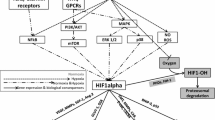
Abstract
There is a need to measure tumor hypoxia in assessing the aggressiveness of tumor and predicting the outcome of therapy. A number of invasive and noninvasive techniques have been exploited to measure tumor hypoxia, including polarographic needle electrodes, immunohistochemical staining, radionuclide imaging (positron emission tomography [PET] and single-photon emission computed tomography [SPECT]), magnetic resonance imaging (MRI), optical imaging (bioluminescence and fluorescence), and so on. This review article summarizes and discusses the pros and cons of each currently available method for measuring tissue oxygenation. Special emphasis was placed on noninvasive imaging hypoxia with emerging new agents and new imaging technologies to detect the molecular events that are relevant to tumor hypoxia.




Similar content being viewed by others
References
Vaupel P, Mayer A (2007) Hypoxia in cancer: significance and impact on clinical outcome. Cancer Metastasis Rev 26:225–239
Hu M, Polyak K (2008) Microenvironmental regulation of cancer development. Curr Opin Genet Dev 18:27–34
Fukumura D, Jain RK (2007) Tumor microvasculature and microenvironment: targets for anti-angiogenesis and normalization. Microvasc Res 74:72–84
Vaupel P, Mayer A, Hockel M (2004) Tumor hypoxia and malignant progression. Methods Enzymol 381:335–354
Gray LH, Conger AD, Ebert M, Hornsey S, Scott OCA (1953) The concentration of oxygen dissolved in tissues at the time of irradiation as a factor in radiotherapy. Br J Radiol 26:638–648
Matthews NE, Adams MA, Maxwell LR, Gofton TE, Graham CH (2001) Nitric oxide-mediated regulation of chemosensitivity in cancer cells. J Natl Cancer Inst 93:1879–1885
Nordsmark M, Bentzen SM, Rudat V et al (2005) Prognostic value of tumor oxygenation in 397 head and neck tumors after primary radiation therapy. An international multi-center study. Radiother Oncol 77:18–24
Brizel DM, Sibley GS, Prosnitz LR, Scher RL, Dewhirst MW (1997) Tumor hypoxia adversely affects the prognosis of carcinoma of the head and neck. Int J Radiat Oncol Biol Phys 38:285–289
Hockel M, Vorndran B, Schlenger K, Baussmann E, Knapstein PG (1993) Tumor oxygenation: a new predictive parameter in locally advanced cancer of the uterine cervix. Gynecol Oncol 51:141–149
Nordsmark M, Loncaster J, Chou SC et al (2001) Invasive oxygen measurements and pimonidazole labeling in human cervix carcinoma. Int J Radiat Oncol Biol Phys 49:581–586
Nordsmark M, Overgaard J (2000) A confirmatory prognostic study on oxygenation status and loco-regional control in advanced head and neck squamous cell carcinoma treated by radiation therapy. Radiother Oncol 57:39–43
Evans SM, Judy KD, Dunphy I et al (2004) Hypoxia is important in the biology and aggression of human glial brain tumors. Clin Cancer Res 10:8177–8184
Powell ME, Collingridge DR, Saunders MI et al (1999) Improvement in human tumour oxygenation with carbogen of varying carbon dioxide concentrations. Radiother Oncol 50:167–171
Gatenby RA, Moldofsky PJ, Weiner LM (1988) Metastatic colon cancer: correlation of oxygen levels with I-131 F(ab’)2 uptake. Radiology 166:757–759
Brizel DM, Scully SP, Harrelson JM et al (1996) Radiation therapy and hyperthermia improve the oxygenation of human soft tissue sarcomas. Cancer Res 56:5347–5350
