Abstract
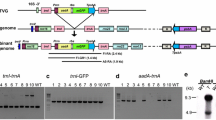
Homologous recombination within plastids directs plastid genome transformation for foreign gene expression and study of plastid gene function. Though transgenes are generally efficiently targeted to their desired insertion site, unintended homologous recombination events have been observed during plastid transformation. To understand the nature and abundance of these recombination events, we analyzed transplastomic tobacco lines derived from three different plastid transformation vectors utilizing two different loci for foreign gene insertion. Two unintended recombinant plastid DNA species were formed from each regulatory plastid DNA element included in the transformation vector. Some of these recombinant DNA species accumulated to as much as 10–60% of the amount of the desired integrated transgenic sequence in T0 plants. Some of the recombinant DNA species undergo further, “secondary” recombination events, resulting in an even greater number of recombinant plastid DNA species. The abundance of novel recombinant DNA species was higher in T0 plants than in T1 progeny, indicating that the ancillary recombination events described here may have the greatest impact during selection and regeneration of transformants. A line of transplastomic tobacco was identified containing an antibiotic resistance gene unlinked from the intended transgene insertion as a result of an unintended recombination event, indicating that the homologous recombination events described here may hinder efficient recovery of plastid transformants containing the desired transgene.








Similar content being viewed by others
References
Ansell SW, Schneider H, Pedersen N, Grundmann M, Russell SJ, Vogel JC (2007) Recombination diversifies chloroplast trnF pseudogenes in Arabidopsis lyrata. J Evol Biol 20:2400–2411. doi:10.1111/j.1420-9101.2007.01397.x
Blowers AD, Bogorad l, Shark KB, Sanford JC (1989) Studies on Chlamydomonas chloroplast transformation: foreign DNA can be stably maintained in the chromosome. Plant Cell 1:123–132
Bohne AV, Ruf S, Borner T, Bock R (2007) Faithful transcription initiation from a mitochondrial promoter in transgenic plastids. Nucleic Acids Res 35:7256–7266. doi:10.1093/nar/gkm679
Bradley RD, Hillis DM (1997) Recombinant DNA sequences generated by PCR amplification. Mol Biol Evol 14:592–593
Buhot l, Horvàth E, Medgyesy P, Lerbs-Mache S (2006) Hybrid transcription system for controlled plastid transgene expression. Plant J 46:700–707. doi:10.1111/j.1365-313X.2006.02718.x
Cerutti H, Osman M, Grandoni P, Jagendorf AT (1992) A homolog of Escherichia coli RecA protein in plastids of higher plants. Proc Natl Acad Sci USA 89:8068–8072. doi:10.1073/pnas.89.17.8068
Chakrabarti SK, Lutz KA, Lertwiriyawong B, Svab Z, Maliga P (2006) Expression of the cry9Aa2 B.t. gene in tobacco chloroplasts confers resistance to potato tuber moth. Transgenic Res 15:481–488. doi:10.1007/s11248-006-0018-z
Corneille S, Lutz KA, Azhagiri AK, Maliga P (2003) Identification of functional lox sites in the plastid genome. Plant J 35:753–762. doi:10.1046/j.1365-313X.2003.01845.x
Gray BN, Ahner BA, Hanson MR (2009) High-level bacterial cellulase accumulation in chloroplast-transformed tobacco mediated by downstream box fusions. Biotechnol Bioeng 102:1045–1054. doi:10.1002/bit.22156
Ho SN, Hunt HD, Horton RM, Pullen JK, Pease LR (1989) Site-directed mutagenesis by overlap extension using the polymerase chain reaction. Gene 77:51–59. doi:10.1016/0378-1119(89)90358-2
Iamtham S, Day A (2000) Removal of antibiotic resistance genes from transgenic tobacco plastids. Nat Biotechnol 18:1172–1176. doi:10.1038/81161
Kanno A, Watanbe N, Nakamura I, Hirai A (1993) Variations in chloroplast DNA from rice (Oryza sativa): differences between deletions mediated by short direct-repeat sequences within a single species. Theor Appl Genet 86:579–584. doi:10.1007/BF00838712
Kawata M, Harada T, Shimamoto Y, Oono K, Takaiwa F (1997) Short inverted repeats function as hotspots of intermolecular recombination giving rise to oligomers of deleted plastid DNAs (ptDNAs). Curr Genet 31:179–184. doi:10.1007/s002940050193
Khakhlova O, Bock R (2006) Elimination of deleterious mutations in plastid genomes by gene conversion. Plant J 46:85–94. doi:10.1111/j.1365-313X.2006.02673.x
Khan MS, Hameed W, Nozoe M, Shiina T (2007) Disruption of the psbA gene by the copy correction mechanism reveals that the expression of plastid-encoded genes is regulated by photosynthesis activity. J Plant Res 120:421–430. doi:10.1007/s10265-007-0082-3
Klaus SMJ, Huang F-C, Golds TJ, Koop H-U (2004) Generation of marker-free plastid transformants using a transiently cointegrated selection gene. Nat Biotechnol 22:225–229. doi:10.1038/nbt933
