Abstract
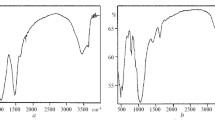
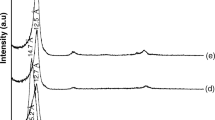
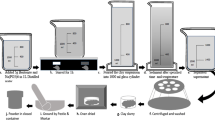
Nowadays, due to the most application of montmorillonite, its purification from raw bentonite has great importance. More than 76% of bentonite is composed of montmorillonite, and its industrial applications are related to its montmorillonite content. In this study, the nanoclay was extracted from bentonite by the use of centrifugal force. The results of the field-emission scanning electron microscopy (FE-SEM) analysis show that the nanosized of purified montmorillonite has a sheet structure with a spacing of 22.41 nm and 45.0 nm. The sharp peaks in X-ray diffraction analysis (XRD) illustrated that the montmorillonite purified successfully, and the results of Fourier-transform infrared spectrophotometry (FT-IR) revealed the successful incorporation of the metabolic extraction within the montmorillonite. By comparison of Brunauer–Emmett–Teller (BET) results with IUPAC, it can be realized that the synthesized montmorillonite nanoclay has a microporous structure (< 2 nm) with a surface area of 11.325 m2 g−1. According to IUPAC classification, the BET isotherms of montmorillonite and bentonite indicate a hysteresis loop belonging to the type H3. Finally, the economic analysis results revealed to this method could be the best option for achieving high purity montmorillonite for future applications.





Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Alhamami M, Doan H, Cheng CH (2014) A review on breathing behaviors of metal-organic-frameworks (MOFs) for gas adsorption. Materials 7:3198–3250. https://doi.org/10.3390/ma7043198
Awad AM, Shaikh SMR, Jalab R, Gulied MH, Nasser MS, Benamor A, Adham S (2019) Adsorption of organic pollutants by natural and modified clays: a comprehensive review. Sep Purif Technol 228:115719. https://doi.org/10.1016/j.seppur.2019.115719
Bananezhad B, Islami MR, Ghonchepour E, Mostafavi H, Tikdari AM, Rafiei HR (2019) Bentonite clay as an efficient substrate for the synthesis of the super stable and recoverable magnetic nanocomposite of palladium (Fe3O4/Bentonite-Pd). Polyhedron 162:192–200. https://doi.org/10.1016/j.poly.2019.01.054
Barakan S, Aghazadeh V (2019) Separation and characterization of montmorillonite from low-grade natural bentonite: using a non-destructive method. Micro &. Nano Lett 14:688–693. https://doi.org/10.1049/mnl.2018.5364
Calabi-Floody M, Bendall JS, Jara AA, Welland ME, Theng BKG, Rumpel C, Mora M d l L (2011) Nanoclays from an Andisol: extraction, properties, and carbon stabilization. Geoderma 161:159–167. https://doi.org/10.1016/j.geoderma.2010.12.013
Chihi R, Blidi I, Trabelsi-Ayadi M, Ayari F (2019) Elaboration and characterization of a low-cost porous ceramic support from natural Tunisian bentonite clay. Comptes Rendus Chimie, Second International Symposium Clays and Ceramics for Environmental Applications 22:188–197. https://doi.org/10.1016/j.crci.2018.12.002
Gong Z, Liao L, Lu G, Wand X (2020) A simple method for physical purification of bentonite. Appl Clay Sci 119:294–300. https://doi.org/10.1016/j.clay.2015.10.031
Hayati-Ashtiani M (2011) Characterization of nano-porous bentonite (montmorillonite) particles using FTIR and BET-BJH analyses. Part Part Syst Charact 28:71–76. https://doi.org/10.1002/ppsc.201100030
Inglethorpe SBJ, Morgan DJ, Highley DE, Bloodworth AJ (1993) Technical report WG/93/20. Mineralogy and petrology Series. British Geology Survey. 1–115
Issaabadi Z, Nasrollahzadeh M, Sajadi SM (2017) Green synthesis of the copper nanoparticles supported on bentonite and investigation of its catalytic activity. J Clean Prod 142:3584–3591. https://doi.org/10.1016/j.jclepro.2016.10.109
Keymirov MA (2018) Water purification of ions of heavy metals by montmorillonite modified with polyamine. J Water Chem Technol 40:320–326. https://doi.org/10.3103/S1063455X18060024
Khedr MH, Omar AA, Abdel-Moaty SA (2006) Reduction of carbon dioxide into carbon by freshly reduced CoFe2O4 nanoparticles. Mater Sci Eng A 432:26–33. https://doi.org/10.1016/j.msea.2006.06.012
Krupskaya V, Zakusin S, Tyupina E, Dorzhieva O, Zhukhlistov A, Belousov P, Timofeeva M (2017) Experimental study of montmorillonite structure and transformation of its properties under treatment with inorganic acid solutions. Minerals 7:49. https://doi.org/10.3390/min7040049
Leal CA, Brunet MNC, Amorim LV, Lira HL, Nogueira FCC, Costa WRP (2019) Influence of reactivity and mineralogical composition on instability due to the disintegration of shales from Paraíba and Ceará States, Brazil. Cerâmica 65:400–406. https://doi.org/10.1590/0366-69132019653752630
