Abstract
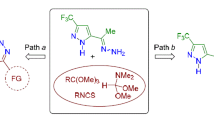
3-Trifluoromethyl-substituted 4-nitrosopyrazolines and 4-nitrosopyrazoles were prepared by a one-pot synthesis from trifluoromethyl-containing 1,3-diketones, sodium nitrite in acetic acid, and hydrazines (hydrazine hydrate, methylhydrazine). 3-Trifluoromethylpyrazolines can be converted to pyrazoles on heating. The use of phenylhydrazine in these reactions led to the formation of regioisomeric 4-hydroxyimino-5-(trifluoromethyl)pyrazoline. The structure of heterocycles synthesized was established using X-ray diffraction study, 1H and 19F NMR spectroscopy. The obtained products exhibited considerable tuberculostatic activity.
Similar content being viewed by others
References
Ya. A. Veselov, E. I. Krasnikova, I. M. Veselovskii, Antibiotiki i khimioterapiya, 1992, 37, 13 [Antibiot. Khimioterap. (Engl. Transl.), 1992, 37]; D. Sinder, S. Dooley, Heart Lung, 1993, 22, 365.
N. M. Rudoi, T. A. Smailova, S. K. Mardanov, Problemy tuberkuleza [Problems of tuberculosis], 1991, 32 (in Russian).
US Pat. 2,510,724; Chem. Abstr., 1950, 44, 8376.
S. F. Torf, N. I. Kudryashova, N. V. Khromov-Borisov, T. A. Mikhailova, Zh. Obshch. Khim., 1962, 32, 1740 [Russ. J. Gen. Chem. (Engl. Transl.), 1962, 32].
V. I. Saloutin, Y. V. Burgart, Z. E. Skryabina, O. G. Kuzueva, J. Fluor. Chem., 1997, 84, 107.
J. C. Sloop, C. L. Bumgardner, W. D. Loehle, J. Fluor. Chem., 2002, 118, 135.
S. P. Singh, J. K. Kapoor, D. Kumar, M. D. Threadgill, J. Fluor. Chem., 1997, 83, 73.
O. G. Khudina, E. V. Shchegol’kov, Y. V. Burgart, M. I. Kodess, O. N. Kazheva, A. N. Chekhlov, G. V. Shilov, O. A. Dyachenko, V. I. Saloutin, O. N. Chupakhin, J. Fluor. Chem., 2005, 126, 1230.
S. P. Singh, D. Kumar, M. D. Threadgill, Ind. J. Chem., Sec. B, 1992, 31, 233; S. P. Singh, L. S. Tarar, R. K. Vaid, J. Elguero, A. Martinez, J. Heterocycl. Chem., 1989, 26, 733.
S. Fustero, R. Román, J. F. Sanz-Cervera, A. Simón-Fuentes, A. C. Cuňat, S. Villanova, M. Murguía, J. Org. Chem., 2008, 73, 3523.
R. D. Pearson, Usp. Khim., 1971, 40, 1259 [Russ. Chem. Rev. (Engl. Transl.), 1971, 40, No. 7].
G. M. Sheldrick, SHELXS-97, Program for the Solution of Crystal Structure, University of Göttingen, Germany, 1997.
G. M. Sheldrick, SHELXL-97, Program for the Refinement of Crystal Structure, University of Göttingen, Germany, 1997.
T. N. Yashchenko, I. S. Mecheva, Rukovodstvo po laboratornym issledovaniyam pri tuberkuleze [Handbook on Tuberculosis Laboratory Studies], Meditsina, Moscow, 1973, 260 pp. (in Russian); V. N. Vasilyov, Mikobakteriozy i mikozy legkikh [Lung Micobacteriose and Micose], Meditsina i Fizkul’tura, Sofia, 1971, p. 377 (in Bulgarian).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 1917–1923, October, 2010.
Rights and permissions
About this article
Cite this article
Khudina, O.G., Burgart, Y.V., Saloutin, V.I. et al. One-pot synthesis of trifluoromethyl- and nitroso-substituted pyrazolines and pyrazoles and their tuberculostatic activity. Russ Chem Bull 59, 1967–1973 (2010). https://doi.org/10.1007/s11172-010-0341-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-010-0341-7