Abstract
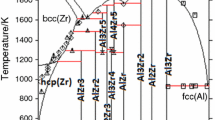
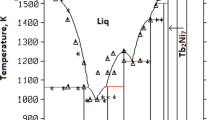
The present study reinvestigates the Al-Ce and Al-Nd phase diagrams and reoptimizes their thermodynamics using the CALPHAD method. First-principles energy calculations play an important role in terms of sublattice formalism and phase-stability prediction, demonstrating that they should be effectively integrated into experimental investigations and thermodynamic assessments. Specifically, current experimental results and theoretical calculations show that Al2Nd (or Al2Ce) should be treated as a stoichiometric compound phase rather than as the solution phase that was proposed in previous studies. Further, a new compound, AlCe2, is found stable at high temperatures (648 °C to 775 °C) in the Al-Ce system. It forms through a peritectic reaction of liquid and AlCe phases at 775 °C, and decomposes into AlCe and βAlCe3 at 648 °C and below. Since the AlCe2 phase is not retained at room temperature by quenching experiments, it is suggested that AlCe2 may be isostructural with the previously known compound AlNd2 (oP12). Based on current differential thermal analysis (DTA) measurements and theoretical calculations, it is also proposed that there is an α/βAl3Ce polymorphous transition occurring at 973 °C in the Al-Ce system and an α/βAl3Nd polymorphous transition occurring at 888 °C in the Al-Nd system. The βAl3RE phase may be isostructural with βAl3Y (hP12). Finally, the previously described βAl11RE3 phase (rare earth elements (RE)=La, Ce, Nd, or Pr) is proposed to have a stoichiometry of Al4RE (tI10), based on direct evidence from differential scanning calorimetry (DSC) measurements.
Similar content being viewed by others
References
Y. He, S.J. Poon, and G.J. Shiflet: Science, 1988, vol. 241, pp. 1640–42.
A. Inoue, K. Ohtera, and T. Masumoto: Jpn. J. Appl. Phys., 1988, vol. 27, pp. L736–39.
W. Biltz and H. Pieper: Z. Anorg. Chem., 1924, vol. 134, p. 13.
G. Canneri and A. Rossi: Gazz. Chim. Ital., 1932, vol. 62, pp. 202–11.
J.H.N. van Vucht: Z. Metallkd., 1957, vol. 48, p. 253.
K.H.J. Buschow and J.H.N. van Vucht: Philips Res. Rep., 1965, vol. 20, pp. 337–48.
K.H.J. Buschow: J. Less-Common Met., 1965, vol. 9, pp. 452–56.
K.H.J. Buschow and J.H.N. van Vucht: Z. Metallkd., 1966, vol. 57, pp. 162–66.
L.F. Yamshchikov, V.A. Lebedev, I.F. Nichkov, S.P. Raspopin, and O.K. Kokoulin: Izv. Vyssh Uchebn. Zaved., Tsv. Metall., 1980, vol. 5, pp. 50–54.
V.I. Kononenko and S.V. Golubev: Izv. Akad Nauk SSSR, Met., 1990, vol. 2, pp. 197–99.
N. Clavaguera and Y. Du: J. Phase Equilibrium, 1996, vol. 17, pp. 107–11.
J. Wang: CALPHAD, 1996, vol. 20, pp. 135–38.
G.B. Kale, A. Biswas, and I.G. Sharma: Scripta Mater., 1997, vol. 37, pp. 999–1003.
A.H. Gowes De Mesquita and K.H.J. Buschow: Acta Crystallogr., 1967, vol. 22, pp. 497–501.
M. Baricco, F. Gaetner, G. Cacciamani, P. Rizzi, L. Battezzati, and A.L. Greer: Mater. Sci. Forum, 1998, vols. 269–272, pp. 553–58.
L. Battezzati, M. Barcicco, and C. Antonione: J. Alloys Compounds, 1994, vol. 209, pp. 341–49.
A. Saccone, A.M. Cardinale, S. Delfino, and R. Ferro: Z. Metallkd., 1996, vol. 87, pp. 82–87.
G. Cacciamani and R. Ferro: CALPHAD, 2001, vol. 25, pp. 583–97.
A.M. Cardinale, G. Cacciamani, G. Borzone, and R. Ferro: CALPHAD, 2003, vol. 27, pp. 221–26.
