Abstract
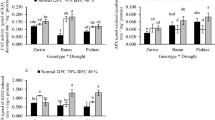
Drought is a major constraint limiting agricultural productivity worldwide. Exposure to drought results in altered physiology of plants and causes oxidative stress. The aim of this study was to evaluate genotypic differences in oxidative stress markers and transcript levels of genes encoding ascorbate peroxidase (APX) and monodehydroascorbate reductase (MDAR) from two finger millet (Eleusine coracana L.) varieties differing in their stress tolerance potential. Seedlings of PR202, a stress-tolerant variety, and PES400, a susceptible variety, were subjected to different polyethylene glycol 6000 (PEG-6000) concentrations (0, 5, 10, 15, and 20%). Increases in activity of APX and MDAR were observed under stress, with maximal increases being recorded at 15 and 20% PEG-6000. At these PEG-6000 levels, PES400 showed comparatively higher accumulations of malondialdehyde and hydrogen peroxide and a lower APX:MDAR ratio than PR202. Adaptive stress response was better in PR202 than in PES400, demonstrating the stress-tolerant character of PR202. Under oxidative stress, the expression of genes encoding APX and MDAR was positively correlated with the respective enzyme activities. The results demonstrated that 15 and 20% PEG-6000 concentrations are optimum for revealing differences in stress response between PR202 and PES400. The results of this study will be useful in understanding the expression of drought-responsive genes in E. coracana.






Similar content being viewed by others
References
Alexieva V, Sergiev I, Mapelli S, Karanov E (2001) The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ 24:1337–1344
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Amador BM, Aguilar RL, Kaya C, Mayoral JL, Hernández AF (2002) Comparative effects of NaCl and polyethylene glycol on germination, emergence and seedling growth of cowpea. J Agron Crop Sci 188:235–247
Asada K, Takahashi M (1987) Production and scavenging of active oxygen in photosynthesis. In: Kyle DJ, Osmond CB, Arntzen CJ (eds) Photoinhibition. Elsevier, Amsterdam, pp 228–287
Bartwal A, Mall R, Lohani P, Guru SK, Arora S (2012) Role of secondary metabolites and brassinosteroids in plant defense against environmental stresses. J Plant Growth Regul 32:216–232
Bartwal A, Pande A, Sharma P, Arora S (2016) Inter-varietal variations in various oxidative stress markers and antioxidant potential of finger millet (Eleusine coracana) subjected to drought stress. J Environ Biol 37(4):517–522
Bhatt D, Negi M, Sharma P, Saxena SC, Dobriyal AK, Arora S (2011) Responses to drought induced oxidative stress in five finger millet varieties differing in their geographical distribution. Physiol Mol Bio Plants 17:347–353
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:246–254
Fridovich I (1998) Oxygen toxicity: a radical explanation. J Exp Bot 201:1203–1209
Grzesiak MT, Grzesiak S, Skoczowski A (2006) Changes of leaf water potential and gas exchange parameters during and after drought in triticale and maize genotypes differing in drought tolerance. Photosynthetica 44:561–568
Halliwell B, Gutteridge JMC (1999) Free radicals in biology and medicine. Oxford University Press, New York
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Horemans N, Hees MV, Hoeck AV, Saenen E, Meutter TD, Nauts R, Blust R, Vandenhove H (2015) Uranium and cadmium provoke different oxidative stress responses in Lemna minor L. Plant Biol 17:91–100
Hossain MA, Asada K (1985) Monodehydroascorbate reductase from cucumber is a flavin adenine dinucleotide enzyme. J Biol Chem 260:12920–12926
Ji H, Liu L, Li K, Xie Q, Wang Z, Zhao X, Li X (2014) PEG-mediated osmotic stress induces premature differentiation of the root apical meristem and outgrowth of lateral roots in wheat. J Exp Bot. doi:10.1093/jxb/eru255