Pauwels EK, Mariani G (2007) Assessment of tumor tissue oxygenation: agents, methods and clinical significance. Drug News Perspect 20:619–626
Rumsey WL, Vanderkooi JM, Wilson DF (1988) Imaging of phosphorescence—a novel method for measuring oxygen distribution in perfused tissue. Science 241:1649–1651
Vinogradov SA, Grosul P, Rozhkov V et al (2003) Oxygen distributions in tissue measured by phosphorescence quenching. Adv Exp Med Biol 510:181–185
Lebedev AY, Cheprakov AV, Sakadzic S et al (2009) Dendritic phosphorescent probes for oxygen imaging in biological systems. ACS Appl Mater Interfaces 1:1292–1304
Pennekamp CW, Bots ML, Kappelle LJ, Moll FL, de Borst GJ (2009) The value of near-infrared spectroscopy measured cerebral oximetry during carotid endarterectomy in perioperative stroke prevention. A review. Eur J Vasc Endovasc Surg 38:539–545
Jobsis FF (1977) Non-invasive, infra-red monitoring of cerebral O2 sufficiency, blood volume, HbO2-Hb shifts and blood flow. Acta Neurol Scand Suppl 64:452–453
Hull EL, Conover DL, Foster TH (1999) Carbogen-induced changes in rat mammary tumour oxygenation reported by near infrared spectroscopy. Br J Cancer 79:1709–1716
Kim JG, Liu H (2008) Investigation of biphasic tumor oxygen dynamics induced by hyperoxic gas intervention: the dynamic phantom approach. Appl Opt 47:242–252
Ljungkvist AS, Bussink J, Kaanders JH, van der Kogel AJ (2007) Dynamics of tumor hypoxia measured with bioreductive hypoxic cell markers. Radiat Res 167:127–145
Chapman JD (1979) Hypoxic sensitizers—implications for radiation therapy. N Engl J Med 301:1429–1432
Raleigh JA, Calkins-Adams DP, Rinker LH et al (1998) Hypoxia and vascular endothelial growth factor expression in human squamous cell carcinomas using pimonidazole as a hypoxia marker. Cancer Res 58:3765–3768
Evans SM, Hahn S, Pook DR et al. Detection of hypoxia in human squamous cell carcinoma by EF5 binding. Cancer Res 60:2018–2024
Evans SM, Koch CJ (2003) Prognostic significance of tumor oxygenation in humans. Cancer Lett 195:1–16
Rajendran JG, Schwartz DL, O’Sullivan J et al (2006) Tumor hypoxia imaging with [F-18] fluoromisonidazole positron emission tomography in head and neck cancer. Clin Cancer Res 12:5435–5441
Raleigh JA, Chou SC, Arteel GE, Horsman MR (1999) Comparisons among pimonidazole binding, oxygen electrode measurements, and radiation response in C3H mouse tumors. Radiat Res 151:580–589
Toma-Dasu I, Dasu A, Brahme A (2009) Quantifying tumour hypoxia by PET imaging—a theoretical analysis. Adv Exp Med Biol 645:267–272
Massoud TF, Gambhir SS (2007) Integrating noninvasive molecular imaging into molecular medicine: an evolving paradigm. Trends Mol Med 13:183–191
Willmann JK, van Bruggen N, Dinkelborg LM, Gambhir SS (2008) Molecular imaging in drug development. Nat Rev Drug Discov 7:591–607
Swartz HM, Clarkson RB (1998) The measurement of oxygen in vivo using EPR techniques. Phys Med Biol 43:1957–1975
Matsumoto K, English S, Yoo J et al (2004) Pharmacokinetics of a triarylmethyl-type paramagnetic spin probe used in EPR oximetry. Magn Reson Med 52:885–892
Krohn KA, Link JM, Mason RP (2008) Molecular imaging of hypoxia. J Nucl Med 49(Suppl 2):129S–148S
