Abstract
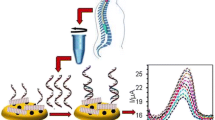
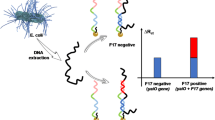
We report an ultrasensitive nanohybrid electrochemical DNA sensor developed to detect the LipL32 gene of Leptospira interrogans that causes leptospirosis. A carboxylated multiwalled carbon nanotubes electrode with gold nanoparticles (c-MWCNTs/nano-Au) was modified with polyamidoamine (PAMAM) and graphene quantum dots using cysteine (Cys) as a linker. The c-MWCNTs/nano-Au electrode was covalently bonded to polyamidoamine (PAMAM) via Cys through Au-SH bonding. The amino group of PAMAM was linked to carboxyl moiety of graphene quantum dots (GQD) to form a c-MWCNTs/nano-Au/Cys/PAMAM/GQD nanohybrid electrode. A 5’-amino (NH2) labeled single-stranded DNA (ssDNA) probe based on the LipL32 gene was immobilized on the surface of a c-MWCNTs/nano-Au/Cys/PAMAM/GQD nanohybrid electrode. The different dilutions of single-stranded genomic DNA (ssG-DNA) of L. interrogans were treated on immobilized DNA probe to allow the hybridization. The hybridization events are measured using cyclic voltammetry (CV), differential pulse voltammetry (DPV), and electrochemical impedance spectroscopy (EIS) using redox indicators of 1 mM methylene blue and potassium ferricyanide. Field emission scanning electron microscopy (FE-SEM) was performed to analyze the modifications done on the surface of the electrode. The developed nanohybrid sensor has a sensitivity of 1952.3 µA cm−2 ng−1 and a lower limit of detection (LOD) of 0.112 pg/µl (R2 = 0.98). The nanohybrid sensor showed good reproducibility over a period of 6 months, exhibiting only a 10% loss in actual DPV current. The developed nanohybrid sensor is found to be specific to L. interrogans with high sensitivity and reproducibility, and it can be used for routine analysis for L. interrogans identification and leptospirosis diagnosis.







Similar content being viewed by others
References
Abad-Valle P, Fernández-Abedul MT, Costa-García A (2005) Genosensor on gold films with enzymatic electrochemical detection of a SARS virus sequence. Biosens Bioelectron 20:2251–2260. https://doi.org/10.1016/j.bios.2004.10.019
Ahmed SN, Shah S, Ahmad FMH (2005) Laboratory diagnosis of leptospirosis. J Postgrad Med 51:195–200
Bahadir EB, Sezginturk MK (2016) Poly (amidoamine) (PAMAM): An emerging material for electrochemical bio (sensing) applications. Talanta 148:427–438. https://doi.org/10.1016/j.talanta.2015.11.022
Bharti AR, Nally JE, Ricaldi JN, Matthias MA, Diaz MM, Lovett MA, Levett PN, Gilmann RH, Willing MR, Gotuzzo E, Vinetz JM, Peru United States Leptospirosis Consortium (2003) Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis 3: 757–771.https://doi.org/10.1016/s1473-3099(03)00830-2
Bini A, Minunni M, Tombelli S, Centi S, Mascini M (2007) Analytical performances of aptamer-based sensing for thrombin detection. Anal Chem 79:3016–3019. https://doi.org/10.1021/ac070096g
Bomfim MK, Barbosa EF, Koury MC (2008) Detection of pathogenic leptospires in urine from naturally infected cattle by nested PCR. Vet J 178:251–256. https://doi.org/10.1016/j.tvjl.2007.07.029
Budihal SV, Perwez K (2014) Leptospirosis diagnosis: competency of various laboratory tests. J Clin Diag Res 8:199–202. https://doi.org/10.7860/jcdr/2014/6593.3950
