Abstract
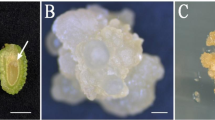
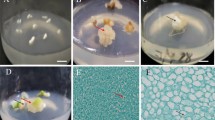
The efficiency of immature embryo-derived in vitro culture of G genome wheats is significantly influenced by various auxins and sugars which are used for induction of embryogenic response, and by regeneration media composition for promotion of plant development from subcultured embryogenic calli. The embryogenic calli of Triticum timopheevii has demonstrated the highest regeneration ability when the initial explants were cultured on the media supplemented with 4 mg l−1 of Picloram (29.0 %), 4 mg l−1 of Dicamba (28.7 %) or 3 mg l−1 of 2,4-D (29.1 %). The media supplemented with 5–6 mg l−1 of Picloram were considered to be the most effective for promotion of embryogenic/regenerable callus production in Triticum kiharae cultures (73.7–75.0 %). Both T. timopheevii and T. kiharae embryogenic structures were characterized by the formation of green and albino plantlets. Generally the medium that was initially supplemented with Picloram promoted the formation of lower albino plants fraction rather than 2,4-D and Dicamba. As it was measured by the total green plant production per initial explant, the overall efficiency has been reduced when sucrose was substituted by glucose or maltose. The regeneration medium supplemented with 0.25 mg l−1 TDZ significantly enhanced the regeneration capacity of embryogenic callus in T. kiharae. In culture of T. timopheevii the difference between the medium lack of growth regulators and the medium supplemented with TDZ was not prominent, though both of the media have demonstrated the greater efficacy as compared to those supplemented with BA and Zeatin.

Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- BA:
-
6-Benzyladenine
- Dicamba:
-
3,6-Dichloro-2-methoxybenzoic acid
- IAA:
-
Indole-3-acetic acid
- MS:
-
Murashige and Skoog medium
- Picloram:
-
4-Amino-3,5,6-trichloro-2-pyridinecarboxylic acid
- TDZ:
-
N-phenyl-N′-(1,2,3-thiadiazol-5-yl)urea (thidiazuron)
References
Afsharsterle S, Peng ECK, Brown JS, Kollmorgen JF (1996) Embryogenic callus induction and plant regeneration in Triticum tauschii, the diploid D-genome donor for bread wheat. Aust J Bot 44:489–497
Ainsley PJ, Aryan AP (1998) Efficient plant regeneration system for immature embryos of triticale (x Triticosecale Wittmack). Plant Growth Regul 24:23–30
Andreev YA, Korostyleva TV, Slavokhotova AA et al (2012) Genes encoding hevein-like defense peptides in wheat: distribution, evolution, and role in stress response. Biochimie 94:1009–1016
Asif M, Eudes F, Randhawa H, Amundsen E, Spaner D (2014) Phytosulfokine alpha enhances microspore embryogenesis in both triticale and wheat. Plant Cell Tissue Organ Cult 116:125–130
Badaeva ED, Shkutina FM, Bogdevich IN, Badaev NS (1986) Comparative study of Triticum aestivum and T.timopheevii genomes using C-banding techniques. Plant Syst Evol 154:183–194
Badaeva ED, Badaev NS, Filatenko AA, Boguslavsky RL, Zelenin AV (1990) Cytological investigation of cereal, hexa- and octoploid species containing G genome. Genetika (Moscow) 26:708–716 (in Russian)
Barro F, Canalejo A, Martin A (1999a) Genomic influence on somatic embryogenesis in the Triticeae. Plant Cell Rep 18:769–772
Barro F, Martin A, Lazzeri PA, Barceló P (1999b) Medium optimization for efficient embryogenesis and plant regeneration from immature inflorescences and immature scutella of elite cultivars of wheat, barley and tritordeum. Euphytica 108:161–167
Bohorova NE, Pfeiffer WH, Mergoum M, Crossa J, Pacheco M, Estanol P (2001) Regeneration potential of CIMMYT durum wheat and triticale varieties from immature embryos. Plant Breed 120:291–295
Brown C, Brooks FJ, Pearson D, Mathias RJ (1989) Control of embryogenesis and organogenesis in immature embryo callus using increased medium osmolarity and abscisic acid. J Plant Physiol 133:727–733
