Abstract
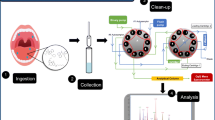
A sensitive method was developed for quantifying a wide range of cannabinoids in oral fluid (OF) by liquid chromatography–tandem mass spectrometry (LC–MS/MS). These cannabinoids include ∆9-tetrahydrocannabinol (THC), 11-hydroxy-∆9-tetrahydrocannabinol (11-OH-THC), 11-nor-9-carboxy-∆9-tetrahydrocannabinol (THCCOOH), cannabinol (CBN), cannabidiol (CBD), ∆9-tetrahydrocannabinolic acid A (THC-A), 11-nor-9-carboxy-∆9-tetrahydrocannabinol glucuronide (THCCOOH-gluc), and ∆9-tetrahydrocannabinol glucuronide (THC-gluc). Samples were collected using a Quantisal™ device. The advantages of performing a liquid–liquid extraction (LLE) of KCl-saturated OF using heptane/ethyl acetate versus a solid-phase extraction (SPE) using HLB copolymer columns were determined. Chromatographic separation was achieved in 11.5 min on a Kinetex™ column packed with 2.6-μm core–shell particles. Both positive (THC, 11-OH-THC, CBN, and CBD) and negative (THCCOOH, THC-gluc, THCCOOH-gluc, and THC-A) electrospray ionization modes were employed with multiple reaction monitoring using a high-end AB Sciex API 5000™ triple quadrupole LC–MS/MS system. Unlike SPE, LLE failed to extract THC-gluc and THCCOOH-gluc. However, the LLE method was more sensitive for the detection of THCCOOH than the SPE method, wherein the limit of detection (LOD) and limit of quantification (LOQ) decreased from 100 to 50 pg/ml and from 500 to 80 pg/ml, respectively. The two extraction methods were successfully applied to OF samples collected from volunteers before and after they smoked a homemade cannabis joint. High levels of THC were measured soon after smoking, in addition to significant amounts of THC-A. Other cannabinoids were found in low concentrations. Glucuronide conjugate levels were lower than the method’s LOD for most samples. Incubation studies suggest that glucuronides could be enzymatically degraded by glucuronidase prior to OF collection.

Similar content being viewed by others
References
Kelly E, Darke S, Ross J (2004) A review of drug use and driving: epidemiology, impairment, risk factors and risk perceptions. Drug Alcohol Rev 23(3):319–344. doi:10.1080/09595230412331289482
Choo RE, Huestis MA (2004) Oral fluid as a diagnostic tool. Clin Chem Lab Med 42(11):1273–1287. doi:10.1515/CCLM.2004.248
Molnar A, Lewis J, Doble P, Hansen G, Prolov T, Fu S (2011) A rapid and sensitive method for the identification of delta-9-tetrahydrocannabinol in oral fluid by liquid chromatography–tandem mass spectrometry. Forensic Sci Int. doi:10.1016/j.forsciint.2011.01.045
Moore C, Rana S, Coulter C, Day D, Vincent M, Soares J (2007) Detection of conjugated 11-nor-Delta9-tetrahydrocannabinol-9-carboxylic acid in oral fluid. J Anal Toxicol 31(4):187–194
Lee D, Milman G, Barnes AJ, Goodwin RS, Hirvonen J, Huestis MA (2011) Oral fluid cannabinoids in chronic, daily Cannabis smokers during sustained, monitored abstinence. Clin Chem 57(8):1127–1136. doi:10.1373/clinchem.2011.164822
Milman G, Barnes AJ, Schwope DM, Schwilke EW, Darwin WD, Goodwin RS, Kelly DL, Gorelick DA, Huestis MA (2010) Disposition of cannabinoids in oral fluid after controlled around-the-clock oral THC administration. Clin Chem 56(8):1261–1269. doi:10.1373/clinchem.2009.141853