Wang X, Xie X, Ku G, Wang LV, Stoica G (2006) Noninvasive imaging of hemoglobin concentration and oxygenation in the rat brain using high-resolution photoacoustic tomography. J Biomed Opt 11:024015
Padhani A (2010) Science to practice: what does MR oxygenation imaging tell us about human breast cancer hypoxia? Radiology 254:1–3
Howe FA, Robinson SP, McIntyre DJ, Stubbs M, Griffiths JR (2001) Issues in flow and oxygenation dependent contrast (FLOOD) imaging of tumours. NMR Biomed 14:497–506
Stubbs M (1999) Application of magnetic resonance techniques for imaging tumour physiology. Acta Oncol 38:845–853
Tumkur SM, Vu AT, Li LP, Pierchala L, Prasad PV (2006) Evaluation of intra-renal oxygenation during water diuresis: a time-resolved study using BOLD MRI. Kidney Int 70:139–143
O’Connor JP, Naish JH, Parker GJ et al (2009) Preliminary study of oxygen-enhanced longitudinal relaxation in MRI: a potential novel biomarker of oxygenation changes in solid tumors. Int J Radiat Oncol Biol Phys 75:1209–1215
Mason RP (2006) Non-invasive assessment of kidney oxygenation: a role for BOLD MRI. Kidney Int 70:10–11
Thomas SR, Pratt RG, Millard RW et al (1996) In vivo PO2 imaging in the porcine model with perfluorocarbon F-19 NMR at low field. Magn Reson Imaging 14:103–114
Mason RP, Shukla H, Antich PP (1993) In vivo oxygen tension and temperature: simultaneous determination using 19F NMR spectroscopy of perfluorocarbon. Magn Reson Med 29:296–302
Zhao D, Ran S, Constantinescu A, Hahn EW, Mason RP (2003) Tumor oxygen dynamics: correlation of in vivo MRI with histological findings. Neoplasia 5:308–318
van der Sanden BP, Heerschap A, Simonetti AW et al. Characterization and validation of noninvasive oxygen tension measurements in human glioma xenografts by 19F-MR relaxometry. Int J Radiat Oncol Biol Phys 44:649–658
McNab JA, Yung AC, Kozlowski P (2004) Tissue oxygen tension measurements in the Shionogi model of prostate cancer using 19F MRS and MRI. Magma 17:288–295
Davda S, Bezabeh T (2006) Advances in methods for assessing tumor hypoxia in vivo: implications for treatment planning. Cancer Metastasis Rev 25:469–480
Yu JX, Kodibagkar VD, Cui W, Mason RP (2005) 19F: a versatile reporter for non-invasive physiology and pharmacology using magnetic resonance. Curr Med Chem 12:819–848
Hunjan S, Zhao D, Constantinescu A et al (2001) Tumor oximetry: demonstration of an enhanced dynamic mapping procedure using fluorine-19 echo planar magnetic resonance imaging in the Dunning prostate R3327-AT1 rat tumor. Int J Radiat Oncol Biol Phys 49:1097–1108
Kwock L, Gill M, McMurry HL et al (1992) Evaluation of a fluorinated 2-nitroimidazole binding to hypoxic cells in tumor-bearing rats by 19F magnetic resonance spectroscopy and immunohistochemistry. Radiat Res 129:71–78
Salmon HW, Siemann DW (2004) Utility of 19F MRS detection of the hypoxic cell marker EF5 to assess cellular hypoxia in solid tumors. Radiother Oncol 73:359–366
Lee CP, Payne GS, Oregioni A et al (2009) A phase I study of the nitroimidazole hypoxia marker SR4554 using 19F magnetic resonance spectroscopy. Br J Cancer 101:1860–1868
Rasey JS, Koh WJ, Evans ML et al (1996) Quantifying regional hypoxia in human tumors with positron emission tomography of [18F]fluoromisonidazole: a pretherapy study of 37 patients. Int J Radiat Oncol Biol Phys 36:417–428
