Abstract
Introduction
Lipids are a diverse group of macromolecules that occur in rice grains and are known to impact rice grain properties. Identifying the relationships between specific lipids and traits of quality is important to improve varietal selection for high quality rice.
Objectives
Using untargeted lipidomics, this study aims to understand the role of lipids on different traits of quality by identifying the genotypic effect of lipids and their impact on traits of cooking and eating quality of a rice mapping population.
Methods
Lipids from milled rice grains of three sets of rice samples were screened by ultra-performance liquid chromatography-mass spectrometry (UPLC-MS) in the positive ionisation mode. Lipid features were putatively identified using analytical standards and online databases. Multivariate statistics were carried out to identify the lipid profile of varieties across three experiments. Correlation analysis was carried out between lipid features and 12 quality traits across a rice mapping population that segregates for grain physical and texture-associated traits.
Results
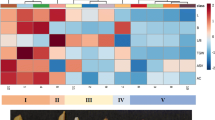
Thousands of features in rice grain lipids were detected, and were grouped into six categories—fatty acyls, glycerolipids, glycerophospholipids, sphingolipids, sterol lipids and prenol lipids. A strong genotypic basis for the lipid profile was observed among the four varieties grown under five nitrogen treatments. Clear differentiation in lipid profiles between waxy and non-waxy rice was observed. Strong correlations were observed for putative lipids that form the amylose–lipid complex and with amylose content and viscosity parameters.
Conclusions
This study demonstrates the strength of untargeted lipidomics in putatively determining features that differentiate varieties from each other, and reveals the role of specific lipids on the physical and textural quality of rice.





Similar content being viewed by others
References
Azudin, M. N., & Morrison, W. R. (1986). Non-starch lipids and starch lipids in milled rice. Journal of Cereal Science,4(1), 23–31. https://doi.org/10.1016/S0733-5210(86)80004-2.
Baisted, D. J. (1981). Turnover of starch-bound lysophosphatidylcholine in germinating barley. Phytochemistry,20(5), 985–988. https://doi.org/10.1016/0031-9422(81)83013-0.
Bergman, C., Goffman, F., & Chen, M. H. (2011). Evaluation of antioxidant, lipid, and protein fractions of accessions of Oryza species. Cereal Chemistry,88(3), 283–290. https://doi.org/10.1094/CCHEM-04-10-0057.
Biliaderis, C. G., & Juliano, B. O. (1993). Thermal and mechanical properties of concentrated rice starch gels of varying composition. Food Chemistry,48(3), 243–250. https://doi.org/10.1016/0308-8146(93)90134-2.
Buléon, A., Colonna, P., Planchot, V., & Ball, S. (1998). Starch granules: Structure and biosynthesis. International Journal of Biological Macromolecules,23(2), 85–112. https://doi.org/10.1016/S0141-8130(98)00040-3.
Chapman, K. D., & Ohlrogge, J. B. (2012). Compartmentation of triacylglycerol accumulation in plants. The Journal of Biological Chemistry,287(4), 2288. https://doi.org/10.1074/jbc.R111.290072.
Choudhury, N. H., & Juliano, B. O. (1980). Effect of amylose content on the lipids of mature rice grain. Phytochemistry,19(7), 1385–1389. https://doi.org/10.1016/0031-9422(80)80179-8.
Concepcion, J. C. T., Ouk, S., Ouk, M., & Fitzgerald, M. A. (2019). Mapping quantitative traits for grain physical and textural quality in Cambodian Jasmine rice PRD. Euphytica,215(7), 135. https://doi.org/10.1007/s10681-019-2455-9.
Concepcion, J. C. T., Ouk, S., Riedel, A., Calingacion, M., Zhao, D., Ouk, M., et al. (2018). Quality evaluation, fatty acid analysis and untargeted profiling of volatiles in Cambodian rice. Food Chemistry,240, 1014–1021. https://doi.org/10.1016/j.foodchem.2017.08.019.
Copeland, L., Blazek, J., Salman, H., & Tang, M. C. (2009). Form and functionality of starch. Food Hydrocolloids,23(6), 1527–1534. https://doi.org/10.1016/j.foodhyd.2008.09.016.
Cuevas, R. P., & Fitzgerald, M. A. (2012). Genetic diversity of rice grain quality. In M. Caliskan (Ed.), Genetic diversity in plants (pp. 285–310). London: IntechOpen.