Li W, Ruf S, Bock R (2006) Constancy of organellar genome copy numbers during leaf development and senescence in higher plants. Mol Genet Genomics 275:185–192. doi:10.1007/s00438-005-0075-7
Lilly JW, Havey MJ, Jackson SA, Jiang J (2001) Cytogenomic analyses reveal the structural plasticity of the chloroplast genome in higher plants. Plant Cell 13:245–254
McBride KE, Schaaf DJ, Daley M, Stalker DM (1994) Controlled expression of plastid transgenes in plants based on a nuclear DNA-encoded and plastid-targeted T7 RNA polymerase. Proc Natl Acad Sci USA 91:7301–7305. doi:10.1073/pnas.91.15.7301
McCabe MS, Klaas M, Gonzalez-Rabade N, Poage M, Badillo-Corona JA, Zhou F, Karcher D, Bock R, Gray JC, Dix PH (2008) Plastid transformation of high-biomass tobacco variety Maryland Mammoth for production of human immunodeficiency virus type 1 (HIV-1) p24 antigen. Plant Biotechnol J 6:914–929. doi:10.1111/j.1467-7652.2008.00365.x
Nakazato E, Fukuzawa H, Tabata S, Takahashi H, Tanaka K (2003) Identification and expression analysis of cDNA encoding a chloroplast recombination protein REC1, the chloroplast RecA homologue in Chlamydomonas reinhardtii. Biosci Biotechnol Biochem 67:608–2613. doi:10.1271/bbb.67.2608
Newell CA, Birch-Machin I, Hibberd JM, Gray JC (2003) Expression of green fluorescent protein from bacterial and plastid promoters in tobacco chloroplasts. Transgenic Res 12:631–634. doi:10.1023/A:1025812309254
Odom OW, Baek K-H, Dani RN, Herrin DL (2008) Chlamydomonas chloroplasts can use short dispersed repeats and multiple pathways to repair a double-strand break in the genome. Plant J 53:842–853. doi:10.1111/j.1365-313X.2007.03376.x
Palmer JD (1983) Chloroplast DNA exists in two orientations. Nature 301:92–93. doi:10.1038/301092a0
Peterson BC, Hashimoto H, Rownd RH (1982) Cointegrate formation between homologous plasmids in Escherichia coli. J Bacteriol 151:1086–1094
Reed ML, Wilson SK, Sutton CA, Hanson MR (2001) High-level expression of a synthetic red-shifted GFP coding region incorporated into transgenic chloroplasts. Plant J 27:257–265. doi:10.1046/j.1365-313x.2001.01088.x
Rogalski M, Ruf S, Bock R (2006) Tobacco plastid ribosomal protein S18 is essential for cell survival. Nucleic Acids Res 34:4537–4545. doi:10.1093/nar/gkl634
Rogalski M, Karcher D, Bock R (2008a) Superwobbling facilitates translation with reduced tRNA sets. Nat Struct Mol Biol 15:192–198. doi:10.1038/nsmb.1370
Rogalski M, Schöttler MA, Thiele W, Schulze WX, Bock R (2008b) Rpl33, a nonessential plastid-encoded ribosomal protein in tobacco, is required under cold stress conditions. Plant Cell 20:2221–2237. doi:10.1105/tpc.108.060392
Ruf S, Biehler K, Bock R (2000) A small chloroplast-encoded protein as a novel architectural component of the light-harvesting antenna. J Cell Biol 149:369–378. doi:10.1083/jcb.149.2.369
Staub JM, Maliga P (1992) Long regions of homologous DNA are incorporated into the tobacco plastid genome by transformation. Plant Cell 4:39–45
Staub JM, Maliga P (1994) Extrachromosomal elements in tobacco plastids. Proc Natl Acad Sci USA 91:7468–7472. doi:10.1073/pnas.91.16.7468
Staub JM, Maliga P (1995) Marker rescue from the Nicotiana tabacum plastid genome using a plastid/Escherichia coli shuttle vector. Mol Gen Genet 249:37–42. doi:10.1007/BF00290233
Svab Z, Maliga P (1991) Mutation proximal to the tRNA binding region of the Nicotiana plastid 16S rRNA confers resistance to spectinomycin. Mol Gen Genet 228:316–319. doi:10.1007/BF00282483
Svab Z, Maliga P (1993) High-frequency plastid transformation in tobacco by selection for a chimeric aadA gene. Proc Natl Acad Sci USA 90:913–917. doi:10.1073/pnas.90.3.913
Yu LX, Gray BN, Rutzke CJ, Walker LP, Wilson DB, Hanson MR (2007) Expression of thermostable microbial cellulases in the chloroplasts of nicotine-free tobacco. J Biotechnol 131:326–369. doi:10.1016/j.jbiotec.2007.07.942
Zhou F, Badillo-Corona J, Karcher D, Gonzalez-Rabade N, Piepenburg K, Borchers AM, Maloney AP, Kavanagh TA, Gray JC, Bock R (2008) High-level expression of human immunodeficiency virus antigens from the tobacco and tomato plastid genomes. Plant Biotechnol J 6:897–913. doi:10.1111/j.1467-7652.2008.00356.x
Acknowledgments
This work was supported by the United States Department of Agriculture [USDA NRI 2007-02133 to B.A.A. and M.R.H.]; and the National Science Foundation [Graduate Research Fellowship, B·N.G.].
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gray, B.N., Ahner, B.A. & Hanson, M.R. Extensive homologous recombination between introduced and native regulatory plastid DNA elements in transplastomic plants. Transgenic Res 18, 559–572 (2009). https://doi.org/10.1007/s11248-009-9246-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-009-9246-3