Lehtio K, Eskola O, Viljanen T et al (2004) Imaging perfusion and hypoxia with PET to predict radiotherapy response in head-and-neck cancer. Int J Radiat Oncol Biol Phys 59:971–982
Souvatzoglou M, Grosu AL, Roper B et al (2007) Tumour hypoxia imaging with [18F]FAZA PET in head and neck cancer patients: a pilot study. Eur J Nucl Med Mol Imaging 34:1566–1575
Koh WJ, Rasey JS, Evans ML et al (1992) Imaging of hypoxia in human tumors with [F-18]fluoromisonidazole. Int J Radiat Oncol Biol Phys 22:199–212
Lee ST, Scott AM (2007) Hypoxia positron emission tomography imaging with 18f-fluoromisonidazole. Semin Nucl Med 37:451–461
Gagel B, Reinartz P, Demirel C et al (2006) [18F]fluoromisonidazole and [18F]fluorodeoxyglucose positron emission tomography in response evaluation after chemo-/radiotherapy of non-small-cell lung cancer: a feasibility study. BMC Cancer 6:51
Eschmann SM, Paulsen F, Reimold M et al (2005) Prognostic impact of hypoxia imaging with 18F-misonidazole PET in non-small cell lung cancer and head and neck cancer before radiotherapy. J Nucl Med 46:253–260
Rajendran JG, Mankoff DA, O’Sullivan F et al (2004) Hypoxia and glucose metabolism in malignant tumors: evaluation by [18F]fluoromisonidazole and [18F]fluorodeoxyglucose positron emission tomography imaging. Clin Cancer Res 10:2245–2252
Koh WJ, Bergman KS, Rasey JS et al (1995) Evaluation of oxygenation status during fractionated radiotherapy in human nonsmall cell lung cancers using [F-18]fluoromisonidazole positron emission tomography. Int J Radiat Oncol Biol Phys 33:391–398
Rajendran JG, Wilson DC, Conrad EU et al (2003) [(18)F]FMISO and [(18)F]FDG PET imaging in soft tissue sarcomas: correlation of hypoxia, metabolism and VEGF expression. Eur J Nucl Med Mol Imaging 30:695–704
Bentzen L, Keiding S, Nordsmark M et al (2003) Tumour oxygenation assessed by 18F-fluoromisonidazole PET and polarographic needle electrodes in human soft tissue tumours. Radiother Oncol 67:339–344
Lehtio K, Oikonen V, Gronroos T et al (2001) Imaging of blood flow and hypoxia in head and neck cancer: initial evaluation with [(15)O]H(2)O and [(18)F]fluoroerythronitroimidazole PET. J Nucl Med 42:1643–1652
Yang DJ, Wallace S, Cherif A et al (1995) Development of F-18-labeled fluoroerythronitroimidazole as a PET agent for imaging tumor hypoxia. Radiology 194:795–800
Barthel H, Wilson H, Collingridge DR et al (2004) In vivo evaluation of [18F]fluoroetanidazole as a new marker for imaging tumour hypoxia with positron emission tomography. Br J Cancer 90:2232–2242
Ziemer LS, Evans SM, Kachur AV et al (2003) Noninvasive imaging of tumor hypoxia in rats using the 2-nitroimidazole 18F-EF5. Eur J Nucl Med Mol Imaging 30:259–266
Evans SM, Kachur AV, Shiue CY et al (2000) Noninvasive detection of tumor hypoxia using the 2-nitroimidazole [18F]EF1. J Nucl Med 41:327–336
Koch CJ, Evans SM (2003) Non-invasive PET and SPECT imaging of tissue hypoxia using isotopically labeled 2-nitroimidazoles. Adv Exp Med Biol 510:285–292
Komar G, Seppanen M, Eskola O et al (2008) 18F-EF5: a new PET tracer for imaging hypoxia in head and neck cancer. J Nucl Med 49:1944–1951
Evans SM, Fraker D, Hahn SM et al (2006) EF5 binding and clinical outcome in human soft tissue sarcomas. Int J Radiat Oncol Biol Phys 64:922–927