Li Z, Hu N (2003) Direct electrochemistry of heme proteins in their layer-by-layer films with clay nanoparticles. J Electroanal Chem 558:155–165. https://doi.org/10.1016/S0022-0728(03)00390-5
Liu XD, Lu XJ, Qiu J, Wang ZM, Wu P (2012) Purification of low-grade Ca-bentonite for iron ore pellets. AMR 454:237–241. https://doi.org/10.4028/scientific5/AMR.454.237
Modabberi S, Namayandeh S, López-Galindo A, Viseras C, Setti M, Ranjbaran M (2015) Characterization of Iranian bentonites to be used as pharmaceutical materials. Appl Clay Sci 116-117:193–201
Morais ICG, Silva IA, Buriti BMB, Fernandes JV, Silva DS, Neves GA, Ferreira HS (2020) Influence of the additivation process on cation exchange capacity and viscosity of bentonitic clay dispersions. Cerâmica 66:81–87. https://doi.org/10.1590/0366-69132020663772695
Nadziakiewicza M, Kehoe S, Micek P (2019) Physico-chemical properties of clay minerals and their use as a health promoting feed additive Animals (Basel) 9. https://doi.org/10.3390/ani9100714
Porebska R, Rybak A, Rapacz-Kmita A (2020) Montmorillonite–triclosan hybrid as effective antibacterial additive with enhanced thermal stability for protection of plastic electrical components. Polym Bull 77:17–31. https://doi.org/10.1007/s00289-019-02699-x
Qiao Z, Liu Z, Zhang S, Yang Y, Wu Y, Liu L, Liu Q (2020) Purification of montmorillonite and the influence of the purification method on textural properties. Appl Clay Sci 187:105491. https://doi.org/10.1016/j.clay.2020.105491
Saha UK, Liu, C, Kozak, LM, Huang, PM (2004) Kinetics of selenite adsorption on hydroxy aluminum- and hydroxy aluminosilicate-montmorillonite complexes. Soil Sci Soc Am J
Slavutsky AM, Bertuzzi MA, Armada M (2012) Water barrier properties of starch-clay nanocomposite films. Braz J Food Technol 15:208–218. https://doi.org/10.1590/S1981-67232012005000014
Thuc CNH, Grillet AC, Reinert L, Ohashi F, Thuc HH, Duclaux L (2010) Separation and purification of montmorillonite and polyethylene oxide modified montmorillonite from Vietnamese bentonites. Appl Clay Sci 49:229–238. https://doi.org/10.1016/j.clay.2010.05.011
Veiskarami M, Mansouri Esfahani M, Nasiri Sarvi M (2017) Qualification of the montmorillonite purification process for nanoclay production. Iran J Crystallogr Mineral 25:557–566. https://doi.org/10.18869/acadpub.ijcm.25.3.557
Wang J, Zhuang S (2017) Removal of various pollutants from water and wastewater by modified chitosan adsorbents. Crit Rev Environ Sci Technol 47:2331–2386. https://doi.org/10.1080/10643389.2017.1421845
Wu H, Xie H, He G, Guan Y, Zhang Y (2016) Effects of the pH and anions on the adsorption of tetracycline on iron-montmorillonite. Appl Clay Sci 119:161–169. https://doi.org/10.1016/j.clay.2015.08.001
Yadav VB, Gadi R, Kalra S (2018) Synthesis and characterization of novel nanocomposite using kaolinite and carbon nanotubes. Appl Clay Sci 155:30–36
Yadav VB, Gadi R, Kalra S (2019) Clay-based nanocomposites for removal of heavy metals from water: a review. J Environ Manag 232:803–817. https://doi.org/10.1016/j.jenvman.2018.11.120
Yoon JW, Seo YK, Hwang YK, Chang JS, Leclerc H, Wuttke S, Bazin P, Vimont A, Daturi M, Bloch E, Llewellyn PL, Serre C, Horcajada P, Grenèche JM, Rodrigues AE, Férey G (2010) Controlled reducibility of a metal-organic framework with coordinatively unsaturated sites for preferential gas sorption. Angew Chem Int Ed 49:5949–5952. https://doi.org/10.1002/anie.201001230
Funding
Financial support and sponsorship from the Foundation for Research Support of the State of Rio Grande do Sul (FAPERGS), National Council for Scientific and Technological Development (CNPq, Brazil), and Coordination of Improvement of Higher Education Personnel (CAPES, Brazil) are acknowledged.
Author information
Authors and Affiliations
Contributions
Zahra Biglari Quchan Atigh: Conducted experimental verification, analyzed the data, and wrote the manuscript
Ava Heidari: Contributed to the research idea and designed the study
Alireza Karimi: Contributed to the conception of the study
Mohammad Ali Pezhman: Helped the cost analysis through constructive discussions
Behnam Asgari Lajayer: Prepared, edited, and submitted the manuscript
Eder Claudio Lima: Helped perform the analysis with constructive discussions and revised the article
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Biglari Quchan Atigh, Z., Heidari, A., Karimi, A. et al. Purification and economic analysis of nanoclay from bentonite. Environ Sci Pollut Res 28, 13690–13696 (2021). https://doi.org/10.1007/s11356-020-11595-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-11595-1