Abstract
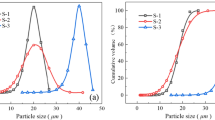
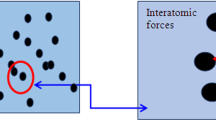
In this work, the effects of nanoparticle size, particle volume fraction and pH on the viscosity of silicon dioxide nanocolloidal dispersions are investigated. Both size and pH are found to significantly affect nanocolloid viscosity. Two models are used to study the effect of aggregate structure on the viscosity of the nanocolloidal dispersion. The fractal concept is introduced to describe the irregular and dynamic aggregate structure. The structure of aggregates, which is considered to play an important role in viscosity, is affected by both intermolecular and electrostatic forces. The particle interaction is primarily affected by particle distance and becomes stronger with decreasing particle size and increasing volume fraction. The aggregate structure is also affected by the pH of the solution. Studying the relationship between pH and zeta-potential shows that with the neutralization of charges on the particle surface and decreasing electrical repulsion force, the particle interaction becomes dominated by attractive forces and the aggregates form a more compact structure.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Eastman J A, Choi S U S, Li S, et al. Anomalously increased effective thermal conductivities of ethylene glycol-based nanofluids containing copper nanoparticles. Appl Phys Lett, 2001, 78: 718–720
Marquis F D S, Chibante L P F. Improving the heat transfer of nanofluids and nanolubricants with carbon nanotubes. JOM, 2005, 57: 32–44
Jin S, Ye K. Nanoparticle-mediated drug delivery and gene therapy. Biotechnol Prog, 2007, 23: 32–41
Yamanaka J, Ise N, Miyoshi H, et al. Experimental examination of the Booth theory on the first-order electroviscous effect in ionic colloidal dispersions. Phys Rev E, 1995, 51: 1276–1281
Wang X, Xu X, Choi S U S. Thermal conductivity of nanoparticle-fluid mixture. J Thermophys Heat Trans, 1999, 13: 474–480
Rubio-Hernandez F J, Ayucar-Rubio M F, Velazquez-Navarro J F, et al. Intrinsic viscosity of SiO2, Al2O3 and TiO2 aqueous suspensions. J Colloid Interface Sci, 2006, 298: 967–972
Putra N, Roetzel W, Das S K. Natural convection of nano-fluids. Heat Mass Trans, 2003, 39: 775–784
Yu W, Xie H, Chen L, et al. Investigation of thermal conductivity and viscosity of ethylene glycol based ZnO nanofluid. Thermochimica Acta, 2009, 491: 92–96
Lee J, Hwang K, Janga S P, et al. Effective viscosities and thermal conductivities of aqueous nanofluids containing low volume concentrations of Al2O3 nanoparticles. Int J Heat Mass Transfer, 2008, 51: 2651–2656
Prasher R, Song D, Wang J, et al. Measurements of nanofluid viscosity and its implications for thermal applications. Appl Phys Lett, 2006, 89: 133108
Prasher R, Phelan P E, Bhattacharya P. Effect of aggregation kinetics on the thermal conductivity of nanoscale colloidal solutions (nanofluid). Nano Lett, 2006, 6: 1529–1534
Rooij R D, Potanin A A, Ende D, et al. Steady shear viscosity of weakly aggregating polystyrene latex dispersions. J Chem Phys, 1993, 99: 9213–9223
Jiang W, Ding G, Peng H, et al. Modeling of nanoparticles’ aggregation and sedimentation in nanofluid. Curr Appl Phys, 2010, 10: 934–941
Wang B, Zhou L, Peng X. A fractal model for predicting the effective thermal conductivity of liquid with suspension of nanoparticles. Int J Heat Mass Transfer, 2003, 46: 2665–2672
Waite T D, Cleaver J K, Beattie J K. Aggregation kinetics and fractal structure of γ-Alumina assemblages. J Colloid Interface Sci, 2001, 241: 333–339
Simha R. The influence of brownian movement on the viscosity of solutions. J Phys Chem, 1940, 44: 25–34
Sun Y, Li X, Duzgunesv N, et al. The shape parameter of liposomes and DNA-lipid complexes determined by viscometry utilizing small sample volumes. Biophys J, 2003, 85: 1223–1232
Berber S, Kwon Y, Tomanek D. Unusually high thermal conductivity of carbon nanotubes. Phys Rev Lett, 2000, 84: 4613–4616
Krieger I M. Rheology of monodisperse lattices. Adv Colloid Interface Sci, 1972, 3: 111–136
Einstein A. Eine neue Bestimmung der Molekiil-dimensionen. Ann Phys, 1911, 34: 591–592
Kok C M, Rudin A. Relationship between the hydrodynamic radius and the radius of gyration of a polymer in solution. Makromol Chem Rapid Commun, 1981, 2: 655–659
Keblinski P, Phillpot S R, Choi S U S, et al. Mechanisms of heat flow in suspensions of nano-sized particles (nanofluids). Int J Heat Mass Transfer, 2002, 45: 855–863
Chen C, Huang C, Tseng W, et al. Dispersion and rheology of surfactant-mediated silver nanoparticle suspensions. Appl Surf Sci, 2010, 257: 650–655
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Wang, T., Ni, M., Luo, Z. et al. Viscosity and aggregation structure of nanocolloidal dispersions. Chin. Sci. Bull. 57, 3644–3651 (2012). https://doi.org/10.1007/s11434-012-5150-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5150-y