, F. Eshteghal, H. Shahbazi-Alavi, Polycycl. Aromat. Compd. 1, 1 (2017)
A. Singh, P. Raj, R. Kaur, A. Singh, N. Kaur, N. Singh, New J. Chem. 41, 3872 (2017)
Z. Karimi-Jaberi, M.R. Nazarifar, B. Pooladian, Chin. Chem. Lett. 23, 781 (2012)
A. Shamsaddini, E. Sheikhhosseini, Int. J. Org. Chem. 4, 135 (2014)
M. BenOsman, S. DialloGarcia, J. Krafft, C. Méthivier, J. Blanchard, T. Yoshiokaand, J. Kubo, Phys. Chem. Chem. Phys. 18, 27837 (2016)
Z. Zarei, B. Akhlaghinia, RSC Adv. 6, 106473 (2016)
R. Jahanshahi, B. Akhlaghinia, RSC Adv. 5, 104087 (2015)
M. Zarghani, B. Akhlaghinia, Appl. Organomet. Chem. 29, 683 (2015)
Z. Zarei, B. Akhlaghinia, Chem. Pap. 69, 1421 (2015)
M. Razavi Fard, B. Akhlaghinia, J. Indian Chem. Soc. 92, 1715 (2015)
M. Zarghani, B. Akhlaghinia, RSC Adv. 5, 87769 (2015)
S. Ghodsinia, B. Akhlaghinia, RSC Adv. 5, 49849 (2015)
N. Razavi, B. Akhlaghinia, RSC Adv. 5, 12372 (2015)
E. Karimian, B. Akhlaghinia, S.S. Ghodsinia, J. Chem. Sci. 128, 429 (2016)
Moghadam B. Nazari, B. Akhlaghinia, S. Rezazadeh, Res. Chem. Intermed. 42, 1487 (2016)
N. Razavi, B. Akhlaghinia, New J. Chem. 40, 447 (2016)
S.S.E. Ghodsinia, B. Akhlaghinia, R. Jahanshahi, RSC Adv. 6, 63613 (2016)
N. Yousefi Siavashi, B. Akhlaghinia, M. Zarghani, Res. Chem. Intermed. 42, 5789 (2016)
R. Jahanshahi, B. Akhlaghinia, RSC Adv. 6, 29210 (2016)
R. Jahanshahi, B. Akhlaghinia, Res. Chem. Intermed. 44, 2451 (2018)
M. Zarghani, B. Akhlaghinia, RSC Adv. 6, 31850 (2016)
M. Zarghani, B. Akhlaghinia, RSC Adv. 6, 38592 (2016)
S. Memar Masjed, B. Akhlaghinia, M. Zarghani, N. Razavi, Aust. J. Chem. 70, 33 (2017)
M. Esmaielpour, B. Akhlaghinia, R. Jahanshahi, J. Chem. Sci. 129, 313 (2017)
R. Jahanshahi, B. Akhlaghinia, New J. Chem. 41, 7203 (2017)
M. Ghasemzadeh, B. Akhlaghinia, Bull. Chem. Soc. Jpn 90, 1119 (2017)
M. Nejatianfar, B. Akhlaghinia, R. Jahanshahi, Appl. Organomet. Chem. 32, 4095 (2017)
Z. Zarei, B. Akhlaghinia, New J. Chem. 41, 15485 (2017)
N. Mohammadian, B. Akhlaghinia, Res. Chem. Intermed. 43, 3325 (2017)
R. Jahanshahi, B. Akhlaghinia, Chem. Pap. 71, 1351 (2017)
A. Mohammadinezhad, B. Akhlaghinia, Aust. J. Chem. 71, 32 (2018)
N. Mohammadian, B. Akhlaghinia, Res. Chem. Intermed. 44, 1085 (2018)
Z. Zarei, B. Akhlaghinia, Turk. J. Chem. 42, 170 (2018)
M. Ghasemzadeh, B. Akhlaghinia, Chemistry Select. 3, 3161 (2018)
M. Zarghani, B. Akhlaghinia, Bull. Chem. Soc. Jpn 89, 1192 (2016)
A. Mohammadinezhad, B. Akhlaghinia, Green Chem. 19, 5625 (2017)
