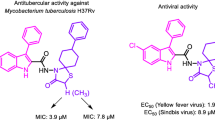
Abstract
Bis-spiro heterocycles containing spiro units at the 1,3-positions of the pyrrolizidine (isothiapyrrolizidine) moiety were synthesized by the reaction of unstabilized azomethine ylides, which were generated in situ from isatin and proline (isothiaproline), with hetarylidene-substituted rhodanines. Quantum chemical calculations of potential energy surface sections and descriptors controlling the regioselectivity of cycloaddition were carried out. For a number of compounds in vitro activity against the influenza virus A/California/07/09 (H1N1)pdm2009 was experimentally established.
Article PDF
Similar content being viewed by others
References
M. E. Heitzman, C. C. Neto, E. Winiarz, A. J. Vaisberg, G. B. Hammond, Phytochemistry, 2005, 66, 5.
C. Marti, E. M. Carreira, Eur. J. Org. Chem., 2003, 2209.
C. V. Galliford, K. A. Scheidt, Angew. Chem., Int. Ed. Engl., 2007, 46, 8748.
A. S. Girgis, Eur. J. Med. Chem., 2009, 44, 91.
M. M. Khafagy, A. H. F. A. El-Wahas, F. A. Eid, A. M. El-Agrody, Farmaco, 2002, 57, 715.
P. R. Sebahar, R. M. Williams, J. Am. Chem. Soc., 2000, 122, 5666.
G. Periyasami, R. Raghunathan, G. Surendiran, N. Mathivanan, Eur. J. Med. Chem., 2009, 44, 959.
G. Wu, L. Ouyang, J. Liu, S. Zeng, W. Huang, B. Han, F. Wu, G. He, M. Xiang, Mol. Divers., 2013, 17, 271.
G. Periyasami, R. Raghunathan, G. Surendiran, N. Mathivanan, Bioorg. Med. Chem. Lett., 2008, 18, 2342.
A. A. Raj, R. Raghunathan, M. R. Sridevi Kumari, N. Raman, Bioorg. Med. Chem., 2003, 11, 407.
R. S. Kumar, S. M. Rajesh, S. Perumal, D. Banerjee, P. Yogeeswari, D. Sriram, Eur. J. Med. Chem., 2010, 45, 411.
A. C. Wei, M. A. Ali, Y. K. Yoon, R. Ismail, T. S. Choon, R. S. Kumar, Bioorg. Med. Chem. Lett., 2013, 23, 1383.
P. Prasanna, K. Balamurugan, S. Perumal, P. Yogeeswari, D. Sriram, Eur. J. Med. Chem., 2010, 45, 5653.
M. A. Ali, R. Ismail, T. S. Choon, Y. K. Yoon, A. C. Wei, S. Pandian, R. S. Kumar, H. Osman, E. Manogaran, Bioorg. Med. Chem. Lett., 2010, 20, 7064.
Y. Kia, H. Osman, R.S. Kumar, V. Murugaiyah, A. Basiri, S. Perumal, I. A. Razak, Bioorg. Med. Chem. Lett., 2013, 23, 2979.
Y. Zhao, D. Bernard, S. Wang, BioDiscovery, 2013, 8, 4.
C. Najera, J. M. Sansano, Curr. Org. Chem., 2003, 7, 1105.
A. A. Shvets, S. V. Kurbatov, Russ. J. Org. Chem. (Engl. Transl.), 2010, 46, 306 [Zh. Org. Khim., 2010, 46, 309].
A. A. Shvets, S. V. Kurbatov, Russ. Chem. Bull. (Int. Ed.), 2010, 59, 1979 [Izv. Akad. Nauk, Ser. Khim., 2010, 1928].
A. A. Shvets, S. V. Kurbatov, Chem. Heterocycl. Compd. (Engl. Transl.), 2009, 45, 866 [Khim. Geterotsikl. Soedin., 2009, 1087].
A. A. Shvets, S. V. Kurbatov, Chem. Heterocycl. Compd. (Engl. Transl.), 2012, 48, 799 [Khim. Geterotsikl. Soedin., 2012, 859].
A. A. Shvets, A. G. Mikhailovskii, D. V. Zimakov, S. V. Kurbatov, O. V. Surikova, Russ. J. Org. Chem. (Engl. Transl.), 2011, 47, 1436 [Zh. Org. Khim., 2011, 47, 1409].
A. A. Shvets, Yu. V. Nelyubina, K. A. Lyssenko, S. V. Kurbatov, Russ. Chem. Bull. (Int. Ed.), 2012, 61, 1659 [Izv. Akad. Nauk, Ser. Khim., 2012, 1642].
D. V. Steglenko, Ph. D. (Chem.) Thesis, Institute of Physical and Organic Chemistry, Southern Federal University, Rostov-on-Don, 2012, 123 pp. (in Russian).
T. G. W. Frisch, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery Jr., T. K. K. N. Vreven, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. G. A. Rega, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. R. A. D. Malick, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. C. M. Nanayakkara, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, J. A. Pople, Gaussian 03, Revision E.01; Gaussian, Inc., Wallingford CT (USA), 2004.
T. V. Pyrkov, I. V. Ozerov, E. D. Balitskaya, R. G. Efremov, Russ. J. Bioorg. Chem. (Engl. Transl.), 2010, 36, 446 [Bioorg. Khim., 2010, 36, 482].
C. Scholtissek, G. Quack, H. D. Klenk, R. G. Webster, Antiviral Res., 1998, 37, 83.
E. De Clercq, Nat. Rev. Drug Discov., 2006, 5, 1015.
A. K. Shetty, L. A. Peek, Expert Rev. Anti Infect. Ther., 2012, 10, 123.
C. J. Vavricka, Q. Li, Y. Wu, J. Qi, M. Wang, Y. Liu, F. Gao, J. Liu, E. Feng, J. He, J. Wang, H. Liu, H. Jiang, G. F. Gao, PLoS Pathog., 2011, 7, e1002249.
F. S. Dawood, S. Jain, L. Finelli, M. W. Shaw, N. Engl. J. Med., 2009, 360, 2605.
Influenza A(H1N1) Virus Resistance to Oseltamivir — 2008/2009 Influenza Season, Northern Hemisphere, 2008; http://www.who.int/influenza/resources/documents/H1N1webupdate20090318-ed-ns.pdf
A. C. Wei, M. A. Ali, Y. K. Yoon, R. Ismail, T. S. Choon, R. S. Kumar, N. Arumugam, A. I. Almansour, H. Osman, Bioorg. Med. Chem. Lett., 2012, 22, 4930.
Yu. M. Pashkevich, Farmatsevtichnii Zh. (Kiev), 1961, 16, 10; Chem. Abstr., 1962, 56, 38460.
E. L. Tarasevichyus, Farmatsevtichnii Zh. (Kiev), 1966, 21, 11; Chem. Abstr., 1967, 66, 104946.
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on the materials of the First Russian Conference on Medicinal Chemistry (“MedChem Russia-2013”) with International Participation (September 8–12, 2013, Moscow).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 5, pp. 1130–1136, May, 2014.
Rights and permissions
About this article
Cite this article
Kurbatov, S.V., Zarubaev, V.V., Karpinskaya, L.A. et al. Synthesis and antiviral activity of bis-spirocyclic derivatives of rhodanine. Russ Chem Bull 63, 1130–1136 (2014). https://doi.org/10.1007/s11172-014-0560-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-014-0560-4