Milman G, Schwope DM, Schwilke EW, Darwin WD, Kelly DL, Goodwin RS, Gorelick DA, Huestis MA (2011) Oral fluid and plasma cannabinoid ratios after around-the-clock controlled oral Delta(9)-tetrahydrocannabinol administration. Clin Chem 57(11):1597–1606. doi:10.1373/clinchem.2011.169490
Moore C, Rana S, Coulter C (2007) Simultaneous identification of 2-carboxy-tetrahydrocannabinol, tetrahydrocannabinol, cannabinol and cannabidiol in oral fluid. J Chromatogr B Analyt Technol Biomed Life Sci 852(1–2):459–464. doi:10.1016/j.jchromb.2007.02.016
Coles R, Clements TT, Nelson GJ, McMillin GA, Urry FM (2005) Simultaneous analysis of the Delta9-THC metabolites 11-nor-9-carboxy-Delta9-THC and 11-hydroxy-Delta9-THC in meconium by GC–MS. J Anal Toxicol 29(6):522–527
ElSohly MA, Feng S (1998) Delta 9-THC metabolites in meconium: identification of 11-OH-delta 9-THC, 8 beta, 11-diOH-delta 9-THC, and 11-nor-delta 9-THC-9-COOH as major metabolites of delta 9-THC. J Anal Toxicol 22(4):329–335
Fabritius M, Staub C, Mangin P, Giroud C (2012) Distribution of free and conjugated cannabinoids in human bile samples. Forensic Sci Int. http://dx.doi.org/10.1016/j.forsciint.2012.08.013
Feng S, ElSohly MA, Salamone S, Salem MY (2000) Simultaneous analysis of delta9-THC and its major metabolites in urine, plasma, and meconium by GC–MS using an immunoaffinity extraction procedure. J Anal Toxicol 24(6):395–402
Gronewold A, Skopp G (2011) A preliminary investigation on the distribution of cannabinoids in man. Forensic Sci Int 210(1–3):e7–e11. doi:10.1016/j.forsciint.2011.04.010
Lin DL, Lin RL (2005) Distribution of 11-nor-9-carboxy-Delta9-tetrahydrocannabinol in traffic fatality cases. J Anal Toxicol 29(1):58–61
Marchei E, Pellegrini M, Pacifici R, Palmi I, Lozano J, Garcia-Algar O, Pichini S (2006) Quantification of Delta9-tetrahydrocannabinol and its major metabolites in meconium by gas chromatographic-mass spectrometric assay: assay validation and preliminary results of the “meconium project”. Ther Drug Monit 28(5):700–706. doi:10.1097/01.ftd.0000245380.95186.13
Moore C, Lewis D, Becker J, Leikin J (1996) The determination of 11-nor-delta 9-tetrahydrocannabinol-9-carboxylic acid (THCCOOH) in meconium. J Anal Toxicol 20(1):50–51
Papoutsis I, Nikolaou P, Dona A, Pistos C, Stefanidou M, Spiliopoulou C, Athanaselis S (2012) A validated GC–MS method for the determination of ∆9-tetrahydrocannabinol and 11-nor-∆9-tetrahydrocannabinol-9-carboxylic acid in bile samples. Forensic Toxicol 30(1):51–58. doi:10.1007/s11419-011-0126-1
Badawi N, Simonsen KW, Steentoft A, Bernhoft IM, Linnet K (2009) Simultaneous screening and quantification of 29 drugs of abuse in oral fluid by solid-phase extraction and ultraperformance LC–MS/MS. Clin Chem 55 (11):2004–2018. doi:10.1373/clinchem.2008.122341
Chi E, Cole J (2010) Detecting Marijuana in Saliva. Forensic Mag 7(5):17–20
Concheiro M, de Castro A, Quintela O, Cruz A, Lopez-Rivadulla M (2008) Determination of illicit and medicinal drugs and their metabolites in oral fluid and preserved oral fluid by liquid chromatography–tandem mass spectrometry. Anal Bioanal Chem 391(6):2329–2338. doi:10.1007/s00216-008-2135-4
Day D, Kuntz DJ, Feldman M, Presley L (2006) Detection of THCA in oral fluid by GC–MS–MS. J Anal Toxicol 30(9):645–650