N. Razavi, B. Akhlaghinia, R. Jahanshahi, Catal. Lett. 147, 360 (2017)
R. Jahanshahi, B. Akhlaghinia, Catal. Lett. 147, 2640 (2017)
M. Zamani, B. Akhlaghinia, A. Mohammadinezhad, ChemistrySelect. 3, 9431 (2018)
F. Heidari, M.E. Bahrololoom, D. Vashaee, L. Tayebi, Ceram. Int. 41, 3094 (2015)
M.E. Bahrololoom, M. Javidi, S. Javadpour, J. Ma, J. Ceram. Process. Res. 10, 129 (2009)
M.M.A. Nikje, L. Sarchami, L. Rahmani, Int. J. Nanosci. Nanotechnol. 11, 39 (2015)
J. Venkatesan, S.K. Kim, Materials 3, 4761 (2010)
X.Y. Lü, Y.B. Fan, D. Gu, W. Cui, Key Eng. Mater. 342, 213 (2007)
Z. Zhang, Z. Yuan, D. Tang, Y. Ren, K. Lv, B. Liu, Chemsuschem 7, 3496 (2014)
M.S. Sajadi, M. Meskinfam, H. Jazdarreh, Int. J. Nano. Dim. 1, 57 (2010)
S. Li, H. Bai, J. Wang, X. Jing, Q. Liu, M. Zhang, R. Chen, L. Liu, C. Jiao, Chem. Eng. J. 193, 372 (2012)
R.J. Abraham, J.J. Byrne, L. Griffiths, M. Perez, Magn. Reson. Chem. 44, 491 (2006)
J.C. Lien, S.C. Chen, L.J. Huang, S.C. Kuo, J. Chin. Chem. Soc. 51, 847 (2004)
G.X. Gong, J.F. Zhou, L.T. An, X.L. Duan, S.J. Ji, Synth. Commun. 39, 497 (2009)
M. Nikpassand, L. Zare Fekri, S. Sahrapeima, Bull. Chem. Soc. Ethiop. 31, 323 (2017)
A. Noorahmadsab Nadaf, K. Shivashankar, J. Heterocycl. Chem. 55, 1375 (2018)
S.S. Kauthale, S.U. Tekale, K.M. Jadhav, R.P. Pawar, Mol. Divers 20, 763 (2016)
M.N. Elinson, A.N. Vereshchagin, O.O. Sokolova, J. Org. Chem. Part iii, 121 (2017)
A. Zhu, S. Bai, L. Li, M. Wang, J. Wang, Catal. Lett. 145, 1089 (2015)
S.C. Azimi, K. Rad-Moghadam, Iran. Chem. Commun. 3, 356 (2015)
A. Sahar, Z. Ali Khan, M. Ahmad, A.F. Zahoor, A. Mansha, A. Iqbal, Trop. J. Pharm. Res. 16, 203 (2017)
R. Karimian, F. Piri, A.A. Safari, S.J. Davarpanah, J. Nanostruct. 3, 52 (2013)
Faisal M, F.A. Larik, A. Saeed, J. Porous Mater. (2018)
V. Padalkar, K. Phatangare, S. Takale, R. Pisal, A. Chaskar, J. Saudi Chem. Soc. 19, 42 (2015)
Acknowledgements
The authors gratefully acknowledge the partial support of this study by the Ferdowsi University of Mashhad Research Council (Grant No. 2/47664).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Akhlaghinia, B., Sanati, P., Mohammadinezhad, A. et al. The magnetic nanostructured natural hydroxyapatite (HAP/Fe3O4 NPs): an efficient, green and recyclable nanocatalyst for the synthesis of biscoumarin derivatives under solvent-free conditions. Res Chem Intermed 45, 3215–3235 (2019). https://doi.org/10.1007/s11164-019-03788-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03788-2