Brown-Guedira GL, Gill BS, Bockus WW, Cox TS, Hatchett JH, Leath S, Peterson CJ, Thomas JB, Zwer PK (1996) Evaluation of a collection of wild timopheevii wheat for resistance to disease and arthropod pests. Plant Dis 80:928–933
Chauhan H, Desai SA, Khurana P (2007) Comparative analysis of the differential regeneration response of various genotypes of Triticum aestivum, Triticum durum and Triticum dicoccum. Plant Cell Tiss Org Cult 91:191–199
Cho MJ, Jiang W, Lemaux PG (1998) Transformation of recalcitrant barley cultivars through improvement of regenerability and decreased albinism. Plant Sci 138:229–244
Cho MJ, Banh J, Yu M, Kwan J, Jones TJ (2015) Improvement of Agrobacterium-mediated transformation frequency in multiple modern elite commercial maize (Zea mays L.) inbreds by media modifications. Plant Cell Tiss Org Cult 121:519–529
Chu C, Hill R, Brule-Babel A (1990) High frequency of pollen embryoid formation and plant regeneration in Triticum aestivum L. Plant Sci 66:255–262
Delporte F, Pretova A, du Jardin P, Watillon B (2014) Morpho-histology and genotype dependence of in vitro morphogenesis in mature embryo cultures of wheat. Protoplasma 251(6):1455–1470
Dorofeev VF, Filatenko AA, Migushova EF, Udachin RA, Jakubtsiner MM (1979) Pshenitsa (Wheat). In: Dorofeev VF, Korovina ON (eds) Cultivated flora of the USSR, vol 1 (in Russian). Kolos, Leningrad
Fahmy AH, Li J, Abou El-Wafa W, Sel-S El-Khodary, El Shihy OM (2012) Effects of different combinations of benzyl adenine and indole acetic acid concentrations on in vitro plant regeneration in hexaploid wheat. GM Crops Food 3(2):111–114
Fennell S, Bohorova N, Ginkel MV, Crossa J, Hoisington D (1996) Plant regeneration from immature embryos of 48 elite CIMMYT bread wheats. Theor Appl Genet 92:163–169
Filippov M, Miroshnichenko D, Vernikovskaya D, Dolgov S (2006) The effect of auxins, time exposure to auxin and genotypes on somatic embryogenesis from mature embryos of wheat. Plant Cell Tiss Org Cult 84:213–222
Ganeshan S, Båga M, Harvey BL, Rossnagel BG, Scoles GJ, Chibbar RN (2003) Production of multiple shoots from thidiazuron-treated mature embryos and leaf-base/apical meristems of barley (Hordeum vulgare L.). Plant Cell Tiss Org Cult 73:57–64
Ganeshan S, Chodaparambil SV, Båga M, Fowler DB, Hucl P, Rossnagel BG, Chibbar RN (2006) In vitro regeneration of cereals based on multiple shoot induction from mature embryos in response to thidiazuron. Plant Cell Tiss Org Cult 85:63–73
Geng PP, La HG, Wang HQ, Stevens EJC (2008) Effect of sorbitol concentration on regeneration of embryogenic calli in upland rice varieties (Oryza sativa L.). Plant Cell Tiss Organ Cult 92:303–313
Ghaemi M, Sarrafi A (1994) The effect of the “D” genome from synthetic wheat lines in anther culture responses. Plant Breed 112:76–79
Goncharov NP (2011) Genus Triticum L. taxonomy: the present and the future. Plant Syst Evol 295:1–11
Goncharov NP, Bannikova SV, Kawahara T (2007) Wheat artificial amphiploids involving the Triticum timopheevii genome: their studies, preservation and reproduction. Genet Resour Crop Evol 54:1507–1516
Hagio T (2002) Adventitious shoot regeneration from immature embryos of sorghum. Plant Cell Tiss Org Cult 68:65–72
Han Y, Jin XL, Wu FB, Zhang GP (2011) Genotypic differences in callus induction and plant regeneration from mature embryos of barley (Hordeum vulgare L.). J Zhejiang Univ Sci B 12(5):399–407
He GY, Lazzeri PA (2001) Improvement of somatic embryogenesis and plant regeneration from durum wheat (Triticum turgidum var. durum Desf.) scutellum and inflorescence cultures. Euphytica 119:369–376
Hou BK, Yu HM, Teng SY (1997) Effects of low temperature on induction and differentiation of wheat calluses. Plant Cell Tiss Org Cult 49:35–38
