Abstract
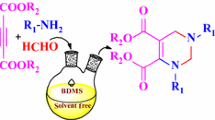
An efficient and eco-friendly synthetic protocol via a one-pot three-component reaction to yield thiophene containing aminonaphthols (Betti bases) is reported. Kaolin was used as a heterogeneous catalyst and a series of Betti bases were synthesized under solvent-free condition. The synthesized derivatives were characterized by various techniques, viz. FTIR, 1H-NMR, 13C-NMR, mass spectrometry and elemental analysis. The synthesized compounds were screened for in vitro anticancer and antioxidant activities. Compounds 4d, 4i, 4j exhibited excellent anticancer activity (GI50 < 10 µg/ml) against all four cell lines which was comparable with the standard drug doxorubicin. Compound 4d was found to be the most potent antioxidant against DPPH radical and H2O2. The reusability studies demonstrated the retention in catalytic activity for up to six cycles. The current synthetic methodology offers several advantages such as excellent yields, mild reaction conditions, short reaction span, ease of work-up, broad substrate scope and reusability.
Graphic abstract





Similar content being viewed by others
References
G. Nagendrappa, Reson. 7, 59 (2002)
W. Nesse, Introduction to Mineralogy, (New York: Oxford University Press) pp. 254 (2000)
P. Prabhakaran, P. Pushpaletha, Int. J. Appl. Chem. 13(3), 461 (2017)
F. Pienkoß, C. Ochoa-Hernández, N. Theyssen, W. Leitner, A.C.S. Sustain, Chem. Eng. 6(7), 8782 (2018)
P. Sahu, P. Sahu, D. Agarwal, RSC Adv. 3, 9854 (2013)
G. Srihari, M. Murthy, Synth. Commun. 41(18), 2684 (2011)
Z. Gordi, M. Vazan, Iran. J. Catal. 6(1), 75 (2016)
M. Hirano, H. Monobe, S. Yakabe, and T. Morimoto, J. Chem. Res. (S), 662 (1998)
M. Heravi, F. Derikvand, L. Ranjbar, H. Oskooie, Heteroat. Chem. 19(2), 215 (2008)
H. Murray, S. Lyons, Clays Clay Miner. 4, 31 (1955)
R. Armstrong, A. Combs, P. Tempest, S. Brown, T. Keating, Acc. Chem. Res. 29(3), 123 (1996)
G. Sharma, B. Devi, K. Reddy, M. Reddy, A. Kondapi, and C. Bhaskar, Heterocycl. Commun. 21 (4) (2015)
M. Dintzner, J. Maresh, C. Kinzie, A. Arena, T. Speltz, J. Chem. Educ. 89, 265 (2012)
S. Sayyahi, S. Jahanbakhshi, and Z. Dehghani, J. Chem. (Hindawi. Online) 2013 (2013)
R. Ballini, F. Bigi, M. Conforti, D. Santis, R. Maggi, G. Oppici, G. Sartori, Catal. Today 60, 305 (2000)
M. Betti, Org. Synth. 9, 60 (1929)
C. Cardellicchio, M. Capozzi, F. Naso, Tetrahedron Asymmetry 21(5), 507 (2010)
I. Szatmári, A. Hetényi, L. Lázár, F. Fülöp, J. Heterocyclic Chem. 41, 367 (2004)
C. Mukhopadhyay, S. Rana, R. Butcher, ARKIVOC 12, 291 (2010)
J. Mou, G. Gao, C. Chen, J. Liu, J. Gao, Y. Liu, D. Pei, RSC Adv. 7, 13868 (2017)
Z. Tang, Z. Zhu, Z. Xia, H. Liu, J. Chen, W. Xiao, X. Ou, Molecules 17(7), 8174 (2012)
K. Waisser, L. Kubicová, V. Buchta, P. Kubanová, K. Bajerová, L. Jirásková, O. Bednarík, O. Bures, P. Holý, Folia Microbiol. 47(5), 488 (2002)