Dolbier WR Jr, Li AR, Koch CJ, Shiue CY, Kachur AV (2001) [18F]-EF5, a marker for PET detection of hypoxia: synthesis of precursor and a new fluorination procedure. Appl Radiat Isot 54:73–80
Kumar P, Emami S, Kresolek Z et al (2009) Synthesis and hypoxia selective radiosensitization potential of beta-2-FAZA and beta-3-FAZL: fluorinated azomycin beta-nucleosides. Med Chem 5:118–129
Postema EJ, McEwan AJ, Riauka TA et al (2009) Initial results of hypoxia imaging using 1-alpha-D: -(5-deoxy-5-[18F]-fluoroarabinofuranosyl)-2-nitroimidazole (18F-FAZA). Eur J Nucl Med Mol Imaging 36:1565–1573
Riedl CC, Brader P, Zanzonico PB et al (2008) Imaging hypoxia in orthotopic rat liver tumors with iodine 124-labeled iodoazomycin galactopyranoside PET. Radiology 248:561–570
Riedl CC, Brader P, Zanzonico P et al (2008) Tumor hypoxia imaging in orthotopic liver tumors and peritoneal metastasis: a comparative study featuring dynamic 18F-MISO and 124I-IAZG PET in the same study cohort. Eur J Nucl Med Mol Imaging 35:39–46
Zanzonico P, O’Donoghue J, Chapman JD et al (2004) Iodine-124-labeled iodo-azomycin-galactoside imaging of tumor hypoxia in mice with serial microPET scanning. Eur J Nucl Med Mol Imaging 31:117–128
Fujibayashi Y, Taniuchi H, Yonekura Y et al (1997) Copper-62-ATSM: a new hypoxia imaging agent with high membrane permeability and low redox potential. J Nucl Med 38:1155–1160
Lewis JS, Herrero P, Sharp TL et al (2002) Delineation of hypoxia in canine myocardium using PET and copper(II)-diacetyl-bis(N(4)-methylthiosemicarbazone). J Nucl Med 43:1557–1569
O’Donoghue JA, Zanzonico P, Pugachev A et al (2005) Assessment of regional tumor hypoxia using 18F-fluoromisonidazole and 64Cu(II)-diacetyl-bis(N 4-methylthiosemicarbazone) positron emission tomography: comparative study featuring microPET imaging, Po2 probe measurement, autoradiography, and fluorescent microscopy in the R3327-AT and FaDu rat tumor models. Int J Radiat Oncol Biol Phys 61:1493–1502
Takahashi N, Fujibayashi Y, Yonekura Y et al (2000) Evaluation of 62Cu labeled diacetyl-bis(N 4-methylthiosemicarbazone) as a hypoxic tissue tracer in patients with lung cancer. Ann Nucl Med 14:323–328
Vavere AL, Lewis JS (2007) Cu-ATSM: a radiopharmaceutical for the PET imaging of hypoxia. Dalton Trans 4893–4902
Dehdashti F, Grigsby PW, Mintun MA et al (2003) Assessing tumor hypoxia in cervical cancer by positron emission tomography with 60Cu-ATSM: relationship to therapeutic response-a preliminary report. Int J Radiat Oncol Biol Phys 55:1233–1238
Lewis J, Laforest R, Buettner T et al (2001) Copper-64-diacetyl-bis(N 4-methylthiosemicarbazone): an agent for radiotherapy. Proc Natl Acad Sci USA 98:1206–1211
Obata A, Kasamatsu S, Lewis JS et al (2005) Basic characterization of 64Cu-ATSM as a radiotherapy agent. Nucl Med Biol 32:21–28
Lewis JS, Laforest R, Dehdashti F et al (2008) An imaging comparison of 64Cu-ATSM and 60Cu-ATSM in cancer of the uterine cervix. J Nucl Med 49:1177–1182
Urtasun RC, Parliament MB, McEwan AJ et al. Measurement of hypoxia in human tumours by non-invasive SPECT imaging of iodoazomycin arabinoside. Br J Cancer Suppl 27:S209–S212
Stypinski D, Wiebe LI, McEwan AJ et al (1999) Clinical pharmacokinetics of 123I-IAZA in healthy volunteers. Nucl Med Commun 20:559–567
Stypinski D, McQuarrie SA, Wiebe LI et al (2001) Dosimetry estimations for 123I-IAZA in healthy volunteers. J Nucl Med 42:1418–1423