Jiang J, Gill BS (1994) Different species-specific chromosome translocations in Triticum timopheevii and T. turgidum support the diphyletic origin of polyploid wheats. Chrom Res 2:59–64
Karsai I, Bedo Z, Hayes PM (1994) Effect of induction medium pH and maltose concentration on in vitro androgenesis of hexaploid winter triticale and wheat. Plant Cell Tiss Org Cult 39:49–53
Kereša S, Bariæ M, Šarèeviæ H, Gunjaèa J (2004) Influence of Zeatin on wheat regeneration from immature embryos. Agric Conspec Sci 69(1):17–20
Kumari M, Clarke HJ, Small I, Siddique KH (2009) Albinism in plants: a major bottleneck in wide hybridization, androgenesis and doubled haploid culture. Crit Rev Plant Sci 28:393–409
Larsen ET, Tuvesson IKD, Andersen SB (1991) Nuclear genes affecting percentage of green plants in barley (Hordeum vulgare L.) anther culture. Theor Appl Genet 82:417–420
Lazar MD, Collins GB, Vian WE (1983) Genetic and environmental effects on the growth and differentiation of wheat somatic cell cultures. J Hered 74:353–357
Machii H, Mizuno H, Hirabayashi T, Hagio T (1998) Screening wheat genotypes for high callus induction and regeneration capability from anther and immature embryo cultures. Plant Cell Tiss Org Cult 53:67–74
Makowska K, Oleszczuk S (2014) Albinism in barley androgenesis. Plant Cell Rep 33:385–392
Mendoza MG, Kaeppler HF (2002) Auxin and sugar effects on callus induction and plant regeneration frequencies from mature embryos of wheat (Triticum aestivum L.). In Vitro Cell Dev Biol Plant 38:39–45
Miroshnichenko D, Filippov M, Dolgov S (2009) Effects of daminozide on somatic embryogenesis from immature and mature embryos of wheat. AJCS 3(2):83–94
Miroshnichenko DN, Filippov MV, Dolgov SV (2013) Medium optimization for efficient somatic embryogenesis and in vitro plant regeneration of spring common wheat varieties. Rus Agric Sci 39(1):24–28
Moieni A, Lokos-Toth K, Sarrafi A (1997) Evidence for genetic control and media effect on haploid regeneration in the anther culture of hexaploid wheat (Triticum aestivum L.). Plant Breed 116:502–504
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Murín R, Mészáros K, Nemeček P, Kuna R, Faragó J (2012) Regeneration of immature and mature embryos from diverse sets of wheat genotypes using media containing different auxins. Acta Agron Hung 60(2):97–108
Navarro-Alvarez W, Baenziger PS, Eskridge KM, Shelton DR, Gustafson VD, Hugo M (1994) Effect of sugars in wheat anther culture media. Plant Breed 112:53–62
Odintsova TI, Egorov TA, Musolyamov AKh (2007) Seed defensins from T.kiharae and related species: genome localization of defensin-encoding genes. Biochimie 89:605–612
Özgen M, Birsin MA, Benlioglu B (2015) Biotechnological characterization of a diverse set of wheat progenitors (Aegilops sp. and Triticum sp.) using callus culture parameters. Plant Genet Resour. doi:10.1017/S1479262115000350
Parmar S, Sainger M, Chaudhary D, Jaiwal P (2012) Plant regeneration from mature embryo of commercial Indian bread wheat (Triticum aestivum L.) cultivars. Physiol Mol Biol Plants 18(2):177–183
Pastori GM, Wilkinson MD, Steele SH, Sparks CA, Jones HD, Parry MAJ (2001) Age-dependent transformation frequency in elite wheat varieties. J Exp Bot 52:857–863
Przetakiewicz A, Orczyk W, Nadolska-Orczyk A (2003) The effect of auxin on plant regeneration of wheat, barley and triticale. Plant Cell Tiss Org Cult 73:245–256
Redha A, Talaat A (2008) Improvement of green plant regeneration by manipulation of anther culture induction medium of hexaploid wheat. Plant Cell Tiss Organ Cult 92:141–146
Satyavathi VV, Jauhar PP, Elias EM, Rao MV (2004) Effects of growth regulators on in vitro plant regeneration in durum wheat. Crop Sci 44:1839–1846
Schulze J (2007) Improvements in Cereal Tissue Culture by Thidiazuron: a Review. Fruit Veg Cereal Sci Biotechnol 1(2):64–79