Laloup M, Ramirez Fernandez Mdel M, Wood M, De Boeck G, Henquet C, Maes V, Samyn N (2005) Quantitative analysis of delta9-tetrahydrocannabinol in preserved oral fluid by liquid chromatography–tandem mass spectrometry. J Chromatogr A 1082(1):15–24
Niedbala RS, Kardos KW, Fritch DF, Kardos S, Fries T, Waga J, Robb J, Cone EJ (2001) Detection of marijuana use by oral fluid and urine analysis following single-dose administration of smoked and oral marijuana. J Anal Toxicol 25(5):289–303
Oiestad EL, Johansen U, Christophersen AS (2007) Drug screening of preserved oral fluid by liquid chromatography–tandem mass spectrometry. Clin Chem 53(2):300–309. doi:10.1373/clinchem.2006.074237
Quintela O, Andrenyak DM, Hoggan AM, Crouch DJ (2007) A validated method for the detection of Delta 9-tetrahydrocannabinol and 11-nor-9-carboxy- Delta 9-tetrahydrocannabinol in oral fluid samples by liquid chromatography coupled with quadrupole-time-of-flight mass spectrometry. J Anal Toxicol 31(3):157–164
Sergi M, Bafile E, Compagnone D, Curini R, D’Ascenzo G, Romolo FS (2009) Multiclass analysis of illicit drugs in plasma and oral fluids by LC–MS/MS. Anal Bioanal Chem 393(2):709–718. doi:10.1007/s00216-008-2456-3
Simoes SS, Ajenjo AC, Franco JM, Vieira DN, Dias MJ (2009) Liquid chromatography/tandem mass spectrometry for the qualitative and quantitative analysis of illicit drugs and medicines in preserved oral fluid. Rapid Commun Mass Spectrom 23(10):1451–1460. doi:10.1002/rcm.4020
Simonin J, Salquebre G, Cirimele V, Kintz P (2007) Screening for illicit drugs in oral fluid by LC–MS/MS. Ann Toxicol Anal 19(2):141–150
Gritti F, Guiochon G (2010) Performance of columns packed with the new shell Kinetex-C18 particles in gradient elution chromatography. J Chromatogr A 1217(10):1604–1615. doi:10.1016/j.chroma.2010.01.008
Gritti F, Leonardis I, Shock D, Stevenson P, Shalliker A, Guiochon G (2010) Performance of columns packed with the new shell particles, Kinetex-C18. J Chromatogr A 1217(10):1589–1603. doi:10.1016/j.chroma.2009.12.079
Moore C, Vincent M, Rana S, Coulter C, Agrawal A, Soares J (2006) Stability of Delta(9)-tetrahydrocannabinol (THC) in oral fluid using the Quantisal collection device. Forensic Sci Int 164(2–3):126–130. doi:10.1016/j.forsciint.2005.12.011
Hubert P, Nguyen-Huu JJ, Boulanger B, Chapuzet E, Chiap P, Cohen N, Compagnon PA, Dewe W, Feinberg M, Lallier M, Laurentie M, Mercier N, Muzard G, Nivet C, Valat L, Rozet E (2007) Harmonization of strategies for the validation of quantitative analytical procedures. A SFSTP proposal—part II. J Pharm Biomed Anal 45(1):70–81. doi:10.1016/j.jpba.2007.06.013
Hubert P, Nguyen-Huu JJ, Boulanger B, Chapuzet E, Cohen N, Compagnon PA, Dewe W, Feinberg M, Laurentie M, Mercier N, Muzard G, Valat L, Rozet E (2007) Harmonization of strategies for the validation of quantitative analytical procedures. A SFSTP proposal—part III. J Pharm Biomed Anal 45(1):82–96. doi:10.1016/j.jpba.2007.06.032
Matuszewski BK, Constanzer ML, Chavez-Eng CM (2003) Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC–MS/MS. Anal Chem 75(13):3019–3030
Chauncey HH, Lionetti F, Winer RA, Lisanti VF (1954) Enzymes of human saliva. I. The determination, distribution, and origin of whole saliva enzymes. J Dent Res 33(3):321–334
Ghadge MS, Raste AS (2006) Place of salivary beta glucuronidase activity in head and neck cancers. Indian J Clin Biochem 21(1):196–198