Cutignano, A., Luongo, E., Nuzzo, G., Pagano, D., Manzo, E., Sardo, A., et al. (2016). Profiling of complex lipids in marine microalgae by UHPLC/tandem mass spectrometry. Algal Research,17, 348–358. https://doi.org/10.1016/j.algal.2016.05.016.
Dunn, W. B., Broadhurst, D., Begley, P., Zelena, E., Francis-McIntyre, S., Anderson, N., et al. (2011). Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nature Protocols,6, 1060. https://doi.org/10.1038/nprot.2011.335.
Fahy, E., Subramaniam, S., Brown, H. A., Glass, C. K., Merrill, A. H., Murphy, R. C., et al. (2005). A comprehensive classification system for lipids. Journal of Lipid Research,46(5), 839–862. https://doi.org/10.1194/jlr.E400004-JLR200.
Fahy, E., Subramaniam, S., Murphy, R. C., Nishijima, M., Raetz, C. R. H., Shimizu, T., et al. (2009). Update of the LIPID MAPS comprehensive classification system for lipids. Journal of Lipid Research,50(Supplement), S9–S14.
Fitzgerald, M. A., Martin, M., Ward, R. M., Park, W. D., & Shead, H. J. (2003). Viscosity of rice four: A rheological and biological study. Journal of Agricultural and Food Chemistry,51(8), 2295–2299. https://doi.org/10.1021/jf020574i.
Fitzgerald, M. A., McCouch, S. R., & Hall, R. D. (2009). Not just a grain of rice: The quest for quality. Trends in Plant Science,14(3), 133–139. https://doi.org/10.1016/j.tplants.2008.12.004.
Galliard, T. (1983). Starch-lipid complexes and other non-starch components of starch granules in cereal grains. In C. Nozzolillo, P. J. Lea, & F. A. Loewus (Eds.), Mobilization of reserves in germination (pp. 111–136). Boston, MA: Springer.
Gao, X., Zhang, Q., Meng, D., Isaac, G., Zhao, R., Fillmore, T. L., et al. (2012). A reversed-phase capillary ultra-performance liquid chromatography–mass spectrometry (UPLC-MS) method for comprehensive top-down/bottom-up lipid profiling. Analytical and Bioanalytical Chemistry,402(9), 2923–2933. https://doi.org/10.1007/s00216-012-5773-5.
Gayral, M., Bakan, B., Dalgalarrondo, M., Elmorjani, K., Delluc, C., Brunet, S., et al. (2015). Lipid partitioning in maize (Zea mays L.) endosperm highlights relationships among starch lipids, amylose, and vitreousness. Journal of Agricultural and Food Chemistry,63(13), 3551–3558. https://doi.org/10.1021/acs.jafc.5b00293.
German, J., Roberts, M.-A., & Watkins, S. (2003). Genomics and metabolomics as markers for the interaction of diet and health: Lessons from lipids. The Journal of Nutrition,133(6S-1), S2078–S2083. https://doi.org/10.1093/jn/133.6.2078S.
Godet, M. C., Buléon, A., Tran, V., & Colonna, P. (1993). Structural features of fatty acid-amylose complexes. Carbohydrate Polymers,21(2), 91–95. https://doi.org/10.1016/0144-8617(93)90003-M.
Halade, G., Dorbane, A., Ingle, K., Kain, V., Schmitter, J.-M., & Rhourri-Frih, B. (2018). Comprehensive targeted and non-targeted lipidomics analyses in failing and non-failing heart. Analytical and Bioanalytical Chemistry,410(7), 1965–1976. https://doi.org/10.1007/s00216-018-0863-7.
Isaac, G., McDonald, S., & Astarita, G. (2011). Lipid separation using UPLC with charged surface hybrid technology. Waters Application Note (720004107en).
Kind, T., Liu, K.-H., Lee, D. Y., DeFelice, B., Meissen, J. K., & Fiehn, O. (2013). LipidBlast in silico tandem mass spectrometry database for lipid identification. Nature Methods,10, 755. https://doi.org/10.1038/nmeth.2551.
Li, Y., Han, D., Hu, G., Sommerfeld, M., & Hu, Q. (2010). Inhibition of starch synthesis results in overproduction of lipids in Chlamydomonas reinhardtii. Biotechnology and Bioengineering,107(2), 258–268. https://doi.org/10.1002/bit.22807.