Chang MK, Cheng YC, Hsu SH, Ma TL, Chou LF, Hsu HH, Tian YC, Chen YC, Sun YJ, Hung CC, Pan RL (2016) Leptospiral outer membrane protein LipL32 induces inflammation and kidney injury in zebrafish larvae. Sci Rep 6:27838. https://doi.org/10.1038/srep27838
Chen F, Gao W, Qiu X, Zhang H, Liu L, Liao P, Fu W, Luo Y (2017) Graphene quantum dots in biomedical applications: Recent advances and future challenges. Front Lab Med 4:192–199. https://doi.org/10.1016/j.flm.2017.12.006
Das R, Sharma MK, Rao VK, Bhattacharya BK, Garg I, Venkatesh V, Upadhyay S (2014) An electrochemical genosensor for Salmonella typhi on gold nanoparticles-mercaptosilane modified screen printed electrode. J Biotechnol 188:9–16. https://doi.org/10.1016/j.jbiotec.2014.08.002
Inbaraj BS, Chen BH (2016) Nanomaterial-based sensors for detection of foodborne bacterial pathogens and toxins as well as pork adulteration in meat products. J Food Drug Anal 24:15–28. https://doi.org/10.1016/j.jfda.2015.05.001
Kaushik R, Chawla P, Kumar N, Janghu S, Lohan A (2018) Effect of premilling treatments on wheat gluten extraction and noodle quality. Food Sci Technol Int 24:627–636. https://doi.org/10.1177/1082013218782368
Khaki P (2016) Clinical laboratory diagnosis of human leptospirosis. Int J Enteric Pathog 4:1–7
Lee KJ, Elgrishi N, Kandemir B, Dempsey JL (2017) Electrochemical and spectroscopic methods for evaluating molecular electrocatalysts. Nat Rev Chem 1:39. https://doi.org/10.3390/asi3030032
Levett PN (2001) Leptospirosis. Clin Microbiol Rev 14:296–326. https://doi.org/10.1128/cmr.14.2.296-326.2001
Liao H, Nehl CL, Hafner JH (2006) Biomedical applications of plasmon resonant metal nanoparticles. Fut Medicine 1:201–208. https://doi.org/10.2217/17435889.1.2.201
Lin XH, Wu P, Chen W, Zhang YF, Xia XH (2007) Electrochemical DNA biosensor for the detection of short DNA species of chronic myelogenous leukemia by using methylene blue. Talanta 72:468–471. https://doi.org/10.1016/j.talanta.2006.11.015
Lin YY, Wang J, Liu G, Wu H, Wai CM, Lin Y (2008) A nanoparticle label/immunochromatographic electrochemical biosensor for rapid and sensitive detection of prostate-specific antigen. Biosens Bioelectron 23:1659–1665. https://doi.org/10.1016/j.bios.2008.01.037
Luka G, Ahmadi A, Najjaran H, Alocilja E, DeRosa M, Wolthers K, Malki A, Aziz H, Althani A, Hoorfar M (2015) Microfluidics integrated biosensors: a leading technology towards lab-on-a-chip and sensing applications. Sensors 15(12):30011–30031. https://doi.org/10.3390/s151229783
Muso D, La Scola B (2013) Laboratory diagnosis of leptospirosis: a challenge. J Microbiol Immunol Infect 46:245–252. https://doi.org/10.1016/j.jmii.2013.03.001
Nagraik R, Kaushal A, Gupta S, Dhar P, Sethi S, Kumar D (2019) Optmized DNA-based bioassay for Leptospira interrogans detection: a novel platform for leptospirosis diagnosis. 3 Biotech 9:284. https://doi.org/10.1007/s13205-019-1815-4
Niloofa R, Fernando N, de Silva NL, Karunanayake L, Wickramasinghe H, Dikmadugoda N, Premawansa G, Wickramasinghe R, de Silva HJ, Premawansa S, Rajapakse S, Hnadunnetti S (2015) Diagnosis of leptospirosis: comparison between microscopic agglutination test, IgM-ELISA and IgM rapid immunochromatography test. PLoS ONE 10:e0129236. https://doi.org/10.1371/journal.pone.0129236
Niu X, Zheng W, Yin C, Weng W, Li G, Sun W, Men Y (2017) Electrochemical DNA biosensor based on gold nanoparticles and partially reduced graphene oxide modified electrode for the detection of Listeria monocytogenes hly gene sequence. J Electroanal Chem 806:116–122. https://doi.org/10.1016/j.jelechem.2017.10.049