Joshi PK, Saxena SC, Arora S (2011) Characterization of Brassica juncea antioxidant potential under salinity stress. Acta Physiol Plant 33:811–822
Karpinski S, Muhlenbock P (2007) Genetic, molecular and physiological mechanisms controlling cell death, defences, and antioxidant network in response to abiotic and biotic stresses in plants. Comp Biochem Physiol A Mol Int Physiol 146:60–66
Lagerwerff JV, Ogata G, Eagle HE (1961) Control of osmotic pressure of culture solutions with polyethylene glycol. Science 133:1486–1487
Lai Q, Bao Z, Zhu Z, Qian Q, Mao B (2007) Effects of osmotic stress on antioxidant enzymes activities in leaf discs of PSAG12-IPT modified gerbera. J Zhejiang Univ Sci B 8:458–464
Lawlor DW (1970) Absorption of polyethylene glycols by plants and their effects on plant growth. New Phytol 69:501–514
Liu YJ, Yuan Y, Liu YY, Liu Y, Fu JJ, Zheng J, Wang GY (2012) Gene families of maize glutathione–ascorbate redox cycle respond differently to abiotic stresses. J Plant Physiol 169:183–192
Liu ZJ, Zhang XL, Bai JG, Suo BX, Xu PL, Wang L (2009) Exogenous paraquat changes antioxidant enzyme activities and lipid peroxidation in drought-stressed cucumber leaves. Sci Hortic 121:138–143
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444:139–158
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Noctor G, Mhamdi A, Foyer CH (2016) Oxidative stress and antioxidative systems: recipes for successful data collection and interpretation. Plant Cell Environ 39:1140–1160
Quan LJ, Zhang B, Shi WW, Li HY (2008) Hydrogen peroxide in plants: a versatile molecule of the reactive oxygen species network. J Integ Plant Biol 50:2–18
Rosales MA, Ruiz JM, Hernández J, Soriano T, Castilla N, Romero L (2006) Antioxidant content and ascorbate metabolism in cherry tomato exocarp in relation to temperature and solar radiation. J Sci Food Agric 86:1545–1551
Secenji M, Bebes A, Hideg E, Györgyey J (2008) Transcriptional changes in ascorbate-glutathione cycle under drought conditions. Acta Biol Szeged 52:93–94
Shehab GG, Ahmed OK, El-Beltagi HS (2010) Effects of various chemical agents for alleviation of drought stress in rice plants (Oryza sativa L.). Not Bot Horti Agrobot Cluj Napoca 38:139–148
Türkan İ, Bor M, Özdemir F, Koca H (2005) Differential responses of lipid peroxidation and antioxidants in the leaves of drought-tolerant P. acutifolius Gray and drought-sensitive P. vulgaris L. subjected to polyethylene glycol mediated water stress. Plant Sci 168:223–231
Uma S, Prasad TG, Kumar MU (1995) Genetic variability in recovery growth and synthesis of stress proteins in response to polyethylene glycol and salt stress in finger millet. Ann Bot 76:43–49
Winter SR, Musick JT, Porter KB (1988) Evaluation of screening techniques for breeding drought-resistant winter wheat. Crop Sci 28:512–516
Yoon HS, Lee H, Lee IA, Kim KY, Jo J (2004) Molecular cloning of the monodehydroascorbate reductase gene from Brassica campestris and analysis of its mRNA level in response to oxidative stress. Biochim Biophys Acta 1658:181–186
Acknowledgment
The authors are thankful to the Department of Biotechnology, Government of India, for the financial support under the Program Mode Support in Agricultural Biotechnology (Phase II) scheme.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Rakhi Chaturvedi
Rights and permissions
About this article
Cite this article
Bartwal, A., Arora, S. Drought stress-induced enzyme activity and mdar and apx gene expression in tolerant and susceptible genotypes of Eleusine coracana (L.). In Vitro Cell.Dev.Biol.-Plant 53, 41–49 (2017). https://doi.org/10.1007/s11627-016-9787-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-016-9787-0