Liu, L., Waters, D. L. E., Rose, T. J., Bao, J., & King, G. J. (2013). Phospholipids in rice: Significance in grain quality and health benefits: A review. Food Chemistry,139(1–4), 1133–1145. https://doi.org/10.1016/j.foodchem.2012.12.046.
López, C. A., de Vries, A. H., & Marrink, S. J. (2012). Amylose folding under the influence of lipids. Carbohydrate Research,364, 1–7. https://doi.org/10.1016/j.carres.2012.10.007.
Luo, J., Jobling, S., Millar, A., Morell, M., & Li, Z. (2015). Allelic effects on starch structure and properties of six starch biosynthetic genes in a rice recombinant inbred line population. Rice,8(1), 1–13. https://doi.org/10.1186/s12284-015-0046-5.
Manier, S. K., Keller, A., Schäper, J., & Meyer, M. R. (2019). Untargeted metabolomics by high resolution mass spectrometry coupled to normal and reversed phase liquid chromatography as a tool to study the in vitro biotransformation of new psychoactive substances. Scientific Reports,9(1), 2741. https://doi.org/10.1038/s41598-019-39235-w.
Mitchell, C. R. (2009). Rice starches: Production and properties. In J. BeMiller & R. Whistler (Eds.), Starch (3rd ed., Chap. 13, pp. 569–578). San Diego: Academic Press.
Morrison, W. R. (1977). Cereal lipids. Proceedings of the Nutrition Society,36(02), 143–148. https://doi.org/10.1079/PNS19770026.
Morrison, W. R. (1983). Acyl lipids in cereals. In P. Barnes (Ed.), Lipids in cereal technology (pp. 403–438). London, England: Academic Press.
Morrison, W. R., Law, R. V., & Snape, C. E. (1993a). Evidence for inclusion complexes of lipids with v-amylose in maize, rice and oat starches. Journal of Cereal Science,18(2), 107–109. https://doi.org/10.1006/jcrs.1993.1039.
Morrison, W. R., Tester, R. F., Snape, C. E., Law, R., & Gidley, M. J. (1993b). Swelling and gelatinization of cereal starches. IV. Some effects of lipid-complexed amylose and free amylose in waxy and normal barley starches. Cereal Chemistry,70(4), 385–391.
Mumm, R., Hageman, J. A., Calingacion, M. N., de Vos, R. C. H., Jonker, H. H., Erban, A., et al. (2016). Multi-platform metabolomics analyses of a broad collection of fragrant and non-fragrant rice varieties reveals the high complexity of grain quality characteristics. Metabolomics,12(2), 38. https://doi.org/10.1007/s11306-015-0925-1.
Paglia, G., & Astarita, G. (2017). Metabolomics and lipidomics using traveling-wave ion mobility mass spectrometry. Nature Protocols,12, 797. https://doi.org/10.1038/nprot.2017.013.
Philpot, K., Martin, M., Butardo, V., Willoughby, D., & Fitzgerald, M. (2006). Environmental factors that affect the ability of amylose to contribute to retrogradation in gels made from rice flour. Journal of Agricultural and Food Chemistry,54(14), 5182–5190. https://doi.org/10.1021/jf060666y.
Putseys, J. A., Derde, L. J., Lamberts, L., Ostman, E., Björck, I. M., & Delcour, J. A. (2010). Functionality of short chain amylose-lipid complexes in starch-water systems and their impact on in vitro starch degradation. Journal of Agricultural and Food Chemistry,58(3), 1939. https://doi.org/10.1021/jf903523h.
R Core Team, R. (2013). R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria
Resurreccion, A. P., & Juliano, B. O. (1975). Fatty acid composition of rice oils. Journal of the Science of Food and Agriculture,26(4), 437–439. https://doi.org/10.1002/jsfa.2740260409.
Shevchenko, A., & Simons, K. (2010). Lipidomics: Coming to grips with lipid diversity. Nature Reviews Molecular Cell Biology,11(8), 593–598. https://doi.org/10.1038/nrm2934.
Sumner, L. W., Amberg, A., Barrett, D., Beale, M. H., Beger, R., Daykin, C. A., et al. (2007). Proposed minimum reporting standards for chemical analysis. Metabolomics, 3(3), 211–221. https://doi.org/10.1007/s11306-007-0082-2.