Picardeau M (2013) Diagnosis and epidemiology of leptospirosis. Med Mal Infect 43:1–9. https://doi.org/10.1016/j.medmal.2012.11.005
Ramadass P, Samuel B, Nachimuthu K (1999) A rapid latex agglutination test for detection of leptospiral antibodies. Vet Microbiol 70:137–140. https://doi.org/10.1016/s0378-1135(99)00133-9
Rao PS, Sridhar VS (2001) Serological evidence in suspected cases of leptospirosis in and around Manipal. Indian J Med Microbiol 19:32. https://doi.org/10.4103/0255-0857.29410
Rowland CE, Brown CW III, Delehanty JB, Medintz IL (2016) Nanomaterial- based sensors for the detection of biological threat agents. Mater Today 19:464–477. https://doi.org/10.1016/j.mattod.2016.02.018
Scott K (2016) Electrochemical principles and characterization of bioelectrochemical systems. Micro Electrochem Fuel Cells 88:29–66
Sehgal SC (2000) Leptospirosis in the horizon. Natl Med J India 13:228–230. https://doi.org/10.4103/0255-0857.29405
Singh S, Kaushal A, Khare S, Kumar P, Kumar A (2014) Gold-mercaptopropionic acid-polyethylenimine composite based DNA sensor for early detection of rheumatic heart disease. Analyst 139:3600–3606. https://doi.org/10.1039/c4an00324a
Sudhakar YN, Selvakumar M, Bhat DK (2018) Biopolymer Electrolytes: Fundamentals and Applications in Energy Storage. Elsevier, Amsterdam, The Netherlands
Tlili C, Korri-Youssoufi H, Ponsonnet L, Martelet C, Jaffrezic-Renault NJ (2005) Electrochemical impedance probing of DNA hybridization on oligonucleotide-functionalized polypyrrole. Talanta 68:131–137. https://doi.org/10.1016/j.msec.2007.10.061
Vijayachari P, Sehgal SC (2006) Recent advances in the laboratory diagnosis of leptospirosis and characterisation of leptospires. Indian J Med Microbiol 24:320–322
Vijayachari P, Sugunan AP, Shriram AN (2008) Leptospirosis: an emerging global public health problem. J Biosci 33:557–569. https://doi.org/10.1007/s12038-008-0074-z
Wang J (2002) Electrochemical nucleic acid biosensors. Anal Chim Acta 469:63–71. https://doi.org/10.1016/s0003-2670(01)01399-x
Wang J (2005) Nanomaterial-based electrochemical biosensors. Analyst 130:421–426. https://doi.org/10.1039/b414248a
Wasinski B, Pejsak Z (2010) Occurrence of leptospiral infections in swine population in Poland evaluated by ELISA and microscopic agglutination test. J Vet Sci 13:695–699. https://doi.org/10.2478/v10181-010-0006-3
Zhang J, Dong S, Lu J, Turner APF (2009) Label free electrochemical nanobiosensor study. Anal Letters 42:2905–2913. https://doi.org/10.1080/00032710903201941
Acknowledgements
The authors would like to thank Prof. P.K. Khosla (Vice Chancellor, Shoolini University, Solan, H.P.) and PGIMER (Chandigarh) for allowing the current study to conduct and complete.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Ethical approval
The work did not involve any human/animal subjects.
Informed consent
Informed consent was taken from all the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nagraik, R., Sethi, S., Sharma, A. et al. Ultrasensitive nanohybrid electrochemical sensor to detect LipL32 gene of Leptospira interrogans. Chem. Pap. 75, 5453–5462 (2021). https://doi.org/10.1007/s11696-021-01737-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01737-1