Iyer RV, Kim E, Schneider RF, Chapman JD (1988) A dual hypoxic marker technique for measuring oxygenation change within individual tumors. Br J Cancer 78:163–169
Mees G, Dierckx R, Vangestel C, Van de Wiele C (2009) Molecular imaging of hypoxia with radiolabelled agents. Eur J Nucl Med Mol Imaging 36:1674–1686
Saitoh J, Sakurai H, Suzuki Y et al (2002) Correlations between in vivo tumor weight, oxygen pressure, 31P NMR spectroscopy, hypoxic microenvironment marking by beta-d-iodinated azomycin galactopyranoside (beta-d-IAZGP), and radiation sensitivity. Int J Radiat Oncol Biol Phys 54:903–909
Ballinger JR, Kee JW, Rauth AM (1996) In vitro and in vivo evaluation of a technetium-99m-labeled 2-nitroimidazole (BMS181321) as a marker of tumor hypoxia. J Nucl Med 37:1023–1031
Hoebers FJ, Janssen HL, Olmos AV et al (2002) Phase 1 study to identify tumour hypoxia in patients with head and neck cancer using technetium-99m BRU 59-21. Eur J Nucl Med Mol Imaging 29:1206–1211
Yutani K, Kusuoka H, Fukuchi K, Tatsumi M, Nishimura T (1999) Applicability of 99mTc-HL91, a putative hypoxic tracer, to detection of tumor hypoxia. J Nucl Med 40:854–861
Liu Z, Stevenson GD, Barrett HH et al. Imaging recognition of multidrug resistance in human breast tumors using 99mTc-labeled monocationic agents and a high-resolution stationary SPECT system. Nucl Med Biol 31:53–65
Bussink J, Kaanders JH, van der Kogel AJ (2003) Tumor hypoxia at the micro-regional level: clinical relevance and predictive value of exogenous and endogenous hypoxic cell markers. Radiother Oncol 67:3–15
Koukourakis MI, Bentzen SM, Giatromanolaki A et al (2006) Endogenous markers of two separate hypoxia response pathways (hypoxia inducible factor 2 alpha and carbonic anhydrase 9) are associated with radiotherapy failure in head and neck cancer patients recruited in the CHART randomized trial. J Clin Oncol 24:727–735
Koukourakis MI, Giatromanolaki A, Polychronidis A et al (2006) Endogenous markers of hypoxia/anaerobic metabolism and anemia in primary colorectal cancer. Cancer Sci 97:582–588
Le QT, Kong C, Lavori PW et al (2007) Expression and prognostic significance of a panel of tissue hypoxia markers in head-and-neck squamous cell carcinomas. Int J Radiat Oncol Biol Phys 69:167–175
Wang GL, Jiang BH, Semenza GL (1995) Effect of protein kinase and phosphatase inhibitors on expression of hypoxia-inducible factor 1. Biochem Biophys Res Commun 216:669–675
Semenza GL (2000) HIF-1: mediator of physiological and pathophysiological responses to hypoxia. J Appl Physiol 88:1474–1480
Semenza GL (2001) Hypoxia-inducible factor 1: control of oxygen homeostasis in health and disease. Pediatr Res 49:614–617
Maxwell P, Salnikow K (2004) HIF-1: an oxygen and metal responsive transcription factor. Cancer Biol Ther 3:29–35
Semenza GL (2000) Expression of hypoxia-inducible factor 1: mechanisms and consequences. Biochem Pharmacol 59:47–53
Zhong H, De Marzo AM, Laughner E et al (1999) Overexpression of hypoxia-inducible factor 1alpha in common human cancers and their metastases. Cancer Res 59:5830–5835
Moon EJ, Brizel DM, Chi JT, Dewhirst MW (2007) The potential role of intrinsic hypoxia markers as prognostic variables in cancer. Antioxid Redox Signal 9:1237–1294