Tenenboim, H., Burgos, A., Willmitzer, L., & Brotman, Y. (2016). Using lipidomics for expanding the knowledge on lipid metabolism in plants. Biochimie,130, 91–96. https://doi.org/10.1016/j.biochi.2016.06.004.
Tong, C., Liu, L., Waters, D. L. E., Huang, Y., & Bao, J. (2015). The contribution of lysophospholipids to pasting and thermal properties of nonwaxy rice starch. Carbohydrate Polymers,133, 187–193. https://doi.org/10.1016/j.carbpol.2015.07.001.
Tufvesson, F., Wahlgren, M., & Eliasson, A.-C. (2003). Formation of amylose-lipid complexes and effects of temperature treatment. Part 2. Fatty acids. Starch Stärke,55(3–4), 138–149. https://doi.org/10.1002/star.200390028.
Wang, S., Wang, J., Yu, J., & Wang, S. (2016). Effect of fatty acids on functional properties of normal wheat and waxy wheat starches: A structural basis. Food Chemistry,190, 285–292. https://doi.org/10.1016/j.foodchem.2015.05.086.
Wasaki, J., Yonetani, R., Kuroda, S., Shinano, T., Yazaki, J., Fujii, F., et al. (2003). Transcriptomic analysis of metabolic changes by phosphorus stress in rice plant roots. Plant, Cell & Environment,26(9), 1515–1523. https://doi.org/10.1046/j.1365-3040.2003.01074.x.
Welti, R., Shah, J., Li, W., Li, M., Chen, J., Burke, J. J., et al. (2007). Discerning biological function by profiling plant complex lipids using mass spectrometry. Frontiers in Biosciences,12, 2494.
Wenk, M. R. (2010). Lipidomics: New tools and applications. Cell,143(6), 888–895. https://doi.org/10.1016/j.cell.2010.11.033.
Wickham, H. (2016). ggplot2: Elegant graphics for data analysis. Cham: Springer.
Yoshida, H., Kuriyama, I., Tomiyama-Sakamoto, Y., & Mizushina, Y. (2012). Profiles of lipid components, fatty acids and triacylglycerol molecular species in lipids of rice bran cultivars. Food Science and Technology Research,18(2), 219. https://doi.org/10.3136/fstr.18.219.
Yoshida, H., Tanigawa, T., Yoshida, N., Kuriyama, I., Tomiyama, Y., & Mizushina, Y. (2011). Lipid components, fatty acid distributions of triacylglycerols and phospholipids in rice brans. Food Chemistry,129(2), 479–484. https://doi.org/10.1016/j.foodchem.2011.04.102.
Yu, D., Rupasinghe, T. W. T., Boughton, B. A., Natera, S. H. A., Hill, C. B., Tarazona, P., et al. (2018). A high-resolution HPLC-QqTOF platform using parallel reaction monitoring for in-depth lipid discovery and rapid profiling. Analytica Chimica Acta. https://doi.org/10.1016/j.aca.2018.03.062.
Acknowledgements
The authors would like to thank Dr Paul Goulding of Waters Corporation for his technical assistance with the lipidomics data of the mapping population. Christopher Proud for his assistance in creating the Manhattan plots in R and for reading the manuscript. Dr Jennifer Waanders of the School of Agriculture and Food Sciences at the University of Queensland for her assistance in using the UPLC-MS. Margrit Martin of the Yanco Agricultural Research Centre for providing samples used in the nitrogen experiment (Experiment 1). The International Rice Research Institute for providing the rice grain samples of the diverse rice varieties. Bingyue Wang for the assistance in sample preparation. We also like to thank the Australian Research Council—Industry Transformation Training Centre (Agents of Change) IC130100011 and the Australian Centre for International Agricultural Research (ACIAR) for funding this research and for the John Allwright Fellowship of JCTC.
Author information
Authors and Affiliations
Contributions
JCTC performed the analytical experiments, data processing, metabolite annotation, PCA, presentation and interpretation of results, MC and MAF optimised the method for the UPLC-MS, JCTC performed the majority of the writing, MAF, MC and MJG provided guidance to the lead author. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Research involved human and animal participants
This article does not contain any studies with human and/or animal participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Concepcion, J.C.T., Calingacion, M., Garson, M.J. et al. Lipidomics reveals associations between rice quality traits. Metabolomics 16, 54 (2020). https://doi.org/10.1007/s11306-020-01670-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-020-01670-6