Lamster IB, Kaufman E, Grbic JT, Winston LJ, Singer RE (2003) Beta-glucuronidase activity in saliva: relationship to clinical periodontal parameters. J Periodontol 74(3):353–359. doi:10.1902/jop.2003.74.3.353
Pil K, Verstraete A (2008) Current developments in drug testing in oral fluid. Ther Drug Monit 30(2):196–202. doi:10.1097/FTD.0b013e318167d563
Hazekamp A, Peltenburg A, Verpoorte R, Giroud C (2005) Chromatographic and spectroscopic data of cannabinoids from Cannabis sativa L. J Liq Chromatogr Relat Technol 28(15):2361–2382
Poklis JL, Thompson CC, Long KA, Lichtman AH, Poklis A (2010) Disposition of cannabichromene, cannabidiol, and Delta(9)-tetrahydrocannabinol and its metabolites in mouse brain following marijuana inhalation determined by high-performance liquid chromatography–tandem mass spectrometry. J Anal Toxicol 34(8):516–520
Barni-Comparini I, Ferri S, Centini F (1984) Cannabinoid level in the leaves as a tool for the early discrimination of cannabis chemiovariants. Forensic Sci Int 24(1):37–42
Skopp G, Potsch L (2002) Stability of 11-nor-delta(9)-carboxy-tetrahydrocannabinol glucuronide in plasma and urine assessed by liquid chromatography–tandem mass spectrometry. Clin Chem 48(2):301–306
Humphrey SP, Williamson RT (2001) A review of saliva: normal composition, flow, and function. J Prosthet Dent 85(2):162–169. doi:10.1067/mpr.2001.113778
Skopp G, Potsch L, Mauden M, Richter B (2002) Partition coefficient, blood to plasma ratio, protein binding and short-term stability of 11-nor-Delta(9)-carboxy tetrahydrocannabinol glucuronide. Forensic Sci Int 126(1):17–23
Cone EJ, Huestis MA (2007) Interpretation of oral fluid tests for drugs of abuse. Ann N Y Acad Sci 1098:51–103. doi:10.1196/annals.1384.037
Smink BE, Hofman BJ, Dijkhuizen A, Lusthof KJ, de Gier JJ, Egberts AC, Uges DR (2008) The concentration of oxazepam and oxazepam glucuronide in oral fluid, blood and serum after controlled administration of 15 and 30 mg oxazepam. Br J Clin Pharmacol 66(4):556–560. doi:10.1111/j.1365-2125.2008.03252.x
Watanabe K, Oguri K, Yoshimura H (1979) Synthesis of delta 8-tetrahydrocannabinol glucuronide and sulfate, and their metabolic disposition in rats. Chem Pharm Bull 27(12):3009–3014
Scheidweiler KB, Desrosiers NA, Huestis MA (2012) Simultaneous quantification of free and glucuronidated cannabinoids in human urine by liquid chromatography tandem mass spectrometry. Clin Chim Acta Int J Clin Chem 413(23–24):1839–1847. doi:10.1016/j.cca.2012.06.034
Wang RE, Tian L, Chang YH (2012) A homogeneous fluorescent sensor for human serum albumin. J Pharm Biomed Anal 63:165–169. doi:10.1016/j.jpba.2011.12.035
Fanali G, Cao Y, Ascenzi P, Trezza V, Rubino T, Parolaro D, Fasano M (2011) Binding of delta9-tetrahydrocannabinol and diazepam to human serum albumin. IUBMB Life 63(6):446–451. doi:10.1002/iub.466
Acknowledgments
The authors thank the Swiss National Science Foundation (FNS_320030_127507/1) and the Faculty of Biology and Medicine (interdisciplinary grant) at the University of Lausanne for their financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fabritius, M., Staub, C., Mangin, P. et al. Analysis of cannabinoids in oral fluid by liquid chromatography–tandem mass spectrometry. Forensic Toxicol 31, 151–163 (2013). https://doi.org/10.1007/s11419-012-0168-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11419-012-0168-z