Birner P, Schindl M, Obermair A et al (2000) Overexpression of hypoxia-inducible factor 1alpha is a marker for an unfavorable prognosis in early-stage invasive cervical cancer. Cancer Res 60:4693–4696
Shibata T, Giaccia AJ, Brown JM (2000) Development of a hypoxia-responsive vector for tumor-specific gene therapy. Gene Ther 7:493–498
Payen E, Bettan M, Henri A et al (2001) Oxygen tension and a pharmacological switch in the regulation of transgene expression for gene therapy. J Gene Med 3:498–504
Vordermark D, Shibata T, Brown JM (2001) Green fluorescent protein is a suitable reporter of tumor hypoxia despite an oxygen requirement for chromophore formation. Neoplasia 3:527–534
Harada H, Kizaka-Kondoh S, Hiraoka M (2005) Optical imaging of tumor hypoxia and evaluation of efficacy of a hypoxia-targeting drug in living animals. Mol Imaging 4:182–193
Harada H, Hiraoka M, Kizaka-Kondoh S (2002) Antitumor effect of TAT-oxygen-dependent degradation-caspase-3 fusion protein specifically stabilized and activated in hypoxic tumor cells. Cancer Res 62:2013–2018
Harada H, Kizaka-Kondoh S, Hiraoka M (2006) Mechanism of hypoxia-specific cytotoxicity of procaspase-3 fused with a VHL-mediated protein destruction motif of HIF-1alpha containing Pro564. FEBS Lett 580:5718–5722
Harada H, Kizaka-Kondoh S, Li G et al (2007) Significance of HIF-1-active cells in angiogenesis and radioresistance. Oncogene 26:7508–7516
Viola RJ, Provenzale JM, Li F et al (2008) In vivo bioluminescence imaging monitoring of hypoxia-inducible factor 1alpha, a promoter that protects cells, in response to chemotherapy. AJR Am J Roentgenol 191:1779–1784
Mayer A, Wree A, Hockel M et al (2004) Lack of correlation between expression of HIF-1alpha protein and oxygenation status in identical tissue areas of squamous cell carcinomas of the uterine cervix. Cancer Res 64:5876–5881
Lehmann S, Stiehl DP, Honer M et al (2009) Longitudinal and multimodal in vivo imaging of tumor hypoxia and its downstream molecular events. Proc Natl Acad Sci USA 106:14004–14009
Potter CP, Harris AL (2003) Diagnostic, prognostic and therapeutic implications of carbonic anhydrases in cancer. Br J Cancer 89:2–7
Ivanov S, Liao SY, Ivanova A et al (2001) Expression of hypoxia-inducible cell-surface transmembrane carbonic anhydrases in human cancer. Am J Pathol 158:905–919
Wykoff CC, Beasley NJ, Watson PH et al. Hypoxia-inducible expression of tumor-associated carbonic anhydrases. Cancer Res 60:7075–7083
Dubois L, Lieuwes NG, Maresca A et al (2009) Imaging of CA IX with fluorescent labelled sulfonamides distinguishes hypoxic and (re)-oxygenated cells in a xenograft tumour model. Radiother Oncol 92:423–428
van Dijk J, Uemura H, Beniers AJ et al. Therapeutic effects of monoclonal antibody G250, interferons and tumor necrosis factor, in mice with renal-cell carcinoma xenografts. Int J Cancer 56:262–268
Stillebroer AB, Oosterwijk E, Oyen WJ, Mulders PF, Boerman OC (2007) Radiolabeled antibodies in renal cell carcinoma. Cancer Imaging 7:179–188
Ahlskog JK, Schliemann C, Marlind J et al (2009) Human monoclonal antibodies targeting carbonic anhydrase IX for the molecular imaging of hypoxic regions in solid tumours. Br J Cancer 101:645–657
