Abstract
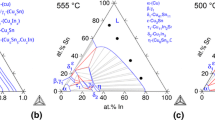
Ternary Sn-Cu-Ni alloys were prepared and annealed at 240°C. The annealed alloys were metallographically examined and the equilibrium phases formed were identified on the basis of compositional determinations and x-ray diffraction (XRD) analysis. The isothermal section of the ternary Sn-Cu-Ni system at 240°C was proposed on the basis of experimental results of this study and related information on phase equilibrium available in the literature. The binary compounds, Cu6Sn5, Ni3Sn2, and Ni3Sn4, have very extensive ternary solubility. Continuous solid solutions form between Cu and Ni as well as between Cu3Sn and Ni3Sn. In addition to the isothermal section, the liquidus projection of the Sn-Cu-Ni system was determined based on results from the existing literature. Interfacial reactions between Sn-Cu alloys and Ni substrate and the primary solidification phases of various Sn-Cu-Ni alloys were also examined in this study.
Similar content being viewed by others
References
M. Abtew and G. Selvaduray, Mater. Sci. Eng. 27, 95 (2000).
J.S. Huang, SMT 15, 60 (2001).
J.T. Eash and C. Upthegrove, Trans. AIME 104, 221 (1933).
B.D. Bastow and D.H. Kirkwood, J. Inst. Met. 99, 277 (1971).
Y. Murakami and S. Kachi, Trans. JIM 24, 9 (1983).
M. Miki and Y. Ogino, Trans. JIM 25, 593 (1984).
E. Wachtel and E. Bayer, Z. Metallkd. 75, 61 (1984).
K.P. Gupta, Ind. Inst. Met. 1, 195 (1990).
D.J. Charkrabarti, D.E. Laughlin, S.-W. Chen, and Y.A. Chang, Ind. Inst. Met. 1, 1442 (1990).
W.T. Chen, C.E. Hoand, and C.R. Kao, J. Mater. Res. 17, 17, 263.
N. Saunders and A.P. Miodownik, Binary Alloy Phase Diagrams, 2nd ed., ed. T.B. Massalski (Materials Park, OH: ASM, 1990), pp. 1481–1483.
P. Nash and A. Nash, Binary Alloy Phase Diagrams, 2nd ed., ed. T.B. Massalski (Materials Park, OH: ASM, 1990), pp. 2863–2864.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lin, CH., Chen, SW. & Wang, CH. Phase equilibria and solidification properties of Sn-Cu-Ni alloys. J. Electron. Mater. 31, 907–915 (2002). https://doi.org/10.1007/s11664-002-0182-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11664-002-0182-8