Shan X, Li D, Qu R (2000) Thidiazuron promotes in vitro regeneration of wheat and barley. In Vitro Cell Dev Biol Plant 36:207–210
Sharma VK, Hänsch R, Mendel RR, Schulze J (2005) Mature embryo axis-based high frequency somatic embryogenesis and plant regeneration from multiple cultivars of barley (Hordeum vulgare L.). J Exp Bot 56(417):1913–1922
Sharma V, Hänsch R, Mendel R, Schulze J (2007) Node-derived cultures with high-morphogenic competence in barley and wheat. Plant Cell Tiss Organ Cult 88(1):21–33
Sirkka A, Immonen T (1993) Comparison of callus culture with embryo culture at different times of embryo rescue for primary triticale production. Euphytica 70:185–190
Tamás C, Szucs P, Rakszegi M, Tamás L, Bedő Z (2004) Effect of combined changes in culture medium and incubation conditions on the regeneration from immature embryos of elite varieties of winter wheat. Plant Cell Tiss Org Cult 79:39–44
Terse M, Xynias IN, Gouli-Vavdinoudi E, Roupakias D (2005) Effect of genome, induction medium and temperature pretreatment on green plant production in durum (Triticum turgidum var. durum) × bread wheat (Triticum aestivum L. em Thell) crosses. Acta Physiol Plant 27:641–649
Timonova EM, Leonova IN, Röder MS, Salina EA (2013) Marker-assisted development and characterization of a set of Triticum aestivum lines carrying different introgressions from T. timopheevii genome. Mol Breeding 31:123–136
Utkina LL, Andreev YA, Rogozhin EA, Korostyleva TV, Slavokhotova AA, Oparin PB, Vassilevski AA, Grishin EV, Egorov TA, Odintsova TI (2013) Genes encoding 4-Cys antimicrobial peptides in wheat Triticum kiharae Dorof. et Migush.: multimodular structural organization, instraspecific variability, distribution and role in defense. FEBS J 280:3594–3608
Winfield MO, Karp A, Lazzeri PA, Davey MR (1995) Chromosome 5D instability in cell lines of Triticum tauschii and morphological variation in regenerated plants. Genome 38(4):737–742
Wu H, Doherty A, Jones HD (2009) Agrobacterium-mediated transformation of bread and durum wheat using freshly isolated immature embryos. Methods Mol Biol 478:93–103
Yin GX, Wang YL, She MY, Du LP, Xu HJ, Ma JX, Ye XG (2011) Establishment of a highly efficient regeneration system for the mature embryo culture of wheat. Agric Sci China 10(1):9–17
Yu Y, Wang J, Zhu ML, Wei ZM (2008) Optimization of mature embryo-based high frequency callus induction and plant regeneration from elite wheat cultivars grown in China. Plant Breed 127:249–255
Zale JM, Borchardt-Wier H, Kidwell KK, Steber CM (2004) Callus induction and plant regeneration from mature embryos of a diverse set of wheat genotypes. Plant Cell Tiss Org Cult 76:277–281
Zhou H, Konzak CF (1992) Genetic control of green plant regeneration from anther culture of wheat. Genome 35:957–961
Zorinyants SE, Smolenskaya IN (1998) Long-term suspension culture of Triticum timopheevii cells: 2. Kinetics of cell population. Russ J Plant Physl 45:82–85
Zorinyants SE, Nosov AV, Badaeva ED, Smolenskaya IN, Badaev NS (1995) Cytogenetic analysis of a long-term Triticum timopheevii (Zhuk.) Zhuk. cell suspension culture. Plant Breed 114:219–225
Zorinyants SE, Smolenskaya IN, Nosov AV, Badaeva ED (1998) Long-term suspension culture of Triticum timopheevii cells: 1. Reorganization of genome. Russ J Plant Physl 45:74–81
Acknowledgments
We are grateful for support from the Ministry of Science and Education of Russian Federation, Grant No.14.M04.12.0015.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miroshnichenko, D., Chernobrovkina, M. & Dolgov, S. Somatic embryogenesis and plant regeneration from immature embryos of Triticum timopheevii Zhuk. and Triticum kiharae Dorof. et Migusch, wheat species with G genome. Plant Cell Tiss Organ Cult 125, 495–508 (2016). https://doi.org/10.1007/s11240-016-0965-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-0965-x