Hoogsteen IJ, Marres HA, Wijffels KI et al (2005) Colocalization of carbonic anhydrase 9 expression and cell proliferation in human head and neck squamous cell carcinoma. Clin Cancer Res 11:97–106
Kim SJ, Shin HJ, Jung KY et al (2007) Prognostic value of carbonic anhydrase IX and Ki-67 expression in squamous cell carcinoma of the tongue. Jpn J Clin Oncol 37:812–819
Mayer A, Hockel M, Vaupel P (2005) Carbonic anhydrase IX expression and tumor oxygenation status do not correlate at the microregional level in locally advanced cancers of the uterine cervix. Clin Cancer Res 11:7220–7225
Westra J, Molema G, Kallenberg CG (2010) Hypoxia-inducible factor-1 as regulator of angiogenesis in rheumatoid arthritis—therapeutic implications. Curr Med Chem 17:254–263
Macheda ML, Rogers S, Best JD (2005) Molecular and cellular regulation of glucose transporter (GLUT) proteins in cancer. J Cell Physiol 202:654–662
Rogers S, Macheda ML, Docherty SE et al (2002) Identification of a novel glucose transporter-like protein-GLUT-12. Am J Physiol Endocrinol Metab 282:E733–E738
Airley RE, Loncaster J, Raleigh JA et al (2003) GLUT-1 and CAIX as intrinsic markers of hypoxia in carcinoma of the cervix: relationship to pimonidazole binding. Int J Cancer 104:85–91
Jonathan RA, Wijffels KI, Peeters W et al (2006) The prognostic value of endogenous hypoxia-related markers for head and neck squamous cell carcinomas treated with ARCON. Radiother Oncol 79:288–297
Grosso AR, Martins S, Carmo-Fonseca M (2008) The emerging role of splicing factors in cancer. EMBO Rep 9:1087–1093
Martinkova J, Gadher SJ, Hajduch M, Kovarova H (2009) Challenges in cancer research and multifaceted approaches for cancer biomarker quest. FEBS Lett 583:1772–1784
Koong AC, Denko NC, Hudson KM et al (2000) Candidate genes for the hypoxic tumor phenotype. Cancer Res 60:883–887
Lal A, Peters H, St Croix B et al (2001) Transcriptional response to hypoxia in human tumors. J Natl Cancer Inst 93:1337–1343
Chen JL, Lucas JE, Schroeder T et al (2008) The genomic analysis of lactic acidosis and acidosis response in human cancers. PLoS Genet 4:e1000293
Starmans MH, Zips D, Wouters BG, Baumann M, Lambin P (2009) The use of a comprehensive tumour xenograft dataset to validate gene signatures relevant for radiation response. Radiother Oncol 92:417–422
Rho JH, Qin S, Wang JY, Roehrl MH (2008) Proteomic expression analysis of surgical human colorectal cancer tissues: up-regulation of PSB7, PRDX1, and SRP9 and hypoxic adaptation in cancer. J Proteome Res 7:2959–2972
Picchio M, Beck R, Haubner R et al (2008) Intratumoral spatial distribution of hypoxia and angiogenesis assessed by 18F-FAZA and 125I-Gluco-RGD autoradiography. J Nucl Med 49:597–605
Acknowledgment
This research was supported by the Intramural Research Program of the National Institute of Biomedical Imaging and Bioengineering (NIBIB), National Institutes of Health (NIH). G.N., who currently has an Imaging Sciences Training Fellowship, is jointly supported by the Radiology and Imaging Sciences Department, NIH Clinical Center, and the Intramural Research Program, NIBIB, NIH.
Conflict of interest disclosure
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sun, X., Niu, G., Chan, N. et al. Tumor Hypoxia Imaging. Mol Imaging Biol 13, 399–410 (2011). https://doi.org/10.1007/s11307-010-0420-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-010-0420-z