G. Cacciamani, A.M. Cardinale, G. Borzone, and R. Ferro: CALPHAD, 2003, vol. 27, pp. 227–33.
K.A. Gschneidner, Jr. and F.W. Calderwood: Bull. Alloy Phase Diagrams, 1988, vol. 9, pp. 669–72.
T. Godecke, W. Sun, R. Luck, and K. Lu: Z. Metallkd., 2001, vol. 92, pp. 723–30.
C. Colinet, A. Pasturel, and K.H.J. Buschow: J. Chem. Thermodyn., 1985, vol. 17, pp. 1133–39.
F. Sommer and M. Keita: J. Less-Common Met., 1987, vol. 136, pp. 95–99.
G. Borzone, G. Cacciamani, and R. Ferro: Metall. Trans. A, 1991, vol. 22A, pp. 2119–23.
G. Borzone, A.M. Cardinale, G. Cacciamani, and R. Ferro: Z. Metallkd., 1993, vol. 84, pp. 635–40.
B. Sundman, B. Jansson, and J.-O. Andersson: CALPHAD, 1985, vol. 9, pp. 153–90.
G. Kresse and J. Hafner: Phys. Rev. B, 1993, vol. 47, pp. 558–61.
G. Kresse and J. Furthmuller: Phys. Rev. B, 1996, vol. 54, pp. 1169–86.
G. Kresse and D. Joubert: Phys. Rev. B, 1999, vol. 59, pp. 1758–75.
J.P. Perdew, K. Burke, and M. Ermzerhoff: Phys. Rev. Lett., 1996, vol. 77, pp. 3865–68.
WWW site http://alloy.phys.cmu.edu (special “published” entries).
M. Mihalkovic and M. Widom: Phys. Rev. B, 2004, vol. 70, pp. 144107–144119.
P. Villars: Pearson’s Handbook, Desk Edition, ASM, Metals Park, OH, 1997.
M. Hillert and L.I. Staffanson: Acta Chem. Scand., 1970, vol. 24, pp. 3618–26.
A.T. Dinsdale: CALPHAD, 1991, vol. 15, pp. 317–425.
O. Redlich and A.T. Kister: Ind. Eng. Chem., 1948, vol. 40, p. 345.
Y.M. Muggianu, M. Gambino, and J.P. Bros: J. Chim. Phys., 1975, vol. 22, pp. 83–88.
T.B. Massalski, H. Okamoto, P.R. Subramanian, and L. Kacprzak: Binary Alloy Phase Diagrams, ASM INTERNATIONAL, Materials Park, OH, 1990.
H. Okamoto: Desk Handbook: Phase Diagrams for Binary Alloys, ASM INTERNATIONAL, Materials Park, OH, 2000.
K. Ozturk, L.Q. Chen, and Z.K. Liu: J. Alloys Compounds, 2002, vol. 340, pp. 199–206.
I. Ansara, T.G. Chart, A. Fernandez Guillermet, F.H. Hayes, U.R. Kattner, D.G. Pettifor, N. Saunders, and K. Zeng: CALPHAD, 1997, vol. 21, pp. 171–218.
M. Zinkevich, N. Mattern, and I. Bacher: Z. Metallkd., 2002, vol. 93, pp. 186–98.
K. Ozturk, Y. Zhong, L.Q. Chen, C. Wolverton, J.O. Sofo, and Z.K. Liu: Metall. Mater. Trans. A, 2005, vol. 36A, pp. 5–13.
L. Kaufman, P.E.A. Turchi, W. Huang, and Z.-K. Liu: CALPHAD, 2001, vol. 25, pp. 419–33.
C. Wolverton: Acta Mater., 2001, vol. 49, pp. 3129–42.
C. Wolverton, X.-Y. Yan, R. Vijayaraghavan, and V. Ozolin: Acta Mater., 2002, vol. 50, pp. 2187–97.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gao, M.C., Ünlü, N., Shiflet, G.J. et al. Reassessment of Al-Ce and Al-Nd binary systems supported by critical experiments and first-principles energy calculations. Metall Mater Trans A 36, 3269–3279 (2005). https://doi.org/10.1007/s11661-005-0001-y
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11661-005-0001-y