J.B. Chylińska, T. Urbański, M. Mordarski, J. Med. Chem. 6(5), 484 (1963)
L. Benameur, Z. Bouaziz, P. Nebois, M. Bartoli, M. Boitard, H. Fillion, Chem. Pharm. Bull. 44(3), 605 (1996)
B. Mathew, A. Kumar, S. Sharma, P. Shukla, M. Nath, Eur. J. Med. Chem. 45(4), 1502 (2010)
N. Georgieva, Z. Yaneva, S. Simova, G. Nikolova, Bulg. Chem. Commun. 49(D), 201 (2017)
A. Puerta, A. Galán, R. Abdilla, K. Demanuele, M. Fernandes, G. Bosica, J. Padrón, J. Mol. Clin. Med. 2(2), 35 (2019)
A. Shen, C. Tsai, C. Chen, Eur. J. Med. Chem. 34, 877 (1999)
A. Jha, N. Paul, S. Trikha, T. Cameron, Can. J. Chem. 84, 843 (2006)
A. Shahrisa, R. Teimuri-Mofrad, M. Gholamhosseini-Nazari, Mol. Divers. 19, 87 (2015)
A. Kumar, M. Gupta, M. Kumar, Tetrahedron Lett. 51, 1582 (2010)
S. Zahiri, M. Mokhtary, M. Torabi, Iran. J. Catal. 5(2), 149 (2015)
M. Lashkari, M. Maghsoodlou, M. Karima, B. Adrom, M. Fatahpour, Acta Chemica Iasi 24(2), 112 (2016)
B. Adrom, N. Hazeri, M. Maghsoodlou, M. Mollamohammadi, Res. Chem. Intermed. 41(7), 4741 (2015)
M. Maghsoodlou, M. Karima, M. Lashkari, B. Adrom, J. Aboonajmi, J. Iran Chem. Soc. 14, 329 (2017)
C. Capello, U. Fischer, K. Hungerbuhler, Green Chem. 9, 927 (2007)
J. Metzger, Angew. Chem. Int. Ed. 37(21), 2975 (1998)
G. Bosica, R. Abdilla, K. Demanuele, Eur. J. Org. Chem. 44, 6127 (2018)
M. Patil, C. Khatri, G. Chaturbhuj, Monatsh. Chem. 149, 1453 (2018)
R. Teimuri-Mofrad, M. Gholamhosseini-Nazari, S. Esmati, A. Shahrisa, Res. Chem. Intermed. 43(12), 6845 (2017)
F. Janati, M. Heravi, A. Shokraie, Synth. React. Inorg. M. 45(1), 1 (2014)
S. Imtiaz, J. Ahmad War, S. Banoo, S. Khan, RSC Adv. 11(19), 11083 (2021)
Y. Gu, Green Chem. 14, 2091 (2012)
J. Ferlay, M. Colombet, I. Soerjomataram, D. Parkin, M. Piñeros, A. Znaor, and F. Bray, Int. J. Cancer. Apr 5 (2021)
V. Vichai, K. Kirtikara, Nat. Protoc. 1, 1112 (2006)
S. Shaikh, G. Pavale, P. Dhavan, P. Singh, J. Uparkar, S. Vaidya, B. Jadhav, M. Ramana, Bioorg. Chem. 110, 104770 (2021)
S. Shaikh, P. Dhavan, M. Ramana, B. Jadhav, Bioorg. Chem. 96, 103589 (2020)
I. Gulcin, M. Buyukokuroglu, O. Kufrevioglu, J. Pineal Res. 34, 278 (2003)
Acknowledgements
We are thankful to Microanalytical Laboratory, University of Mumbai for providing characterization facilities. We are also thankful to the Tata Memorial Centre’s Advanced Centre for Treatment Research and Education in Cancer (ACTREC) AntiCancer Drug Screening Facility (ACDSF), Kharghar, Navi Mumbai, for providing anticancer activity.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yellapurkar, I., Shaikh, S., Pavale, G. et al. Kaolin-catalysed one-pot synthesis of thiophene containing aminonaphthols under solvent-free condition and their in vitro anticancer and antioxidant activity. Res Chem Intermed 47, 4067–4082 (2021). https://doi.org/10.1007/s11164-021-04516-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04516-5